
Plasma zinc status in critically ill patients with chronic liver disease
Introduction
Zinc is an essential trace element in the body and has crucial roles in numerous enzymatic and metabolic processes, including gene transcription, immunoregulation, cellular restoration and protein synthesis (1). The liver plays an important role in zinc homeostasis. Zinc levels are abnormally decreased in chronic liver disease (CLD) and may lead to metabolic complications such as hepatic encephalopathy (2). Hypozincaemia is observed in the critically ill and is associated with increased mortality and illness severity scores (3). Zinc deficiency in CLD patients may increase their susceptibility to endotoxaemia by increasing intestinal barrier permeability via oxidative stress pathways (4). Epithelial dysfunction causing intestinal hyperpermeability with alteration of intestinal microbiota is also described in critical illness. This leads to local and distant tissue injury resulting in multiple organ dysfunction syndrome (5).
Plasma zinc levels in critically ill patients with a background of CLD have not been well defined. Therefore, we conducted a prospective study to assess the plasma zinc levels in CLD patients requiring intensive care unit (ICU) admission and compared them with concurrent critically ill patients without CLD. We also evaluated the association of plasma zinc levels and outcomes in these patients.
We present the following article in accordance with the STROBE reporting checklist (available at https://dx.doi.org/10.21037/jeccm-21-48).
Methods
Subjects
Sixty-nine critically ill patients with CLD formed the CI-CLD group, whereas 73 critically ill controls without a background of CLD formed the CI-CONTROL group. The study was conducted between November 2011 and December 2015 across two university hospital ICUs. Patients with established CLD were enrolled on ICU admission. Controls were matched for age, gender, admission diagnosis and organ support required; admitted to ICU concurrently without any history of CLD or significant alcohol intake. Patients with end stage renal failure on regular haemodialysis, receiving parenteral nutrition and with a history of receiving radiologic contrast in the previous 4 days were excluded from the study. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Hospital Human Research and Ethics Committee of The Queen Elizabeth Hospital (ethical approval ID number 2011121). Informed consent was taken from all the patients.
Data collection and laboratory analysis
Patient demographic data such as age and gender, admission diagnosis, physiologic parameters and clinical characteristics were collected at time of admission. Acute Physiology and Chronic Health Evaluation III (APACHE-III) and Sequential Organ Failure Assessment (SOFA) scores were recorded. Severity of liver disease was evaluated using the Model For End Stage Liver Disease (MELD) score which utilises serum creatinine, bilirubin and prothrombin time to predict survival (6).
In all subjects, blood samples were collected at admission to examine haemoglobin, white cell count, and platelet count, coagulation profile including international normalised ratio (INR), C-reactive protein, liver functions and renal function. Zinc levels were collected during ICU admission on inclusion in the study. Blood for zinc levels were collected in metal-free vacutainer tubes, at the same time point (0600) in all patients. Plasma zinc was measured by mass spectrometry which has a validated normal range (9.02–11.32 µmol/L) (7). Twenty-four-hour urinary zinc levels were measured in a subgroup of patients with normal renal function in both groups (36% of CI-CLD patients, 33% of CI-CONTROLs). Urine was collected in a specialised metal-free container with 50 mL dilute nitric acid. All patients were followed up for the length of stay (LOS) in ICU and hospital as well as for mortality in ICU, in-hospital and at 6 months post-discharge, using hospital patient information systems.
Statistical analysis
The categorical variables are presented as absolute frequency and percentages, and the continuous variables are presented as mean ± standard deviation (SD). The clinical characteristics of the two groups; CI-CLD and CI-CONTROL, were compared using the Pearson’s chi-squared test or Fisher’s exact test for categorical variables and Student’s t-test for continuous variables.
Survival was estimated using the Kaplan-Meier method, and differences in survival between groups were assessed using the log-rank test.
Cox proportional-hazard regression analysis was used to evaluate the association between variables and mortality, with an adjusted P value <0.05 considered statistically significant. The hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated. Within the Cox model the explained variation (R2) was assessed using the user-written Stata™ module “str2d”, with 95% CI determined by the bootstrap procedure (1,000 samples) (8). Variable collinearity was evaluated by the condition number (in non-linear models, values >15) and the correlation between variables (rho >0.8). Non-linearity of continuous covariate effect was determined using multivariate fractional polynomials. Model development was guided by clinical expert opinion, progressive reduction of Akaike Information Criteria and Bayesian Information Criteria.
All statistical analyses were performed using Stata statistical software version 14.2 (College Station, TX 2015) and MedCalc Statistical Software version 12.7.7 (MedCalc Software, Ostend, Belgium)
Results
Baseline characteristics and parameters in study population of CI-CLD and CI-CONTROL patients
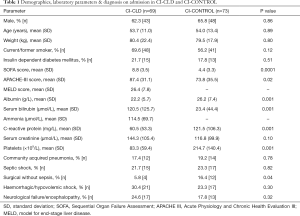
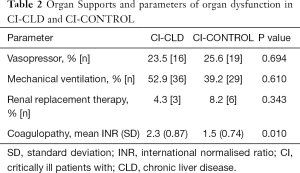
Patient demographics and admission diagnoses were well matched (Table 1). APACHE-III and SOFA scores were significantly higher in the CLD group, reflected by high mean MELD scores of 26.4±7.8 (Table 1). Hypoalbuminaemia and hyperammonaemia were noted in the CI-CLD group. Serum creatinine was not different between the groups (Table 1). The requirement of organ support with vasopressors, mechanical ventilation and renal replacement therapy was similar in both study groups. Predictably, coagulopathy was statistically significant in the CI-CLD group as compared to CI-CONTROL (mean INR 2.3 vs. 1.5, P=0.010) (Table 2).
Full table
Full table
Decreased plasma zinc levels in CI-CLD patients
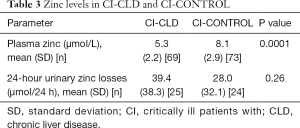
Plasma zinc concentrations in CI-CLD patients were significantly lower than CI-CONTROLs (CI-CLD vs. CI-CONTROLs, 5.3±2.2 vs. 8.1±2.9 µmol/L, P=0.0001) (Table 3). Notably, patients in the CI-CONTROL group had zinc levels either low or at the lower limit of normality. Time to collection of zinc levels from ICU admission varied from 0 to 30 days [P50 2 days, interquartile range (IQR) 4 days]. There was a time relationship of zinc assay (β coefficient 0.156, P=0.003), but the explained variance (R2) of plasma zinc levels by time to assay was small at 0.06.
Full table
Twenty-four-hour urinary zinc losses
Twenty-four-hour urinary zinc was analysed in 1/3 of the study population (n=49). Urinary zinc losses were higher but statistically non-significant in the CI-CLD group as compared to CI-CONTROL (39.4±38.3 vs. 28.0±32.1 µmol/24 h, P=0.26) (Table 3).
Association of plasma zinc concentrations with mortality
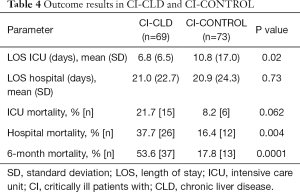
Our data demonstrated a significant increase in the unconditional mortality rate associated with hypozincaemia across the domains of ICU (21.7% vs. 8.2%, P=0.062), hospital (37.7% vs. 16.4%, P=0.004) in the CI-CLD group compared to CI-CONTROL and at 6 months follow up (53.6% vs. 17.8%, P=0.0001) (Table 4).
Full table
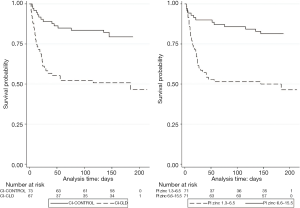
Kaplan-Meier survival estimates and numbers at risk, by CLD and median split (6.55 µmol) of plasma zinc levels are shown in Figure 1. The adverse effect of low plasma zinc levels was consistent for both patient groups as CI-CLD (median 5.15 µmol/L) log-rank P=0.038 and CI-CONTROL (median 7.65 µmol/L) log-rank P=0.048.
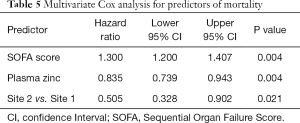
A multivariate Cox model identified SOFA score, plasma zinc levels and site as predictors of 6-month survival (Table 5). We found that per unit increment in SOFA score increased the risk of mortality by 30% (HR 1.300, 95% CI: 1.200, 1.407; P=0.004). Per μmol/L increment in plasma zinc levels reduced mortality by 17% (HR 0.835, 95% CI: 0.739, 0.943; P=0.004). Patients at Site 2 had a 50% less chance of mortality than patients in Site 1 (HR 0.505, 95% CI: 0.328, 0.902; P=0.021). Model R2 was high at 0.567 (95% CI: 0.447, 0.702) suggesting a good data fit for the model. Gender, age, weight, smoking status, diabetes, time to zinc assay and APACHE-III score were non-significant predictors (P≥0.33). There was no effect of time to collection of zinc levels as a time varying covariate within the Cox model (P=0.86). The binary variable denoting the presence or absence of CLD (univariate CLD HR 3.275, 95% CI: 1.786, 6.006; P=0.001) failed to achieve significance in the multivariate model (P=0.267).
Full table
Discussion
This prospective study demonstrates quantitatively severe hypozincaemia in CI-CLD compared to low levels in the CI-CONTROL group. While the existing body of literature acknowledges the existence of hypozincaemia in CLD (2,9) and critical illness (2,10), there is scarcity of literature describing the status of zinc levels when these two conditions coexist.
Zinc deficiency is common in CLD, reported in up to 83% of patients (9). Normally, zinc is absorbed in the small intestine, 11% being stored in the liver and skin and is excreted by the kidneys. Zinc transporters play a key role in absorption, excretion and transport of zinc (4,11). Patients with CLD have decreased zinc absorption, altered zinc transport, decreased hepatic content of zinc and increased urinary zinc excretion (9).
Hypozincaemia, as observed in CI-CONTROL, is noted in 95% of ICU patients (12). This is partly due to increased tissue sequestration of zinc as an acute phase response in critical illness and is associated with reduced survival (9). In an experimental murine model of polymicrobial sepsis, subacute zinc deficiency was associated with increased systemic inflammation, organ damage and mortality (13). Suboptimal zinc concentrations in critical illness with increased oxidative stress and inflammatory biomarkers may contribute to damage of crucial proteins (14).
Chronic zinc deficiency in CLD combined with altered zinc homeostasis during acute illness may explain the significant difference in plasma zinc levels observed between the two study groups.
Urinary zinc excretion is normally inhibited in states of acute severe dietary zinc deficiency (15). Contrarily, urinary zinc levels were high in both study groups. Tissue zinc redistribution, hypoalbuminaemia and diuretic use (all common in both critical illness and CLD) result in increased urinary zinc losses irrespective of its intake (16,17). In hypoalbuminaemia, unbound zinc binds to amino acids such as alpha-2-macroglobulin (itself an acute phase reactant) with increased affinity to zinc, leading to increased zinc excretion (18). This association is so compelling that hypoalbuminaemia is used as a surrogate marker of underlying zinc deficiency in CLD (19).
We unsurprisingly observed hyperammonaemia in the CI-CLD group (114.5±69.7 µmol/L, reference range <50 µmol/L) (20). Zinc deficiency has been linked with altered ammonia metabolism since 1978, when Prasad et al. unexpectedly discovered hyperammonaemia in volunteers who consumed a zinc-free diet (21). Zinc deficiency reduces hepatic ornithine transcarboxylase activity, a crucial enzyme in the urea cycle required for the metabolism of ammonia. Randomised control trial data had demonstrated the effect of zinc supplementation in reducing ammonia levels in patients with CLD (22).
Hyperbilirubinaemia, as noted in our study, had previously shown a negative correlation with plasma zinc concentration in CLD (19). Zinc inhibits the enterohepatic circulation of bilirubin, leading to higher total circulating bilirubin levels in zinc deficiency (23). Thus, zinc supplementation is used to treat hyperbilirubinemia in specific high-risk patient groups (24). Hyperbilirubinemia is also common in critical illness and linked to increased mortality despite a lack of explanation as to its mechanism (25). Vanwijngaerden et al. reported the absence of association between cholestasis and inflammation in critically ill patients while confirming the strong association between the pro-inflammatory state of critical illness with hyperbilirubinemia, most likely due to altered handling of bile acids (26) which has been associated with zinc deficiency (24).
Declining plasma zinc levels were previously associated with increased illness severity scores in severe infections and increased mortality in ICU (27,28). It concurs with our findings of SOFA score and zinc levels as predictors of survival at 6 months. The observed association of hypozincaemia with increased mortality may suggest its prognostic value. Disruption of zinc’s critical role in antioxidant defence, DNA repair and immunoregulation in CLD patients (29), further worsened by critical illness may explain reduced survival in CI-CLD group. The study site difference identified in predictability of outcome was presumably related to our small study population and case-mix.
There is an established body of literature outlining the value of zinc in maintaining homeostasis, as well as the harmful sequelae of zinc deficiency (8,11,13,14). However, the effect of zinc supplementation in CI-CLD patients remains unclear and has not been investigated. Current guidelines do not make any specific recommendation regarding zinc supplementation. A systematic review by Diglio et al. concluded that zinc supplementation in CLD did not show benefit in clinical improvement or halting of disease progression, except possible benefit in hepatic encephalopathy (22,30). Similarly, supplemental antioxidants in critically ill mechanically ventilated patients with multisystem organ failure did not improve clinical outcome (31).
Although to our knowledge, this study is the first to assess the status of zinc levels in CLD patients with acute critical illness, it has some limitations. Only 1/3 of patients had urinary zinc analysis, so the results cannot be generalised. Ammonia levels were assessed only in patients with hepatic pathology. Critically ill populations may possess risk factors for non-hepatic causes for hyperammonaemia (32). Our small sample size, more recruitment of CI-CONTROL from one site and differences in case-mix may have affected the study outcome. Zinc levels were not repeated on discharge from ICU to ascertain if levels showed any improvement upon resolution of acute illness. It is possible that CLD patients predisposed to chronically low zinc levels may drop their levels further during an acute episode, while controls may have normal baseline zinc levels and merely drop acutely during illness.
In conclusion, this study demonstrates significantly low plasma zinc levels in acutely ill patients with CLD compared to hypozincaemia seen in other critically ill patients. Our data identifies the association of hypozincaemia with increased ICU, hospital and 6-month mortality. A large clinical trial is required to evaluate role of monitoring zinc levels and maintenance of normal zinc levels with supplementation in CI-CLD patients.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://dx.doi.org/10.21037/jeccm-21-48
Data Sharing Statement: Available at https://dx.doi.org/10.21037/jeccm-21-48
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://dx.doi.org/10.21037/jeccm-21-48). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Hospital Human Research and Ethics Committee of The Queen Elizabeth Hospital (Ethical Approval ID Number 2011121). Informed consent was taken from all the patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Vallee BL, Falchuk KH. The biochemical basis of zinc physiology. Physiol Rev 1993;73:79-118. [Crossref] [PubMed]
- Himoto T, Masaki T. Associations between Zinc Deficiency and Metabolic Abnormalities in Patients with Chronic Liver Disease. Nutrients 2018;10:88. [Crossref] [PubMed]
- Cander B, Dundar ZD, Gul M, et al. Prognostic value of serum zinc levels in critically ill patients. J Crit Care 2011;26:42-6. [Crossref] [PubMed]
- Grüngreiff K, Reinhold D, Wedemeyer H. The role of zinc in liver cirrhosis. Ann Hepatol 2016;15:7-16. [Crossref] [PubMed]
- Otani S, Coopersmith CM. Gut integrity in critical illness. J Intensive Care 2019;7:17. [Crossref] [PubMed]
- Kamath PS, Wiesner RH, Malinchoc M, et al. A model to predict survival in patients with end-stage liver disease. Hepatology 2001;33:464-70. [Crossref] [PubMed]
- International Zinc Nutrition Consultative Group (IZiNCG). International Zinc Nutrition Consultative Group (IZiNCG) technical document #1. Assessment of the risk of zinc deficiency in populations and options for its control. Food Nutr Bull 2004;25:S99-203. [PubMed]
- Royston P. Explained variation for survival models. Stata Journal 2006;6:83-96. [Crossref]
- Koop AH, Mousa OY, Pham LE, et al. An Argument for Vitamin D, A, and Zinc Monitoring in Cirrhosis. Ann Hepatol 2018;17:920-32. [Crossref] [PubMed]
- Lee YH, Bang ES, Lee JH, et al. Serum Concentrations of Trace Elements Zinc, Copper, Selenium, and Manganese in Critically Ill Patients. Biol Trace Elem Res 2019;188:316-25. [Crossref] [PubMed]
- Cousins RJ. Gastrointestinal factors influencing zinc absorption and homeostasis. Int J Vitam Nutr Res 2010;80:243-8. [Crossref] [PubMed]
- Besecker BY, Exline MC, J, Hollyfield G, et al. A comparison of zinc metabolism, inflammation, and disease severity in critically ill infected and noninfected adults early after intensive care unit admission Am J Clin Nutr 2011;93:1356-64. [Crossref] [PubMed]
- Knoell DL, Julian MW, Bao S, et al. Zinc deficiency increases organ damage and mortality in a murine model of polymicrobial sepsis. Crit Care Med 2009;37:1380-8. [Crossref] [PubMed]
- Mertens K, Lowes DA, Webster NR, et al. Low zinc and selenium concentrations in sepsis are associated with oxidative damage and inflammation. Br J Anaesth 2015;114:990-9. [Crossref] [PubMed]
- King JC, Shames DM, Woodhouse LR. Zinc homeostasis in humans. J Nutr 2000;130:1360S-6S. [Crossref] [PubMed]
- Boosalis MG, Solem LD, Cerra FB, et al. Increased urinary zinc excretion after thermal injury. J Lab Clin Med 1991;118:538-45. [PubMed]
- Vincent JL, Dubois MJ, Navickis RJ, et al. Hypoalbuminemia in acute illness: is there a rationale for intervention? A meta-analysis of cohort studies and controlled trials. Ann Surg 2003;237:319-34. [Crossref] [PubMed]
- Schechter PJ, Giroux EL, Schlienger JL, et al. Distribution of serum zinc between albumin and alpha-2-macroglobulin in patients with decompensated hepatic cirrhosis. Eur J Clin Investig 1976;31:147-50. [Crossref]
- Katayama K, Kawaguchi T, Shiraishi K, et al. The prevalence and implication of zinc deficiency in patients with chronic liver disease. J Clin Med Res 2018;10:437-44. [Crossref] [PubMed]
- Burton BK. Urea cycle disorders. Clin Liver Dis 2000;4:815-30. [Crossref] [PubMed]
- Prasad AS, Rabbani P, Abbasii A, et al. Experimental zinc deficiency in humans. Ann Intern Med 1978;89:483-90. [Crossref] [PubMed]
- Katayama K, Saito M, Kawaguchi T, et al. Effect of zinc on liver cirrhosis with hyperammonemia: A preliminary randomized, placebo controlled double-blind trial. Nutrition 2014;30:1409-14. [Crossref] [PubMed]
- Méndez-Sánchez N, Roldán-Valadez E, Flores MA, et al. Zinc salts precipitate unconjugated bilirubin in vitro and inhibit enterohepatic cycling of bilirubin in hamsters. Eur J Clin Invest 2001;31:773-80. [Crossref] [PubMed]
- Méndez-Sánchez N, Martínez M, González V, et al. Zinc sulfate inhibits the enterohepatic cycling of unconjugated bilirubin in subjects with Gilbert's syndrome. Ann Hepatol 2002;1:40-3. [Crossref] [PubMed]
- Kramer L, Jordan B, Druml W, et al. Incidence and prognosis of early hepatic dysfunction in critically ill patients - a prospective multicenter study. Crit Care Med 2007;35:1099-104. [Crossref] [PubMed]
- Vanwijngaerden YM, Wauters J, Langouche L, et al. Critical illness evokes elevated circulating bile acids related to altered hepatic transporter and nuclear receptor expression. Hepatology 2011;54:1741-52. [Crossref] [PubMed]
- Hoeger J, Simon TP, Beeker T. Persistent low serum zinc is associated with recurrent sepsis in critically ill patients - A pilot study. PLoS One 2017;12:e0176069 [Crossref] [PubMed]
- Florea D, Molina-López J, Hogstrand C, et al. Changes in zinc status and zinc transporters expression in whole blood of patients with Systemic Inflammatory Response Syndrome (SIRS). J Trace Elem Med Biol 2018;49:202-9. [Crossref] [PubMed]
- Katayama K. Zinc and protein metabolism in chronic liver diseases. Nutr Res 2020;74:1-9. [Crossref] [PubMed]
- Diglio DC, Fernandes SA, Stein J, et al. Role of zinc supplementation in the management of chronic liver diseases: A systematic review and meta-analysis. Ann Hepatol 2020;19:190-6. [Crossref] [PubMed]
- van Zanten AR, Hofman Z, Heyland DK. Consequences of the REDOXS and METAPLUS Trials. JPEN J Parenter Enteral Nutr 2015;39:890-2. [Crossref] [PubMed]
- Prado FA, Delfino VD, Grion CM, et al. Hyperammonemia in ICU patients: a frequent finding associated with high mortality. J Hepatol 2015;62:1216-8. [Crossref] [PubMed]
Cite this article as: Subramaniam S, Jacobs S, Moran J, Kanhere M. Plasma zinc status in critically ill patients with chronic liver disease. J Emerg Crit Care Med 2021;5:23.


