
Generalizability of pediatric major trauma experience to severe pediatric traumatic brain injury at level 1 and 2 trauma centers
Introduction
Among the pediatric population, traumatic brain injury (TBI) is the number one cause of hospitalization, death, or disability (1). With more than 6,000 deaths, 50,000 hospitalizations, and 600,000 emergency department visits occurring annually, pediatric TBI contributes to over $1 billion of hospital charges each year (2,3). The ongoing, significant burden of moderate and severe pediatric TBI compels the study of factors that may influence outcomes (4). Several studies have focused on the relationship between trauma center designation and outcomes for adult TBI, finding more favorable outcomes at Level I centers, including improvements in complication rates, mortality, and higher odds of favorable discharge (5-7). The association between patient volume and outcomes is also well-established across a variety of specialties (8-10) and has prompted the study of regionalization practices for a variety of procedures (11,12).
Notably, prior studies of neurosurgical and trauma volume-outcome relationships (VORs) assessed volume in different ways, demonstrating that higher pediatric trauma volume, adult TBI volume, and adult trauma volume are correlated with improved outcomes in their respective domains (8,13-15). For example, Tepas et al. found that higher adult TBI volume was associated with improved outcomes for adult TBI patients (14). Despite broad evidence for the VOR for TBI in adults (13,14), the impact of hospital volume on treatment outcomes of severe pediatric TBI patients has yet to be elucidated. Moreover, because prior VOR assessments typically examined only a single volume metric, no study has taken the important step of directly comparing multiple volume metrics to determine which might be more or less relevant to outcomes. This is particularly important in pediatric neurotrauma care because an institution’s broader and often more voluminous experience with adult trauma might inform or influence its approach to pediatric cases. Furthermore, experience with general (non-TBI) trauma may also be relevant to pediatric TBI care, particularly for the management of polytrauma patients. One of our primary goals, therefore, was to understand what aspects of trauma experience may contribute to management and outcomes in severe pediatric TBI. Specifically, we tested the impact of four distinct volume variables on severe pediatric TBI outcomes.
In this study, we used the largest U.S. trauma registry, the National Trauma Databank (NTDB), to examine the VOR in the pediatric population. Because treatment at American College of Surgeons (ACS) Level I and II pediatric trauma centers has been shown to improve outcomes and may be a significant confounder due to the requirements for ACS accreditation (16), we restricted our analysis to Level I and II pediatric ACS-designated facilities. We hypothesized that, given the relatively smaller numbers of severe pediatric TBI cases, the VOR for pediatric TBI might depend to a greater extent on broader institutional trauma experience. Our objectives were (I) to establish whether volume—rather than trauma center level and resource availability—was related to severe pediatric TBI short-term outcomes and (II) to analyze the influence of different measures of volume on these outcomes. We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/jeccm-21-24).
Methods
Study population
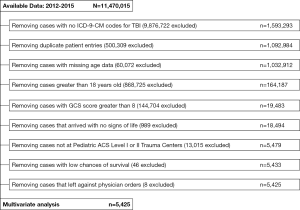
This study was conducted using data from the 2012‒2015 National Trauma Data Bank (NTDB) Research Data Set. The NTDB is managed and compiled by the ACS Committee on Trauma (17). The study population was composed of patients between ages 0 and 18, who were treated for severe TBI at trauma centers with a pediatric ACS level designation of I or II. Only patients presenting to these institutions were included to minimize inter-institutional heterogeneity in resource and personnel availability. However, a subsequent sensitivity analysis was also conducted to examine the effects of volume across all institutions. TBI patients were identified using International Classification of Diseases, 9th Revision Clinical Modification codes (ICD-9-CM) corresponding to the following injury types: fractures of the skull (800.0‒801.9, 803.0‒804.9), intracranial injuries (850.0‒854.1), “shaken baby syndrome” (995.55), and other unspecified head injury (959.01). Following earlier research and guidelines (18), severe TBI was defined as having a total Glasgow Coma Score (GCS) of 3‒8 upon admission to the hospital. Patients with an unknown age were excluded. These criteria make the inclusion of minor TBI cases unlikely. Accordingly, the average ICU and hospital length of stay (LOS) were 7.07 and 11.42 days, indicating that patients required substantial treatment. Due to the absence of a “Dead Upon Arrival” variable in the NTDB, we removed patients with a low chance of survival by following a similar method as Alarhayem et al. (19), eliminating patients who exhibited no signs of life (admission systolic blood pressure =0 and pulse rate =0) and no neurological activity (GCS =3) (Figure 1).
Demographic and clinical data including sex, age, race, Injury Severity Score derived from hospital-submitted Abbreviated Injury Scores (ISSAIS score), comorbidities present, injury type (blunt vs. penetrating vs. other/unspecified), procedures done on the first day of admission, presence of complications, mortality, LOS in the ICU, total LOS in the hospital, and hospital discharge disposition were isolated for each patient. Only comorbidities and complications that were present in at least 1% of the patient population were investigated, in order to reduce the risk of model overfitting. Comorbidities included congenital anomalies (1%) and respiratory disease (3.3%). Complications included cardiac arrest (4.3%), deep vein thrombosis (1.8%), cerebrovascular accident (1.1%), pneumonia (6.5%), urinary tract infection (1.8%), acute respiratory distress syndrome (3.3%), unplanned return to the ICU (1.1%), and unplanned return to the OR (1.0%). Only patients who developed these conditions after initial presentation were counted as having a complication. Procedures done on the first day of admission were condensed into a single variable which represented whether the patient had any of the following procedures performed within their first 24 hours in the hospital: craniotomy or craniectomy, ICP monitoring, transfusions for coagulopathy (transfusion of platelets or coagulation factors), other transfusions (transfusion of PRBCs, whole blood, blood expander, exchange transfusion, other uncategorized transfusions), and ventriculostomy. In order to assess inter-hospital differences in treatment patterns for severe pediatric TBI influenced by hospital volume, we also investigated each procedure as a separate outcome variable, which denoted its occurrence at any point during the patient’s hospital stay. Following a similar method as Dams-O'Connor et al. (6), the native hospital discharge disposition variable within the NTDB was dichotomized as a binary variable into favorable (routine discharge, discharged home without home services, discharge home with home services, discharged home under care of organized home service, transfer to a long-term care facility, transfer to an intermediate care facility, transfer to a skilled nursing facility, transferred to rehab) or unfavorable (death or hospice). Patients who left against medical advice or whose outcomes were unknown were excluded from the study population. For each patient, facility data including hospital teaching status (community or university), region of the hospital, adult major trauma volume, adult severe TBI volume, pediatric major trauma volume, and pediatric severe TBI volume were also obtained from the NTDB.
Volume variables
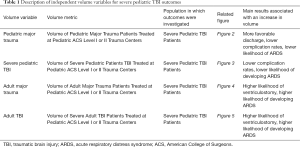
The VOR was investigated using four different independent variables: pediatric severe TBI volume, adult severe TBI volume, pediatric major trauma volume, and adult major trauma volume (Table 1). For each of the four models, we considered the cumulative volume of each pediatric ACS Level I or II trauma center that fit the inclusion criteria to account for longitudinal changes in volume for an institution across the study period. We derived the volume variables from the 2012–2015 NTDB datasets. Severe pediatric TBI volume was computed as the number of patients admitted to an institution who were between 0–18 years old, had ICD-9-CM diagnosis codes corresponding to TBI, and had an admission GCS score between 3–8. We used similar criteria for adult severe TBI volume, but only included patients older than 18 years in the volume calculation. We identified pediatric major trauma volume as the number of cases recorded in the NTDB, for a given hospital, where the patient was 18 years or younger and had an ISS score greater than or equal to 15 (20). Similarly, we calculated adult major trauma volume by only considering NTDB admissions older than 18 years with ISS score of at least 15. These criteria were only used to calculate the volume variables. The study population for each of the four distinct volume models, identified by the severe pediatric TBI criteria, remained the same (Table 1).
Full table
Statistical analysis
Four separate analyses based on adult major trauma volume, pediatric major trauma volume, adult severe TBI volume, and pediatric severe TBI volume were conducted. Each independent volume variable was analyzed on a continuous scale. Combined pediatric and adult centers and stand-alone pediatric institutions were treated together. Outcome variables included mortality, ICU LOS, total hospital LOS, discharge disposition status, presence of any complication, presence of each complication subtype, performance of each procedure, and performance of any procedure on the first day of admission. We analyzed discharge disposition as our primary outcome variable and controlled for patient demographics, hospital characteristics, ISS score, GCS score, comorbidities, and field-to-hospital transport time.
Ethics
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was exempt from Institutional Board Review due to the publicly available and de-identified nature of the data in the NTDB. The National Trauma Data Bank is available to researchers in all partner institutions that contribute data to the dataset. Ethical review and approval were not required for this study on human participants in accordance with the local legislation and institutional requirements. Written informed consent from the patients was not required to participate in this study in accordance with the national legislation and the institutional requirements. These data are reported according to the STROBE guidelines.
Results
Study population
There were 5,425 severe pediatric TBI admissions to 92 unique Pediatric ACS Level I or II trauma centers from 2012–2015 (Figure S1). Patients were an average of 9.5 years old [standard deviation (SD)=6.0, range, 1 month to 18 years], and the majority of cases were white (59.5%) and male (65.1%). Injuries were most commonly blunt (72.9%), with an average GCS admission score of 4.2 (SD= 1.8, pre-defined range, 3–8) and an average injury severity score of 23.0 (SD= 13.7, range, 1–75). Additionally, most patients were admitted to university hospitals (75.6%), and most cases occurred in the Midwest (33.4%) or the South (40.0%; Table 2).

Full table
Outcomes: overview of outcomes for the four volume analyses
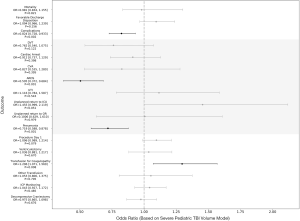
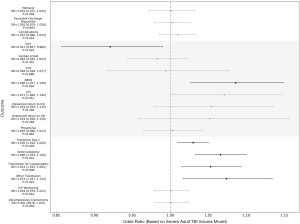
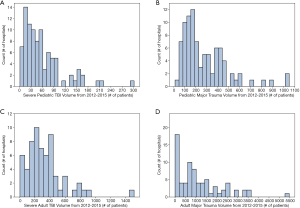
Following multivariate adjustment, only pediatric major trauma volume significantly impacted discharge disposition for severe pediatric TBI patients [odds ratio (OR) = 1.073 per +100 patients, P<0.001; Figure 2]. Treatment at centers with greater severe pediatric TBI and pediatric major trauma volume was associated with lower complication rates (Severe pediatric TBI: OR =0.824 per +100 patients, P=0.002; Pediatric major trauma: OR =0.956 per +100 patients, P=0.018; Figures 2,3). Complication subtype analysis revealed that the likelihood of developing ARDS, specifically, was significantly reduced at these institutions (Severe pediatric TBI: OR =0.505 per +100 patients, P<0.001; Pediatric major trauma: OR =0.851 per +100 patients, P<0.001). Analysis based on the adult TBI and adult major trauma volume variables demonstrated no difference in discharge disposition for severe pediatric TBI patients based on adult volume (Severe adult TBI: OR =1.002 per +100 patients, P=0.864; Adult major trauma: OR =0.998 per +100 patients, P=0.553). However, higher adult volume was associated with a greater likelihood of receiving a ventriculostomy on the first day of admission (Severe adult TBI: OR =1.066 per +100 patients, P<0.001; Adult major trauma: OR =1.018 per +100 patients, P<0.001) and developing ARDS (Severe adult TBI: OR =1.086 per +100 patients, P=0.004; Adult major trauma: OR =1.024 per +100 patients, P=0.002; Figure 4-5). Interestingly, the majority of institutions experienced lower volumes of each neurotrauma type (Figure 6A,B,C,D).
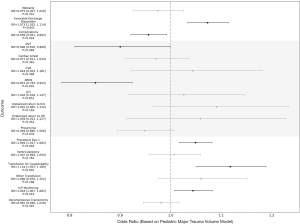
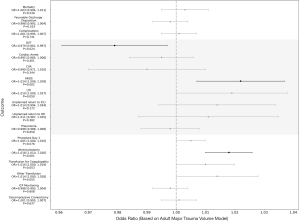
Severe pediatric TBI outcomes related to pediatric major trauma volume
For the period 2012–2015, the highest volume center treated 1,049 pediatric major trauma patients, while the lowest volume center admitted none (Figure 6B). Hospitals with higher pediatric major trauma volume also saw more severe cases, as measured by Injury Severity Score (+0.03 per +100 patients, P<0.001). Additionally, lower volume centers contributed most to the variability in outcomes (Figure S2). Following multivariate adjustment, treatment at hospitals with higher pediatric major trauma volume was associated with higher odds of favorable discharge (OR =1.073 per +100 patients, P<0.001; Figure 2, Figure 7). A subsequent sensitivity analysis demonstrated that this significant relationship was strengthened when patients treated at all trauma center types were included (OR =1.109 per +100 patients, P<0.001; Figure S3). Greater pediatric major trauma volume was also correlated with shorter hospital (‒0.21 days per +100 patients, P=0.035) and ICU LOS (‒0.16 days per +100 patients, P=0.011), and a lower risk of complications (OR =0.956 per +100 patients, P=0.018), particularly ARDS (OR =0.851 per +100 patients, P<0.001; Figure 2). To account for the possibility that patients who initially presented with pulmonary injury may have been more likely to develop ARDS, and that the presence of this factor could interact with trauma and TBI experience more generally, we repeated the analysis while controlling for pulmonary injury at presentation. In this model, patients treated at higher pediatric major trauma volume hospitals remained less likely to develop ARDS (OR =0.850 per +100 patients, P<0.001). Greater volume was also associated with a higher likelihood of receiving a transfusion for coagulopathy within the first 24 hours (OR =1.118 per +100 patients, P<0.001) and ICP monitoring (OR =1.044 per +100 patients, P=0.019). However, pediatric major trauma volume did not significantly impact mortality (OR =0.975 per +100 patients, P=0.331). Consistent with these results, after running an identical analysis that was not restricted to pediatric ACS level I and II trauma centers, mortality was not significantly decreased at higher pediatric trauma volume institutions (P=0.056; Figure S3).
Discussion
Pediatric major general trauma experience informs pediatric severe TBI outcomes
In this study, we found more favorable discharge dispositions for severe pediatric TBI patients at hospitals with higher pediatric major general trauma volume exclusively. However, both pediatric severe TBI and major trauma experience were correlated with lower complication rates, particularly for ARDS. Analyses based on adult volume demonstrated no significant effect of adult major trauma or TBI experience on outcomes for severe pediatric TBI patients. VORs have been established for neurosurgery, trauma (21-23), and adult TBI (13,14). Additionally, the difference in outcomes between pediatric-ACS designated and non-pediatric centers, as well as Level I vs. Level II institutions has already been described. Therefore, rather than focusing on center designation or resource availability, we investigated whether volume influences outcomes in the pediatric severe TBI population at similarly-designated centers. Level I and Level II centers typically have a high level of expertise and meet a set of stringent requirements for certification. Thus, similar to previous research performed for the general trauma population (8), limiting our analysis to these hospitals was particularly informative about the beneficial impact of volume at even the most experienced institutions with comparable resource availability.
Contrary to earlier neurosurgical and trauma VOR research, which only examined the impact of volume for the procedure or patient population being studied, we analyzed four different measures of volume and found that pediatric major trauma was the only volume metric that significantly impacted discharge disposition, our primary outcome variable. Analysis based on this measure demonstrated that centers with greater pediatric major trauma volume had significantly higher odds of favorable discharge (Figure 2). This trend is consistent with previous VOR studies in neurotrauma patients (9,14,23,24). Together, these findings suggest that greater pediatric major trauma experience is informative for the care of TBI patients. There are several plausible explanations for this observation. One possibility is that the management of systemic (rather than neurologic) sequelae of trauma that includes a TBI component is more critical to outcomes, and so greater general pediatric trauma experience is more directly impactful in these cases. Alternatively or in addition, because non-pediatric subspecialty physicians (including neurosurgeons) and care technicians (e.g., respiratory care specialists) at some institutions may cross-cover to treat pediatric neurotrauma, higher pediatric major trauma volume may improve these individuals’ expertise at pediatric neurotrauma care. Particularly at institutions with low TBI volume, insights from handling more pediatric major trauma may translate to better care for these patients presenting with severe TBI. Conversely, higher volume centers may have sufficient staffing to ensure care is delivered exclusively by pediatric neurosurgeons, pediatric intensivists, and other pediatric care providers. Patients presenting with polytrauma may also benefit from treatment at centers with greater severe pediatric trauma experience. Greater volume may also influence the development of an institution’s management policies and processes of care for pediatric trauma cases, allowing for more effective multidisciplinary treatment of severe TBI patients. The methodology and findings of the present study may inform future VOR research by expanding focus towards multiple volume metrics, rather than a singular measure of experience directly related to the disease process of interest.
Previous work has suggested that hospital volume can impact a variety of factors such as surgeon experience with pediatric procedures, performance during time-sensitive cases, and hospital refinement of management strategies (22,25). For severe pediatric TBI patients, who have distinct needs and require multidisciplinary care, hospital volume related to different trauma care processes may exert an especially important effect on outcomes. The present study found that for the pediatric major trauma and pediatric severe TBI volume models, higher volume centers had lower rates of complications (OR =0.956 per +100 patients, P=0.018; Figure 2). By comparison, a study that investigated adult TBI outcomes based on adult TBI volume saw no significant effect on complication rates (13). Importantly, the authors used a different database, considered all center types in their analysis, and only considered the effects of one measure of volume.
Of particular note was the highly significant relationship between greater pediatric volume and lower rates of ARDS in both primary and sensitivity analyses. While the connection between central nervous system insults and the onset of respiratory complications is still an area of developing research, TBI has been associated with increased risk of a pulmonary edema (26). Treating comorbid ARDS and TBI is especially difficult due to the competing effects of mechanical ventilation on TBI (27,28). Our analysis suggests that pulmonary complications might be a factor affecting variability in outcomes among institutions. Centers with higher pediatric TBI and trauma volume may have more advanced protocols for multidisciplinary care by pediatric, neurocritical care, and pulmonology specialists, and thus better equipped to treat this complex set of conditions. Efforts to improve the management of pulmonary complications may offer benefits for pediatric TBI patients, particularly at low-volume centers. However, further research is needed to understand the connection between treatment for comorbid TBI and respiratory issues. Although evidence is conflicting, previous studies have also suggested that platelet transfusions, proinflammatory effects of platelets, and elevated ICP may contribute to the development of ARDS (28-30). Our present findings demonstrate that patients at higher volume centers are more likely to undergo early ICP monitoring and transfusions for coagulopathy (Figure 2). Further research is necessary to determine the extent to which these practices impact the pathogenesis of ARDS after severe TBI.
Although favorable discharge was more likely with higher pediatric major trauma volume, mortality rates for severe pediatric TBI patients between low and high-volume institutions were about the same across all volume analyses (Figures 2-5). This is in contrast to some previous studies that saw improved mortality rates at higher volume centers for trauma patients (9,21). However, our findings may have been a result of a relatively smaller sample size (n=5,425). To further investigate this, we re-ran an identical analysis, but included all trauma center types. Consistent with our previous results, mortality was not significantly different at higher pediatric major trauma volume centers (P=0.056; Figure S3). Mortality rates may also be impacted by residual confounding. For example, hospitals with greater volume may have higher case complexity, which the severity metrics in our multivariate model were not entirely capable of capturing. Trauma center type, which is controlled in our analysis by limiting the study population to Pediatric ACS Level I and II centers, has also been connected to improved mortality rates for TBI patients (5,7,31). However, most of these studies focused on adults rather than children. There may be relevant differences even within the pediatric population, dependent on age; one study found that mortality rates for the pediatric trauma population at different trauma center types differ based on age of the child (32). Nevertheless, mortality is not the only salient endpoint for children with trauma. Ensuring favorable functional outcomes and status are also critical, and the present study suggests that high pediatric major trauma volume centers do have higher rates of favorable discharge disposition for children affected by TBI.
Unfortunately, however, the NTDB lacks information on longer term or more granular functional outcomes. The database also does not record measures of specific neurologic features related to brainstem function upon admission, such as pupillary response to light, due to these variables’ unavailability in the NTDB. While we followed validated earlier methods to remove patients with no signs of life of neurological activity (19), these exclusion criteria may not have completely accounted for this subpopulation with uniquely poor outcomes.
Implications of the pediatric severe TBI VOR
Centralization of care is an important strategy that several states and organizations have begun to implement for a variety of conditions (11,12,33). Research examining the management of pediatric TBI patients has suggested that optimal care of this younger patient group requires multidisciplinary approaches that are distinct from those used to manage adult neurotrauma (34,35). Our present results also demonstrate that higher pediatric major trauma volume translates to improved severe pediatric TBI outcomes, underscoring the value of general trauma experience in TBI management. As such, examining system-based factors may inform initiatives to benefit pediatric TBI patients at a population level and optimize hospital resource utilization. Discussing centralization of care is especially relevant to the pediatric TBI population because the time-sensitive nature of TBI typically does not allow patients to make fully informed decisions on where they are treated (36). In these situations, it is important that the routine intervention strategies and transfer or referral networks to high-performing institutions optimize outcomes for these critical care patients. However, there are some important drawbacks to heed for such strategies. Because the treatment of severe TBI is often time-sensitive, the additional time needed to present or transfer to higher volume institutions may negatively impact outcomes. Cost of treatment, both for the provider, as well as the patient, is another moderating factor. Additionally, although some hospitals treated fewer than 10 patients over a four-year period, these institutions may be the only place for trauma care in underserved regions. Lack of access to a center with the resources to manage TBI is an important concern that should be addressed in conversations about regionalization processes, and understanding intervention and practice differences between high- and low-performing institutions may help ameliorate these disparities. For example, coordination of care between centers with heterogenous availability of specialists may refine inter-hospital communication, educational partnerships, and management policies, allowing experienced institutions to serve as resources to local care and providing educational opportunities for practitioners and trainees at lower-volume centers (36,37). Regionalization of care may also offer benefits by catalyzing the study of best practices at high-volume institutions that may be disseminated to hospitals with less trauma experience.
Limitations
The present study has several potential limitations. First, the NTDB contains data voluntarily submitted by hospitals and may not be a fully complete and accurate representation of true trauma burden and care. However, because this database is the largest trauma registry in the United States, it was the best resource for this study. Additionally, because only initial admission data are included in the NTDB, analysis of long-term functional status and re-admissions was not possible; the short-term outcomes assessed here may not precisely reflect the ultimate functional outcomes of the studied population. Future research that includes follow-up data and analysis of long-term outcomes is necessary. Furthermore, multivariate regression only identifies associations between variables, providing an incomplete understanding of the relationship between disease processes and outcomes—causality cannot be assumed based on the present analysis. Adjusting for all the possible physiological variables that may affect outcomes presents another challenge. For example, information about the volume of intracranial hemorrhage for each case was not given, so residual confounding related to differences in severity of case presentation may still exist. Not all complications are able to be captured through the NTDB. Information about sepsis without organ dysfunction, for example, was not available. As with any hospital or national database, the potential for recording inaccuracies is present. However, the NTDB also contains several quality assurance measures to minimize the incidence of these coding inaccuracies (38). Changes in pediatric ACS verification since the end of our study period may merit consideration when interpreting the present results, as well.
Due to the selection criteria of this investigation, our data set sample size was also relatively small (n=5,425). To address this issue, we conducted a sensitivity analysis that included patients treated at institutions of any level (n=18,410) and found that the VOR of our primary outcome variable using the pediatric major trauma model was strengthened (Figure S3). Our analyses might be further refined by including a greater range of years of data from the NTDB or utilizing a different national database, such as the National Inpatient Sample. Finally, although the present analysis focused on the impacts of pediatric major trauma on our primary outcome variable, the other volume metrics may also be informative on outcomes. Further modeling to highlight the most predictive volume model for severe pediatric TBI is necessary.
Conclusions
Among different volume metrics, pediatric experience, rather than adult, appears to benefit severe pediatric TBI short-term outcomes. At Level I and II pediatric trauma care facilities, treatment at higher pediatric trauma volume hospitals was associated with improved outcomes, specifically higher likelihood of favorable discharge and lower rates of ARDS.
Acknowledgments
Preliminary data related to this study were presented at the Congress of Neurological Surgeons Annual Meeting, San Francisco, CA, October 19-23, 2019. The full manuscript has not been published elsewhere.
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/jeccm-21-24
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jeccm-21-24
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jeccm-21-24). WFA reports grants from Vivonics Inc., grants from Biogen Inc., outside the submitted work. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was exempt from Institutional Board Review due to the publicly available and de-identified nature of the data in the NTDB. Written informed consent from the patients was not required to participate in this study in accordance with the local legislation and the institutional requirements.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Araki T, Yokota H, Morita A. Pediatric Traumatic Brain Injury: Characteristic Features, Diagnosis, and Management. Neurol Med Chir (Tokyo) 2017;57:82-93. [Crossref] [PubMed]
- Faul M, Xu L, Wald M, et al. Traumatic Brain Injury in the United States: Emergency Department Visits, Hospitalizations and Deaths 2002-2006. Atlanta (GA): Center for Disease Control and Prevention, National Center for Injury Prevention and Control, 2010.
- Schneier AJ, Shields BJ, Hostetler SG, et al. Incidence of pediatric traumatic brain injury and associated hospital resource utilization in the United States. Pediatrics 2006;118:483-92. [Crossref] [PubMed]
- Bowman SM, Bird TM, Aitken ME, et al. Trends in hospitalizations associated with pediatric traumatic brain injuries. Pediatrics 2008;122:988-93. [Crossref] [PubMed]
- DuBose JJ, Browder T, Inaba K, et al. Effect of trauma center designation on outcome in patients with severe traumatic brain injury. Arch Surg 2008;143:1213-7; discussion 1217. [Crossref] [PubMed]
- Dams-O'Connor K, Cuthbert JP, Whyte J, et al. Traumatic brain injury among older adults at level I and II trauma centers. J Neurotrauma 2013;30:2001-13. [Crossref] [PubMed]
- Chalouhi N, Mouchtouris N, Saiegh FA, et al. Comparison of Outcomes in Level I vs Level II Trauma Centers in Patients Undergoing Craniotomy or Craniectomy for Severe Traumatic Brain Injury. Neurosurgery 2020;86:107-11. [Crossref] [PubMed]
- Bennett KM, Vaslef S, Pappas TN, et al. The volume-outcomes relationship for United States Level I trauma centers. J Surg Res 2011;167:19-23. [Crossref] [PubMed]
- Davies JM, Ozpinar A, Lawton MT. Volume-outcome relationships in neurosurgery. Neurosurg Clin N Am 2015;26:207-18. viii. [Crossref] [PubMed]
- Sewalt CA, Wiegers EJA, Venema E, et al. The volume-outcome relationship in severely injured patients: A systematic review and meta-analysis. J Trauma Acute Care Surg 2018;85:810-9. [Crossref] [PubMed]
- Long DM, Gordon T, Bowman H, et al. Outcome and cost of craniotomy performed to treat tumors in regional academic referral centers. Neurosurgery 2003;52:1056-63; discussion 1063-5. [PubMed]
- Gordon TA.. BGP, Tielsch J.M., Cameron J.L. The Effects of Regionalization on Cost and Outcome for One General High-Risk Surgical Procedure. Ann Surg 1995;221:43-9. [Crossref] [PubMed]
- Alali AS, Gomez D, McCredie V, et al. Understanding Hospital Volume-Outcome Relationship in Severe Traumatic Brain Injury. Neurosurgery 2017;80:534-42. [Crossref] [PubMed]
- Tepas JJ 3rd, Pracht EE, Orban BL, et al. High-volume trauma centers have better outcomes treating traumatic brain injury. J Trauma Acute Care Surg 2013;74:143-7; discussion 147-8. [Crossref] [PubMed]
- Tilford JM, Simpson PM, Green JW, et al. Volume–Outcome Relationships in Pediatric Intensive Care Units. Pediatrics 2000;106:289-94. [Crossref] [PubMed]
- Choi PM, Hong C, Woods S, et al. Early impact of American College of Surgeons-verification at a level-1 pediatric trauma center. J Pediatr Surg 2016;51:1026-9. [Crossref] [PubMed]
- American College of Surgeons. About NTDB. Chicago, IL: American College of Surgeons, 2019 [cited 2019 September 12]. Available online: https://www.facs.org/quality-programs/trauma/tqp/center-programs/ntdb/about
- Kortbeek JB, Al Turki SA, Ali J, et al. Advanced trauma life support. 8th edition, the evidence for change. J Trauma 2008;64:1638-50.
- Alarhayem AQ, Cohn SM, Eastridge BJ, et al. Natural History of Trauma Patients Presenting “Dead on Arrival”: Should We Resuscitate? J Am Coll Surg 2015;221:S165. [Crossref]
- Champion HR, Copes WS, Sacco WJ, et al. The Major Trauma Outcome Study: Establishing National Norms for Trauma Care. J Trauma 1990;30:1356-65. [Crossref] [PubMed]
- Nathens AB, Jurkovich GJ, Maier RV, et al. Relationship between trauma center volume and outcomes. JAMA 2001;285:1164-71. [Crossref] [PubMed]
- Clement RC, Carr BG, Kallan MJ, et al. Volume-outcome relationship in neurotrauma care. J Neurosurg 2013;118:687-93. [Crossref] [PubMed]
- Berman MF, Solomon RA, Mayer SA, et al. Impact of hospital-related factors on outcome after treatment of cerebral aneurysms. Stroke 2003;34:2200-7. [Crossref] [PubMed]
- Tang OY, Yoon JS, Kimata AR, et al. Volume-outcome relationship in pediatric neurotrauma care: analysis of two national databases. Neurosurg Focus 2019;47:E9 [Crossref] [PubMed]
- McAteer JP, LaRiviere CA, Drugas GT, et al. Influence of surgeon experience, hospital volume, and specialty designation on outcomes in pediatric surgery: a systematic review. JAMA Pediatr 2013;167:468-75. [Crossref] [PubMed]
- Holland MC, Mackersie RC, Morabito D, et al. The development of acute lung injury is associated with worse neurologic outcome in patients with severe traumatic brain injury. J Trauma 2003;55:106-11. [Crossref] [PubMed]
- Contant CF, Valadka AB, Gopinath SP, et al. Adult respiratory distress syndrome: a complication of induced hypertension after severe head injury. J Neurosurg 2001;95:560-8. [Crossref] [PubMed]
- Della Torre V, Badenes R, Corradi F, et al. Acute respiratory distress syndrome in traumatic brain injury: how do we manage it? J Thorac Dis 2017;9:5368-81. [Crossref] [PubMed]
- Matthay MA, Zemans RL. The acute respiratory distress syndrome: pathogenesis and treatment. Annu Rev Pathol 2011;6:147-63. [Crossref] [PubMed]
- Kornblith LZ, Robles AJ, Conroy AS, et al. Predictors of postinjury acute respiratory distress syndrome: Lung injury persists in the era of hemostatic resuscitation. J Trauma Acute Care Surg 2019;87:371-8. [Crossref] [PubMed]
- Oyetunji TA, Haider AH, Downing SR, et al. Treatment outcomes of injured children at adult level 1 trauma centers: are there benefits from added specialized care? Am J Surg 2011;201:445-9. [Crossref] [PubMed]
- Sathya C, Alali AS, Wales PW, et al. Mortality Among Injured Children Treated at Different Trauma Center Types. JAMA Surg 2015;150:874-81. [Crossref] [PubMed]
- Surgical Volume: The Leapfrog Group; 2019 [cited 2019 June 4]. Available online: https://www.leapfroggroup.org/ratings-reports/surgical-volume
- Giza CC, Mink RB, Madikians A. Pediatric traumatic brain injury: not just little adults. Curr Opin Crit Care 2007;13:143-52. [Crossref] [PubMed]
- Figaji AA. Anatomical and Physiological Differences between Children and Adults Relevant to Traumatic Brain Injury and the Implications for Clinical Assessment and Care. Front Neurol 2017;8:685. [Crossref] [PubMed]
- Lorch SA, Myers S, Carr B. The regionalization of pediatric health care. Pediatrics 2010;126:1182-90. [Crossref] [PubMed]
- Davies JM, Lawton MT. Improved outcomes for patients with cerebrovascular malformations at high-volume centers: the impact of surgeon and hospital volume in the United States, 2000-2009. J Neurosurg 2017;127:69-80. [Crossref] [PubMed]
- Newgard CD, Fildes JJ, Wu L, et al. Methodology and analytic rationale for the American College of Surgeons Trauma Quality Improvement Program. J Am Coll Surg 2013;216:147-57. [Crossref] [PubMed]
Cite this article as: Kimata AR, Tang OY, Asaad WF. Generalizability of pediatric major trauma experience to severe pediatric traumatic brain injury at level 1 and 2 trauma centers. J Emerg Crit Care Med 2021;5:21.


