
Systemic vasospasms induced by vasopressor therapy resulting in diffuse tissue necrosis and death: a case report and review of the literature
Introduction
Vasopressor medications are commonly used in intensive care practice to maintain hemodynamic function during shock. Such medications are not without adverse effects. One such effect is excessive vasoconstriction and vasospasms leading to organ and limb ischemia. In vasodilatory shock unresponsive to volume resuscitation, there is a general consensus that norepinephrine is the first-choice vasopressor. Though not seen in this case, norepinephrine, known to have both agonistic alpha-1, and beta-1 adrenergic receptor activity, has been reported to cause vasospasms in the coronary arteries, inducing ST-segment elevation (1). It has also been reported to aggravate middle and anterior cerebral artery vasospasms during aneurysmal subarachnoid hemorrhage resulting in upper motor neuron damage (2). Similar to vasospasms, tissue necrosis is also a rare, but more reported side-effect of norepinephrine treatment. This side-effect has been reported in the extremities (3-5). Unlike norepinephrine, the addition of a second vasopressor to vasodilatory shock is often patient specific and based on physician preference. However, vasopressin may often be preferred due to its lower risk of atrial fibrillation in patients with septic shock. Due to its V-1 agonist effects, limb necrosis has also been reported with the use of vasopressin (6). The patient in this case report was administered both norepinephrine and vasopressin and subsequently experienced tongue and extremity necrosis. Tongue necrosis caused by excessive vasoconstriction secondary to vasopressor administration was first reported in 2010 in a patient who had presented for cardiogenic shock after overdosing on antihypertensive medications in an attempted suicide. The patient was treated with norepinephrine, as well terlipressin, a V-1 agonist, epinephrine, a non-selective adrenergic agonist, and dobutamine and milrinone which are inotropic agents. This resulted in the patient requiring extensive debridement of the tongue (7). There is also one report of necrosis affecting both the upper lip and extremities while under treatment of norepinephrine for septic shock (8). Both of the aforementioned patients survived their complications. The only reported case of tongue necrosis while under vasopressor support that has resulted in death occurred post-operatively after treatment for enteritis. The death was attributed to hypoperfusion caused by septic shock and not as a side effect of the norepinephrine and epinephrine therapy (9).
The previously reported cases of vasopressor-induced necrosis have not demonstrated the level of severity as demonstrated here. Systemic vasospasms have also not been previously reported. In this case report, we present for the first time a case in which necrosis of the extremities as well as the tongue occurred as a consequence of severe systemic vasopressor-induced vasospasms, which ultimately, resulted in death. In the absence of evidence-based guidelines, we provide a detailed description of how we identified and treated this condition to give critical care providers more information on how to do so. We present the following case in accordance with the CARE reporting checklist (available at http://dx.doi.org/10.21037/jeccm-20-161).
Case presentation
A 71-year-old male presented to the emergency department (ED) from a nursing home due to acute hypoxic respiratory failure and septic shock secondary to acute bacterial cystitis and bacteremia. While in the ED, the patient went into asystole and returned to spontaneous circulation after 1 round of chest compressions. The patient was intubated and started on norepinephrine and phenylephrine with a goal of a mean arterial pressure (MAP) greater than 65 mmHg along with stress-dose steroids. There were initial signs of improvement in overall clinical status with antibiotic therapy. The patient was extubated to room air on day 2 and weaned off phenylephrine soon after. On day 3, the patient started to experience abdominal pain correlating with lactic acidosis. A computed tomography angiogram (CTA) of the abdomen demonstrated complete proximal superior mesenteric artery (SMA) occlusion (Figure 1) for which the patient was started on a heparin drip. Due to persistent non-fluid responsive shock, phenylephrine was added again that same day and vasopressin was also started. On day 4, the patient was transitioned to bilevel positive airway pressure (BiPAP). He also developed oliguric acute kidney injury (AKI) due to ischemic-related acute tubular necrosis (ATN) for which sustained low efficiency dialysis (SLED) was initiated. At that time, epinephrine had to be added as the fourth vasopressor. On the same day, the upper extremities were found to intermittently have no pulse or bounding pulses. The lower extremities were cyanotic and pulseless, for which an arteriogram was done demonstrating vasospastic occlusion of the right internal femoral artery and right popliteal artery as well as the left internal femoral artery and midsegment of the left superficial femoral artery. A lower MAP goal was then used at 60 mmHg to facilitate weaning of vasopressors, which appeared to be aggravating the vasospasms. Vasopressin and epinephrine were weaned off that same day with phenylephrine actively being weaned. Despite that, the patient developed livedo reticularis in all extremities and tongue necrosis began developing. On day 5, the goal was changed to a systolic blood pressure of 90 mmHg as phenylephrine was weaned off and norepinephrine was decreased again, however, by day 6, the legs began to necrosis and blisters developed on the arms (Figure 2A). On day 7, the lower extremities (Figure 2A,B) and the tongue (Figure 3A,B) were fully necrosed. At this time, the family decided to proceed with inpatient hospice care. On the tenth day, the patient passed away from cardiopulmonary arrest. Details on the vasopressors and doses that were administered throughout the patient’s hospital course can be found in Table 1. The doses are reported as the average for each respective day.
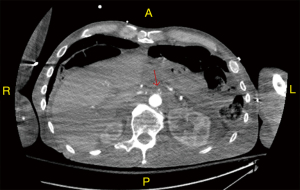
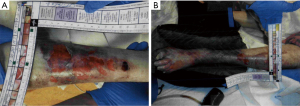
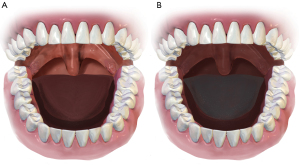
Table 1
| Day | Goal pressure | Vasopressor support | Dosing (average) | Hours per day under treatment |
|---|---|---|---|---|
| 1 | MAP >65 mmHg | Norepinephrine | 0.36 μg/kg/min | 15.50 |
| Phenylephrine | 2.35 μg/kg/min | 14.00 | ||
| Vasopressin | No dose | 0.00 | ||
| Epinephrine | No dose | 0.00 | ||
| 2 | MAP >65 mmHg | Norepinephrine | 0.37 μg/kg/min | 24.00 |
| Phenylephrine | 1.07 μg/kg/min | 16.75 | ||
| Vasopressin | No dose | 0.00 | ||
| Epinephrine | No dose | 0.00 | ||
| 3 | MAP >65 mmHg | Norepinephrine | 0.27 μg/kg/min | 24.00 |
| Phenylephrine | 1.37 μg/kg/min | 2.25 | ||
| Vasopressin | 0.04 U/min | 7.25 | ||
| Epinephrine | No dose | 0.00 | ||
| 4 | MAP >65 mmHg | Norepinephrine | 0.38 μg/kg/min | 24.00 |
| Phenylephrine | 4.29 μg/kg/min | 24.00 | ||
| Vasopressin | 0.04 U/min | N/A | ||
| Epinephrine | 0.10 μg/kg/min | 10.75 | ||
| 5 | MAP =60 mmHg | Norepinephrine | 0.30 μg/kg/min | 24.00 |
| Phenylephrine | 1.98 μg/kg/min | 8.00 | ||
| Vasopressin | No dose | 0.00 | ||
| Epinephrine | No dose | 0.00 | ||
| 6 | Systolic = 90 mmHg | Norepinephrine | 0.11 μg/kg/min | 24.00 |
| Phenylephrine | No dose | 0.00 | ||
| Vasopressin | No dose | 0.00 | ||
| Epinephrine | No dose | 0.00 | ||
| 7 | Systolic = 90 mmHg | Norepinephrine | 0.04 μg/kg/min | 24.00 |
| Phenylephrine | No dose | 0.00 | ||
| Vasopressin | No dose | 0.00 | ||
| Epinephrine | No dose | 0.00 |
Each day of the patient’s hospital course is shown with the goal blood pressure, the average dose of vasopressors administered each respective day, and how many hours each respective vasopressor was administered each day. MAP, mean arterial pressure.
It is important to note that the hospital course of the patient, who had a past medical history of gastroesophageal reflux disease, was complicated by a gastrointestinal bleed. Coffee ground blood was found on suction and fecal occult blood test was positive at admission. Hemoglobin was 9.1 g/dL at admission with the lowest level at 7.6 g/dL on day 3 of hospitalization with one unit of leukocyte-reduced packed red blood cells given that day. Also of note, the patient’s lactic acidosis progressed during the hospital course and the lactic acid reached a high of 5.6 mmol/L on day 4. Table 2 shows detailed lab values relevant to the patient’s clinical portrait. Thromboembolism was ruled out as a cause of the necrotic sequelae. See supplemental data 1 (Appendix 1) for details.
Table 2
| Variable | Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 | Day 7 | Normal range |
|---|---|---|---|---|---|---|---|---|
| Bleed | ||||||||
| Hemoglobin (g/dL) | 10.3 | 10.3 | 8.2 | 8.6 | 8.9 | 9.4 | 9.3 | 12.0–16.0 |
| Hematocrit (%) | 31.3 | 30.5 | 24.6 | 25.7 | 26.4 | 27.8 | 28.9 | 37.0–47.0 |
| Respiratory/acid-base status | ||||||||
| Respiration | Intubated | Intubated | Room Air | BiPAP | BiPAP | BiPAP | BiPAP | – |
| Arterial pH | 7.55 | 7.45 | 7.48 | 7.58 | 7.62 | 7.57 | 7.54 | 7.35–7.45 |
| Arterial pCO2 (mmHg) | 36.0 | 29.0 | 23.5 | 20.0 | 24.0 | 24.0 | 27.0 | 35.0–45.0 |
| Arterial pO2 (mmHg) | 82.0 | 89.5 | 71.4 | 285.5 | 155.0 | 236.0 | 247.0 | 60.0–100.0 |
| Bicarbonate (mmol/L) | 17.5 | 20.0 | 22.0 | 20.0 | 25.0 | 25.0 | 20.2 | 21.0–32.0 |
| Lactic acid (mmol/L) | 2.0 | N/A | 1.6 | 5.1 | 4.8 | 4.7 | 3.9 | 0.4–1.9 |
| Infection | ||||||||
| White blood cells (k/mm3) | 27.1 | 15.9 | 21.1 | 20.0 | 24.3 | 27.6 | 43.8 | 4.0–10.0 |
| C-reactive protein (mg/dL) | 13.4 | N/A | N/A | N/A | N/A | N/A | N/A | 0.0–0.9 |
| Renal function (SLED on day 4) | ||||||||
| Blood urea nitrogen (mg/dL) | 26.0 | 34.0 | 28.0 | 23.0 | 12.3 | 19.0 | 29.0 | 7.0–18.0 |
| Creatinine (mg/dL) | 1.25 | 1.3 | 0.90 | 0.70 | 0.43 | 0.90 | 1.47 | 0.60–1.30 |
| Urine output (mL/kg/h) | 1.85 | 0.57 | 0.84 | 0.21 | 0.08 | 0.05 | N/A | – |
Labs relevant to the patient’s hospital course are shown here. If multiple labs were drawn in a day, the average of those labs are reported for each respective day. BiPAP, bilevel positive airway pressure; SLED, sustained low efficiency dialysis.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and division research director as well as the Helsinki Declaration (as revised in 2013). The patient was deceased at the time of the writing of this manuscript. Written informed consent was obtained from the patient’s next of kin.
Discussion
Tongue necrosis secondary to vasopressor administration is a rare condition noted in only a handful of cases due to the rich vasculature and collateral circulation supplying the tongue. Use of vasopressors was warranted in our patient as the patient’s blood pressure failed to improve with resuscitative fluids. However, despite the need for vasopressor administration, it was important to wean the vasopressors down in an attempt to preserve the dying tissue and prevent further necrosis. The decisions to reduce the blood pressure goals were done in the absence of established guidelines, thus prompting further discussion.
Circulation of the extremities and the tongue, along with occlusion of the SMA on CTA of the abdomen (Figure 1), as well as ATN, was found to be compromised after initiation of vasopressors. Furthermore, the emergence of ATN on day 4 of hospitalization was unusual, as the patient was hypotensive on day 1 of admission, and yet did not present with prerenal AKI. This unexpected timing of ATN later on in the hospital course could indicate that the etiology of the ATN was renal vascular vasospasms. We recognize that hypoperfusion in the setting of septic shock alone could have been the cause of mesenteric ischemia in the context of possible underlying chronic SMA occlusion. However, the vasospasms of the lower extremity arteries, as shown in the angiogram, as well as the proposed vasospasms of the renal arteries are more suggestive of severe systemic vasospasms, of which the SMA may have been a part of. Furthermore, even if the patient had chronic SMA occlusion, it is likely that it was acutely exacerbated by something, as symptoms of severe mesenteric ischemia did not occur prior to the hospital course.
While we propose that vasopressor-induced vasospasm was the cause or a cause of the SMA occlusion and ATN, the main limitation of this case report is the difficulty of being able to accurately identify the cause without an autopsy, which could have provided more information about underlying vascular comorbidities. A Naranjo algorithm score was calculated to determine the likelihood that the various sequelae that the patient experienced were caused by vasopressors. We calculate a Naranjo algorithm score of 6 for attributing the tongue and limb necrosis to an adverse drug reaction to vasopressor therapy, indicating a “probable” likelihood. We also calculate a Naranjo algorithm score of 3 for attributing the SMA occlusion and ATN to vasopressor-induced vasospasms, indicating a “possible” likelihood (10).
It is possible that the patient’s prior history of smoking complicated the vasospasms. Though he had not smoked for at least the past five months that he had been under nursing home care, he did have an extensive history of smoking. Smoking has been associated with symptomatic vasospasms in the context of aneurysmal subarachnoid hemorrhage with a relative risk of 4.7 (11).
Vasopressor-induced vasospasms have previously been noticed in isolated arteries, such as cerebral arteries and coronary arteries (1,2). The possibility that the patient had such severe systemic vasospasms is, however, remarkable, as such severe vasospasms in the context of vasopressor therapy have not been found to be previously reported. Furthermore, the fact that such vasospasms continued throughout the hospital course with worsening necrosis, even while being weaned down to such low vasopressor doses is remarkable.
Symptoms including pain, erythema, and glossodynia were not found in this patient on presentation. The patient’s initial intubation may have caused some reduction in continuous circulation to the tongue. However, symptoms were not noted until days after extubation. Multiple differential diagnosis can be mentioned that could have caused the tongue necrosis including shock, giant cell arteritis, disseminated intravascular coagulation, and Wegener’s granulomatosis. All except shock appear unlikely.
Tongue necrosis is one of the rare complications seen in patients using vasopressors. Treatment of tongue necrosis in granulomatous diseases includes steroids and resection of the necrotic debris under general anesthesia (12). Debridement and pain management were the treatments considered in our case. Had debridement been chosen, the severity of the necrosis was such that two-thirds or even up to complete amputation of the tongue would have been necessary. The quality-of-life issues that this would have created was a significant factor in the decision not to treat the tongue necrosis. Instead, the family opted for hospice care.
Some other side effects noted with the use of vasopressors include hypoperfusion (13), dysrhythmias (14), and local vasoconstriction resulting in skin necrosis, which can occur either with or without extravasation (15).
Throughout the hospital course, we titrated the vasopressor doses down as much as possible in an attempt to determine a balance between preventing further necrosis in the already dying tissues and maintaining adequate perfusion to other organs. Adhering to the standard pressure goal of a MAP greater than 65 mmHg throughout the necrotic process would have worsened the already extreme tongue and limb necrosis. We used our clinical judgment in establishing our blood pressure goals and vasopressor doses, as there are currently no published guidelines for maintaining permissive hypotension while attempting to minimize tissue damage in the setting of necrotic side effects of vasopressors. The only published data we found to guide our decisions was Lamontagne el al. (2020), which showed that a MAP goal of 60–65 mmHg in patients older than 65 did not show a decrease in mortality at 90 days compared to a MAP goal of greater than 65 mmHg (16). There are also no published guidelines on the pharmacologic management of vasopressor-induced necrosis related to systemic vasospasms or vasoconstriction. The only existing guidelines for treatment of necrosis caused by vasopressor are related to extravasation (17,18). In these guidelines, pharmacologic treatments include phentolamine, a non-selective alpha-adrenergic antagonist, terbutaline, a beta-2 adrenergic agonist, and topical nitroglycerin, which causes vasodilation through the production of nitric oxide. Our patient was not a candidate for these medications, as he would not have been able to tolerate the hypotensive side-effects associated with these medications.
We also wonder how common systemic vasospastic side effects to vasopressors actually are. The case reports/studies we cited that were related to tissue necrosis in the setting of vasopressors all attributed the necrosis to either vasoconstrictive ischemia from the vasopressors or to hypoperfusion from shock. Is it possible that any of those patients may have had systemic vasospasms? Isolated vasospasm in coronary and cerebral arteries were previously noted with use of vasopressors (1,2), but this case showed severe generalized vasospasms affecting multiple organ systems. Perhaps our physical exam findings from pulse checks as well as the angiographic studies we did allowed us to discover the systemic vasospasms that might have otherwise been attributed to vasoconstrictive ischemia or hypoperfusion. Vasospastic ischemia may be considered a form of vasoconstriction but is certainly a unique form of vasoconstriction that we have discovered.
In conclusion, complete limb necrosis is a rare complication seen with the use of vasopressors. Tongue necrosis is even rarer. Our reported sequelae were likely induced by systemic vasospasm and would necessitate prompt action in order to preserve and restore any damage caused in the process. Daily visualization of the tongue during physical exam to look for discoloration as well as thorough daily inspection and pulse checks of the limbs can help identify vasospasms while on vasopressor support and allow prompt action that might minimize permanent injury and decrease recovery time.
Acknowledgments
We would like to thank artmessy for a detailed and accurate artist’s rendition of the tongue necrosis (Figure 3A,B). Illustration services from artmessy can be found at https://www.fiverr.com/artmessy. This research was supported (in whole or in part) by HCA Healthcare and/or an HCA Healthcare affiliated entity. The views expressed in this publication represent those of the author(s) and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at http://dx.doi.org/10.21037/jeccm-20-161
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jeccm-20-161). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and division research director as well as the Helsinki Declaration (as revised in 2013). The patient was deceased at the time of the writing of this manuscript. Written informed consent was obtained from the patient’s next of kin.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Elkaryoni A, Ramakrishnan D, Abdelkarim I, et al. Vasopressor-induced generalized coronary vasospasm presenting as inferior ST-segment elevation in post-cardiopulmonary resuscitation. JACC Case Rep 2019;1:94-8. [Crossref] [PubMed]
- Zeiler FA, Silvaggio J, Kaufmann AM, et al. Norepinephrine as a potential aggravator of symptomatic cerebral vasospasm: two cases and argument for milrinone therapy. Case Rep Crit Care 2014;2014:630970. [Crossref] [PubMed]
- Khundkar R, Wilson PA, Khan U. A cautionary tale. J Plast Reconstr Aesthet Surg 2009;62:253. [Crossref] [PubMed]
- Simman R, Phavixay L. Bilateral toe necrosis resulting from norepinephrine bitartrate usage. Adv Skin Wound Care 2013;26:254-6. [Crossref] [PubMed]
- Daroca-Pérez R, Carrascosa MF. Digital necrosis: a potential risk of high-dose norepinephrine. Ther Adv Drug Saf 2017;8:259-61. [Crossref] [PubMed]
- Cho AR, Kim JI, Kim EJ, et al. Skin necrosis after high dose vasopressor infusion in septic shock: two case reports. Korean J Crit Care Med 2012;27:182-6. [Crossref]
- Noordally SO, Sohawon S, Duttmann R, et al. Tongue necrosis as a complication of vasoconstrictor agents in the intensive care setting. Intern Emerg Med 2011;6:183-5. [Crossref] [PubMed]
- Shin JY, Roh SG, Lee NH, et al. Ischemic Necrosis of Upper Lip, and All Fingers and Toes After Norepinephrine Use. J Craniofac Surg 2016;27:453-4. [Crossref] [PubMed]
- Cho J, Sung K, Lee D. Ischemic necrosis of the tongue in surgical patients with septic shock: a case report. BMC Surg 2016;16:48. [Crossref] [PubMed]
- Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 1981;30:239-45. [Crossref] [PubMed]
- Lasner TM, Weil RJ, Riina HA, et al. Cigarette smoking-induced increase in the risk of symptomatic vasospasm after aneurysmal subarachnoid hemorrhage. J Neurosurg 1997;87:381-4. [Crossref] [PubMed]
- Llorente Pendás S, De Vicente Rodríguez JC, González García M, et al. Tongue necrosis as a complication of temporal arteritis. Oral Surg Oral Med Oral Pathol 1994;78:448-51. [Crossref] [PubMed]
- Gregory JS, Bonfiglio MF, Dasta JF, et al. Experience with phenylephrine as a component of the pharmacologic support of septic shock. Crit Care Med 1991;19:1395-400. [Crossref] [PubMed]
- De Backer D, Biston P, Devriendt J, et al. Comparison of dopamine and norepinephrine in the treatment of shock. N Engl J Med 2010;362:779-89. [Crossref] [PubMed]
- HARDIE GH. HUNTER DC Jr. Skin necrosis with the intravenous use of norepinephrine. Med Bull (Ann Arbor) 1955;21:213-9. [PubMed]
- Lamontagne F, Richards-Belle A, Thomas K, et al. Effect of Reduced Exposure to Vasopressors on 90-Day Mortality in Older Critically Ill Patients With Vasodilatory Hypotension: A Randomized Clinical Trial. JAMA 2020;323:938-49. [Crossref] [PubMed]
- Reynolds PM, MacLaren R, Mueller SW, et al. Management of extravasation injuries: a focused evaluation of noncytotoxic medications. Pharmacotherapy 2014;34:617-32. [Crossref] [PubMed]
- Plum M, Moukhachen O. Alternative Pharmacological Management of Vasopressor Extravasation in the Absence of Phentolamine. P T 2017;42:581-92. [PubMed]
Cite this article as: Arian MU, Medina CS, Matus G, Hazari AD, Chozet L, Omar F. Systemic vasospasms induced by vasopressor therapy resulting in diffuse tissue necrosis and death: a case report and review of the literature. J Emerg Crit Care Med 2022;6:10.


