An unusual cause of a toddler with a big belly, abdominal lymphatic malformation case report
Introduction
Abdominal pain is one of the most common complaints seen in the pediatric emergency department (PED). Because of the broad range of potential diagnoses, it can pose challenges in diagnosis of the young child, given speech has not yet developed and description of symptoms is lacking. We present a case of a young child presenting to the Emergency Department with one day of abdominal pain, recent weight gain, and abdominal distention. Laboratory studies and imaging did not establish a diagnosis until surgical resection and pathology confirmed a macrocystic lymphatic malformation. We present the following article in accordance with the CARE reporting checklist (available at http://dx.doi.org/10.21037/jeccm-20-146).
Case presentation
A 3-year-old Caucasian female presented to our PED with one day of abdominal pain, tactile fever and poor appetite. The patient developed abdominal pain with poor appetite that worsened throughout the evening. She was also having a harder time bending over. The pain improved by curling her knees up toward her abdomen. Pain was localized to the lower abdomen, per parents. Associated symptoms include tactile fever, rhinorrhea, and constipation (no bowel movement the last 2 days). Pertinent negatives included no vomiting, diarrhea, documented fevers, sore throat, cough, hematuria, dysuria, or rash. The parents report a history of the patient having a large abdomen that her pediatrician felt was normal for age. The parents were concerned because the abdominal pain did not improve with MiraLAX and fruit juice, and the child rarely complained. Past medical history was negative for prior medical illnesses or surgeries. Of note, her weight-for-age percentile increased from the 8th percentile to the 33rd percentile in the last 6 months.
The patient weighed 13.2 kg and her vital signs at presentation were: heart rate 120, respiratory rate of 24, blood pressure 101/75 mmHg, and temperature of 37.5 °C (99.5 °F). On initial examination in the PED, the patient was tired appearing but non-toxic in no acute distress. Her mucous membranes were dry, and her cardiovascular exam was significant for tachycardia. She had strong peripheral pulses, good distal perfusion, and a normal pulmonary exam. Her abdominal exam was significant for a tense distended abdomen, taut skin with prominent abdominal wall vasculature, and shifting gravity dependent tympany. There was generalized fullness and tenderness to palpation. There was no palpable abdominal mass or hepatosplenomegaly.
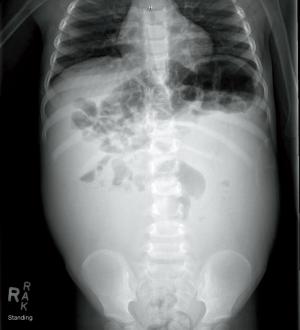
In the PED, an abdominal radiograph (X-ray) and abdominal ultrasound (US) was obtained. The abdominal X-ray revealed a “nonspecific but abnormal bowel gas pattern with air-filled loops in the right upper quadrant but paucity of bowel gas in the remained of the abdomen” (Figure 1). Abdominal US showed a “large volume of complex intra-abdominal ascites. No definite intraperitoneal mass identified, but a neoplastic process should be considered. Additional diagnostic considerations include chylous ascites or infection/hemorrhage.”
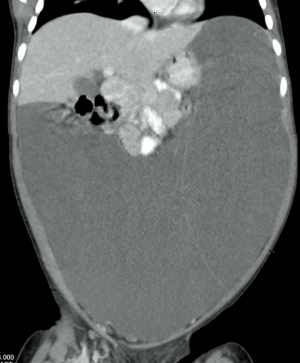
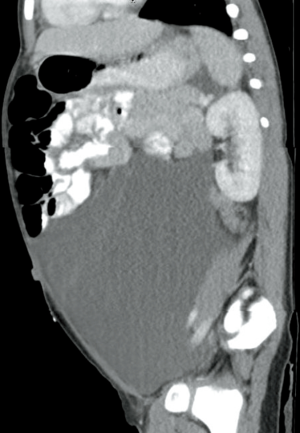
An intravenous catheter (IV) was placed and initial laboratory studies obtained included a complete blood count (CBC), basic metabolic profile (BMP), hepatic profile, pancreatic enzymes and C-reactive protein (CRP). Pertinent laboratory results were as follows: white blood cell count (WBC) of 16.45 K/mcL (reference range, 6.0–17.0 K/mcL) with 69% segmented neutrophils, hemoglobin 11.2 gm/dL (reference range, 11.5–13.5 gm/dL), CRP of 5.92 mg/dL (reference range, ≤0.30 mg/dL), and lactic acid of 1.1 (1.0–2.4 mmol/L). The rest of her labs were normal including a hepatic profile, BMP and pancreatic enzymes. The patient was given IV Toradol for pain and a normal saline (NS) bolus. Pediatric general surgery was consulted who evaluated the patient in the ED and stated that there was no acute surgical intervention. Gastroenterology recommended obtaining an abdominal/pelvis computed tomography (CT) scan with IV contrast, lactic acid dehydrogenase (LDH), uric acid, CA 19-9, alpha fetoprotein (AFP), coagulation studies, urinalysis (UA), and urine protein:creatinine ratio. All laboratory studies were normal, except for the UA with 40+ ketones. The CT abdomen/pelvis scan was obtained prior to admission and showed: “mass-like accumulation of fluid within the abdomen, appears relatively simple by CT though more complexity was apparent by US. Septations in and around the fluid appear to largely correspond with peritoneal reflections, some of which contain vessels and fat. Solid organs and hollow abdominal viscera are normal, with the exception of mass effect. Differential diagnosis includes lymphatic malformation and cystic neoplasm” (Figures 2,3).
The patient was admitted to the hospital medicine service. Oncology recommended obtaining a magnetic resonance imaging (MRI) study of the abdomen/pelvis and the following laboratory studies: cancer antigen 125 (Ca-125), luteinizing hormone (LH), follicle stimulating hormone (FSH), inhibin B, dihydrotestosterone (DHT), total testosterone, and beta human chorionic gonadotropin (HCG). Lab studies were obtained, and the only abnormality was elevated CA-125 of 94.4 unit/mL. (reference range, ≤30.2 unit/mL). The patient received an interventional radiology (IR) guided paracentesis of the abdominal fluid for diagnostic and symptom relief purposes under general anesthesia. Approximately 20 milliliters (mL) of dark fluid was aspirated and sent to the laboratory for analysis. Peritoneal fluid results showed turbid brown fluid, WBC 3,489 mm3, RBC 29,000 mm3, serum ascites albumin gradient (SAAG) <1.1, amylase 21, glucose 77, LDH 686, triglyceride (TG) 31, protein 3.9, and cholesterol crystals were noted. Gram stain showed many white blood cells, no epithelial cells, no malignant cells, and no organisms. MRI of the abdomen/pelvis and CT scan of the chest was completed 3 days after admission. The CT scan of the chest showed no abnormalities. The MRI of the abdomen/pelvis showed a “large amount of intraperitoneal fluid with component of internal complexity, septations/loculation but no areas of suspicious enhancement to suggest overt malignant process. Differential considerations include potential lymphatic malformation or unusual mesothelial cyst”. At the time of discharge, the patient had scheduled appointments with surgery, IR, and vascular malformation.
Approximately six weeks following discharge, the patient was evaluated in the Center for Hemangiomas and Vascular Malformations. The physicians recommended a diagnostic laparoscopy with biopsies and resection. Three months after the initial PED presentation, the patient underwent surgery. A total of 1.8 liters (L) of straw-colored proteinaceous fluid was sent for pathology. A very large cystic mass extending from the upper quadrants of the omentum down into the pelvis was mobilized and decompressed. An omentectomy was performed to fully resect the cyst. Postoperative diagnosis was a large multi-lobular congenital omental cyst. Pathology was consistent with a macrocystic lymphatic malformation (Appendix 1). At her postoperative visit one month later, abdominal distention and pain had completely resolved. The patient will continue to have abdominal US surveillance in 3–6 months, then 1 year, and again at puberty to assess for interval recurrence of lymphatic ascites.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Discussion
This case report of a child with a rare abdominal lymphatic malformation includes a detailed description of the clinical presentation, emergency medicine evaluation and management, continued descriptive evaluation and clinical course of the child during their hospital stay, and ultimate diagnosis with definitive management. Limitations of the case report include inability to follow the patient multiple years after their surgical resection to assess for recurrence.
Abdominal pain is a common complaint of pediatric patients presenting to the emergency department. The most common causes of abdominal pain in toddlers includes constipation, gastroenteritis, urinary tract infection, pyelonephritis, and streptococcal pharyngitis; congenital abdominal anomalies are often not included in the differential diagnosis (1).
Lymphatic malformations are exceedingly rare in children, accounting for only 5% of benign tumors in childhood (2,3). They comprise of dilated lymphatic channels that form cyst-like structures with fibrous septa (3). Common locations include neck (75%) and axillae (20%), only rarely involving the gastrointestinal tract (<1%) (2,4). The clinical presentation depends on the location and size of the tumor. In children, they are frequently discovered as incidental findings when imaging studies are obtained. Over ninety percent of lymphatic malformations in the abdomen occur before the age of 2 years (2,5). Symptoms that may occur from these lesions include abdominal pain, vomiting, changes in bowel habits, obstruction, volvulus, intussusception, and/or secondary infectious or hemorrhagic complications (2,4). The abdominal pain in our patient likely occurred secondary to massive ascites from obstruction resulting in a tense distended abdomen.
The etiology of lymphatic malformations continues to be unknown, although many theories have been hypothesized (2). The early sequestration of lymphatic vessels may fail to create normal connections with draining lymphatic ducts, causing dilation and cyst development (2). Another idea is that acquired factors may contribute to cyst development such as inflammation, trauma, or degeneration of lymphatic ducts (2).
Diagnosis is best initially accomplished through US, which is easily accessible and is sensitive to define cystic masses (2,3). An important aspect of diagnosis is the detection of intracystic echogenic septae within the lesion (2). US can occasionally be limiting if the lesion is large, making it difficult to determine the site of origin as well as the borders (2). Plain radiographs often fail to show these cystic structures but may reveal bowel displacement, as in our patient (2). CT scans are useful to describe the origin and extension of the cystic structures (2). CT scanners are fast, may require no sedation, and are available in the emergency department, where some lymphatic malformations are first diagnosed (3). When using CT to better evaluate the lymphatic malformation, administration of oral and IV contrast improves contrast resolution (3). MRI is best used for large and diffuse infiltrative as well as microcystic lymphatic malformations, as well as being sensitive and specific for hemorrhagic complications (2-6).
There is a paucity of literature regarding congenital lymphatic malformations, especially in the toddler age. This age presents a difficult diagnosis as speech has not yet fully developed and may be difficult to specify symptoms. Also, younger children and toddlers often have larger abdomens, and a mass may not actually be palpable. Multiple family members had noted the patient’s large abdomen in the prior year, but her pediatrician had reassured her that her abdominal size was normal for her age. The general pediatrician may have been clued into the diagnosis sooner with regards to multiple family member concerns that her abdomen was growing at a greater rate than the rest of her body and she had gained significant weight from her developing ascites, from the 8th percentile to the 33rd percentile in the last 6 months.
Surgical resection is the definitive treatment for abdominal cystic lymphangiomas, with bowel resection occasionally being required (2,7). US is the preferred imaging modality for postoperative surveillance (2). An approximately 10% recurrence rate has been described in the literature for patients who have undergone partial resections (7). There are no current guidelines regarding frequency of US surveillance to determine recurrence (2).
In conclusion, abdominal pain in a young child presenting with abdominal distention and increasing weight should be evaluated with imaging studies for intra-abdominal manifestations. Although rare, lymphatic malformations can occur in children in multiple regions of the body. They pose a diagnostic challenge given the rarity of their disease process, but clinicians should be considering lymphatic malformations when evaluating for a mass in particular regions of the body.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The author has completed the CARE reporting checklist. Available at http://dx.doi.org/10.21037/jeccm-20-146
Conflicts of Interest: The author has completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jeccm-20-146). The author has no conflicts of interest to declare.
Ethical Statement: The author is accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Reust CE, Williams A. Acute Abdominal Pain in Children. Am Fam Physician 2016;93:830-6. [PubMed]
- Rana A, Katzman PJ, Pegoli W, et al. An unusual cause of abdominal pain: duodenal cystic lymphangioma. Gastroenterol Hepatol (N Y) 2013;9:192-5. [PubMed]
- Francavilla ML, White CL, Oliveri B, et al. Intraabdominal Lymphatic Malformations: Pearls and Pitfalls of Diagnosis and Differential Diagnoses in Pediatric Patients. AJR Am J Roentgenol 2017;208:637-49. [Crossref] [PubMed]
- Calmés D, Médart L. Abdominal Lymphatic Malformation. J Belg Soc Radiol 2018;102:57. [Crossref]
- Chateil JF, Brun M, Vergnes P, et al. Abdominal cystic lymphangiomas in children: presurgical evaluation with imaging. Eur J Pediatr Surg 2002;12:13-8. [Crossref] [PubMed]
- Iyer R, Eftekhari F, Varma D, et al. Cystic retroperitoneal lymphangioma: CT, ultrasound and MR findings. Pediatr Radiol 1993;23:305-6. [Crossref] [PubMed]
- Steyaert H, Guitard J, Moscovici J, et al. Abdominal cystic lymphangioma in children: benign lesions that can have a proliferative course. J Pediatr Surg 1996;31:677-80. [Crossref] [PubMed]
Cite this article as: Riney LC. An unusual cause of a toddler with a big belly, abdominal lymphatic malformation case report. J Emerg Crit Care Med 2021;5:24.