
Non-pulmonary manifestations of coronavirus disease 2019 (COVID-19): case report
Introduction
The 2019 novel coronavirus [now called severe acute respiratory failure coronavirus 2 (SARS-CoV-2), causing the disease coronavirus disease 2019 (COVID-19)] was declared a global pandemic by the World Health Organisation (WHO) on 11th March 2020. Early reports from China suggested that COVID-19 resulted in a viral pneumonia. Patients present with the cardinal features of fever (88.7%), cough (67.8%) and variable shortness of breath (18.7%) (1). Alternative clinical features, such as diarrhoea (3.7%), rashes (0.2%) and conjunctivitis (0.9%) were rare (1). The vast majority of patients presented with the aforementioned cardinal features, in keeping with pulmonary COVID-19, thus allowing for easier triage, diagnosis and management. The disease spectrum ranges from asymptomatic carriage through to multi-organ failure. Complications were primarily single organ, pneumonia (91.1%) and in those with extra-pulmonary manifestations, such as myocarditis, patients often had significant COVID-19 lung disease (2).
In this case report, we discuss the diagnostic difficulties of COVID-19 in a previously well 30-year-old female, who presented with a 1-week history of atypical symptoms and subsequently developed new onset acute cardiac failure. She had no pulmonary involvement, making the diagnosis of COVID-19 challenging. She was the first patient to present atypically to our hospital. Confounding factors were the lack of the classic pulmonary COVID-19 symptoms, no COVID-19 specific radiological findings and serial negative reverse transcriptase-polymerase chain reaction (RT-PCR) tests. A chronological clinical course will be presented, alongside the various investigations, advice from complimentary specialties and subsequent management. We present the following article in accordance with the CARE reporting checklist (available at http://dx.doi.org/10.21037/jeccm-20-81).
Case presentation
A 30-year-old female presented to Accident & Emergency (A&E) with a 5-day history of diarrhoea, vomiting, muscle aches and intermittent fevers. She also complained of red eyes for 2 days which had resolved by admission. She denied any cough, dyspnoea and had no history of recent travel or unwell contacts. Her past medical history was of coeliac disease and a thoracic scoliosis, managed conservatively. She was on no regular medication and had no allergies. She did not smoke, had minimal alcohol intake and an unlimited exercise tolerance.
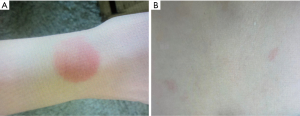
On initial examination, she was shocked, hypotensive (blood pressure 68/46) and tachycardic (sinus at 133 beats per minute) with cool peripheries, and no oedema. Her respiratory examination (saturations 99% on room air, respiratory rate 16 breaths/minute, with a clear chest on auscultation) and abdominal examination were unremarkable. Of note, one distinct rash was noticed, and another found incidentally on examination (Figure 1). She was lucid with no focal neurology, but with intermittent pyrexias.
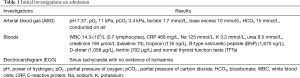
Initial significant investigations are displayed in Table 1.
Full table
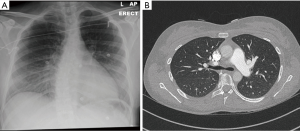
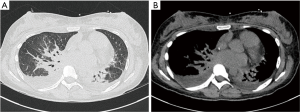
A chest X-ray (CXR), and then a computerised tomography (CT) chest/abdomen/pelvis (C/A/P) was performed to identify the source of sepsis (Figure 2).
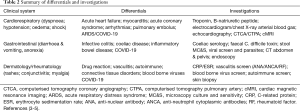
The working diagnosis was atypical COVID-19; however, her initial RT-PCR swab was negative. Based on the evidence thus far, she was treated with antibiotics for infective gastroenteritis. The shock responded to fluid therapy and was attributed to hypovolaemia, combined with an element of distributive shock (sepsis). Despite adequate resuscitation, she remained hypotensive and was escalated to critical care for vasopressors. A summary of the differential diagnoses alongside the patient’s systems are demonstrated in Table 2.
Full table
On critical care, she developed rapidly worsening dyspnoea and hypotension, although her diarrhoea had settled. COVID-19 was still suspected, however the repeat swab, the following day, was negative. Repeat CT imaging was requested, suspecting evolving COVID-19 lung involvement or a pulmonary embolism (PE) (Figure 3). The findings were consistent with acute heart failure, with no classic COVID-19 lung changes or PE. The repeat troponin increased (from 116 to 705 ng/L), despite normal renal function with no new electrocardiogram (ECG) changes. A formal transthoracic echocardiogram (TTE) revealed new biventricular failure with a dilated, impaired right ventricle [tricuspid annular plane systolic excursion (TAPSE) 0.9 cm] and global left ventricular systolic dysfunction [left ventricular ejection fraction (LVEF) 44%] with severe tricuspid regurgitation. She required the addition of an inotrope (milrinone), alongside low dose noradrenaline. The combination of careful diuresis and continuous positive airway pressure (CPAP) allowed safe offloading and she improved quickly. The addition of cardiogenic shock to her clinical picture required her to be discussed and subsequently transferred to our tertiary cardiac centre for suspected COVID-19 myocarditis, despite the negative tests thus far.
Whilst awaiting transfer, screens for autoimmune, vasculitis, blood borne viruses and atypical respiratory infections (including legionella) were reviewed and all negative. All her microbiological samples were negative. She was reviewed by multiple specialties: gastroenterology; dermatology; cardiology; and rheumatology. With no clear unifying diagnosis or underlying medical history and a lingering suspicion of COVID-19 a third swab was performed, despite improving oxygenation, which was positive.
After transfer, she spent two nights in cardiac critical care and was weaned off noradrenaline and milrinone. Her myocarditis screen was negative and a computed tomography coronary angiogram (CTCA) excluded an acute coronary syndrome. Her other symptoms had resolved and her serial TTE showed progression towards normal cardiac function. The cardiac magnetic resonance imaging (cMRI) was performed once she was COVID-19 negative on swab; this was three weeks after her symptoms. The result was essentially normal by the time it had been performed. Despite this the diagnosis was still considered COVID-19 myocarditis, with no other identifiable cause found. The patient’s symptoms continued to resolve, and she was successfully discharged home, after making a good recovery, after a total of 29 days in hospital (Figure 4). There were no concerns over compliance to treatment or adverse events we were aware of, throughout her hospital stay.

All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). The patient’s consent was obtained, in writing, for the use of identifiable data related to this manuscript.
Discussion
As the evidence emerges, COVID-19 appears to be a more heterogeneous disease, making the diagnosis difficult, especially in those with a paucity of pulmonary involvement (6). Published case reports comment on extra-pulmonary manifestations, however often these patients have the classic acute severe respiratory failure caused by COVID-19. To improve awareness of extra-pulmonary COVID-19, we aim to discuss the incidence, investigations and specific management, using this case as an example.
A recent review of 2,023 patients found the incidence of diarrhoea (49.5%) and vomiting (66.7%) to be much higher than initially thought. Other gastrointestinal symptoms reported included anorexia (up to 50.2%), nausea (up to 29.4%), abdominal pain (up to 6%) and gastrointestinal bleeding (up to 13.7%). Gastrointestinal symptoms were even occasionally the first symptom, sometimes in the absence of fever or cough, leading to potential delays and difficulties in diagnosis, such as in this case (7). Similar findings, from the USA, found that gastrointestinal symptoms were the predominant presenting complaint in 20.3% of patients and the initial presenting symptom in 14.2% of patients (3).
Coronavirus can be detected in faeces by RT-PCR (8). A meta-analysis revealed that 48.1% of stool samples were positive for virus ribonucleic acid (RNA), and they often remained positive, even once respiratory specimens were negative (8). In our centre, like many others, testing stool samples in this manner is not possible. Therefore, an appropriate knowledge of the COVID-19 gastrointestinal presentations, exclusion of differentials (Table 2) and advice from gastroenterologists is required.
The largest study of COVID-19 cutaneous manifestations, by Casas et al., reviewed 375 patients with suspected or confirmed COVID-19. Rashes were categorised into five major groups: urticarial lesions (19%); livedo or necrosis (6%); vesicular eruptions (9%); pseudo-chilblain (19%); and other maculopapules (47%). Maculopapular lesions, as demonstrated by this patient, were associated with more severe COVID-19 illness (4). However, it was noted that urticarial and maculopapular lesions may not aid diagnostics, due to their commonality and multiple potential causes (4).
Another method of categorising cutaneous lesions are by splitting them up into two groups: viral exanthems (an immune response to viral nucleotides); and cutaneous lesions (a systemic consequence of COVID-19) in particular vasculitis and thrombotic vasculopathy) (9). Ideally, a biopsy is required to further classify and investigate rashes, however this was not feasible in our case.
Most rashes in critical care are manifestations of underlying systemic disease. This patient has a predisposition to one autoimmune disease (coeliac disease) and should be investigated for others. Drug reactions with rashes are common in critical care; however, in this case the rashes were noticed prior to admission. No new rashes or evolution of the existing rashes were noted. The differential diagnosis required exclusion of allergic reactions, autoimmune, vasculitis and connective tissue diseases (Table 2).
The patient’s sudden respiratory deterioration, rising troponin and B-type natriuretic peptide (BNP) levels, and new biventricular failure, suggested this was a new cardiac event. However, initial bloods were confounded by shock and acute kidney injury (AKI). The difficulty here is the timing of the myocarditis. It is possible that the pre-hospital fluid losses (diarrhoea, vomiting and pyrexia) may have concealed any clinical evidence of acute cardiac failure. The fluids she received, although significant in volume, were felt to be appropriate in a 30-year-old, with no known cardiac disease. The appearances on the CT scan were in keeping with pulmonary oedema rather than pulmonary COVID-19. The acute cardiac failure could have been myocarditis or decompensation of a pre-existing undiagnosed cardiomyopathy. This combined with cardiogenic shock was the rationale for specialist centre referral. With a normal ECG, no cardiac risk factors and the absence of chest pain, an acute coronary syndrome was felt to be unlikely.
Cardiac complications are a common feature amongst COVID-19 patients and can present in numerous ways: acute coronary event; acute heart failure; arrhythmias; and myocarditis (10). A study of 416 patients with COVID-19 showed that cardiac injury was present in 19.7% of cases. These patients had higher levels of complications (58.5% vs. 14.7%), and significantly higher mortality (51.2% vs. 4.5%) compared to patients without cardiac injury respectively (11). Another study identified that 27.8% of patients exhibited myocardial injury, as indicated by raised troponin levels, as in this case (12). Of those with acute cardiac injury, 94% had CT features consistent with COVID-19 lung infiltration. This suggests isolated myocarditis without significant pulmonary involvement is rare (5).
At present, the incidence of COVID-19 myocarditis is not evident in the literature. Our patient had no cardiac family history or past medical history, and a myocarditis screen was negative. She had normal coronary arteries, and a cMRI was normal, however, this was performed for logistic reasons after she was RT-PCR negative (active transplant centre). It is possible if the cMRI was performed earlier it may have shown myocarditis.
Radiological changes, primarily on CT, have been shown to be more sensitive (98%) than RT-PCR (71%) (13). Potential sources of error are: sampling technique; specimen source; and sampling timing, ideally within 2 to 7 days of symptom onset (14). In this case the timing of testing was within the ideal range, and a possible cause for the negative results could be sampling technique or site.
The CT scans taken after the patient’s respiratory deterioration are shown and reported in Figure 3.
The potential pitfalls in the case report are as follows. Firstly, as the evidence base expands for COVID-19, clinicians are becoming more aware of the various presentations. At the time of writing this was our first case of extra-pulmonary COVID-19, and the evidence base at the time (May 2020) was still expanding, making the diagnosis challenging. Secondly the potential errors from COVID-19 swab testing are now more apparent. False negatives from inappropriate sampling techniques and timing are now more defined (14). It is possible this could have occurred with the first two swabs, and that she may have indeed been positive for COVID-19 on presentation. The use of CXR/CT scans as a screening tool for COVID-19 is widespread, however, the use in those without pulmonary symptoms, as in this case is less defined, however, combined with negative swab results we believed at the time she was initially COVID-19 negative. The underlying myocarditis may have been masked by sepsis and hypovolaemia on admission. Bedside screening tests, such as lung ultrasound or echocardiography may have helped identify, the cardiogenic component sooner, for example whilst in A&E. Thirdly, although it is the consensus opinion of multiple specialists, without histological samples, it is not possible to categorically confirm this case report as COVID-19 myocarditis.
A correct diagnosis of COVID-19 has significant implications for the patient, staff and the institution. For the patient, although no targeted effective treatment is currently available, as research progresses a potential drug treatment may be developed. Early diagnosis also avoids unnecessary investigation and treatments, which may have side effects. Additionally, a positive diagnosis has a psychological impact on the patient and alters isolating guidelines for relatives.
For staff, appropriate personal protective equipment (PPE) is essential for health and safety. This relies on an accurate diagnosis of COVID-19 and depends on the procedures performed, e.g., CPAP which is an aerosol generating procedure. This is especially important during a pandemic to minimise staff shortages through sickness.
Institutionally, many hospitals have COVID-19 and non-COVID-19 wards, and therefore appropriate placement depends upon correct early diagnosis. The risk of cross infection to staff and other patients is a significant consideration, especially for those with risk factors for COVID-19.
The original, classic pulmonary COVID-19, will remain the most common clinical presentation, however clinicians may be faced with atypical, extra-pulmonary COVID-19 presentations. As the numbers of cases reduce, making this infrequent atypical diagnosis will become increasingly challenging. Methodical clinical assessment, appropriate exclusion of differential diagnoses and early involvement of appropriate medical specialties is essential. A strong index of suspicion for atypical COVID-19 remains imperative for correct patient management and staff safety.
Patient perspective
Following discharge the patient was re-united with her family and expressed her utmost gratitude to the care that she had received. A thank you card was received addressed to all the staff at Chelsea and Westminster Hospital, involved with her care.
Acknowledgments
The authors wish to thank the Chelsea & Westminster NHS Foundation Trust personnel, the Chelwest AICU COVID-19 consortium and especially the Adult Intensive Care Unit nurses, for the delivery of personalised care to all patients admitted with COVID-19 infection. The authors would also wish to thank the CW Plus charity for invaluable support throughout the COVID-19 outbreak. We would like to thank the patient for providing her informed written consent for this case report.
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at http://dx.doi.org/10.21037/jeccm-20-81
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jeccm-20-81). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). The case report was written during the COVID-19 infection outbreak as part of the clinical care the patient received at Chelsea & Westminster NHS Foundation Trust. As the case report involves routinely collected non-identifiable clinical data, no ethical approval was required under the UK policy framework for Health and Social Care. The patient’s consent was obtained, in writing, for the use of identifiable data related to this manuscript.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Guan WJ, Ni ZY, Hu Y, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382:1708-20. [Crossref] [PubMed]
- Doyen D, Moceri P, Ducreux D, et al. Myocarditis in a patient with COVID-19: a cause of raised troponin and ECG changes. Lancet 2020;395:1516. [Crossref] [PubMed]
- Redd WD, Zhou JC, Hathorn KE, et al. Prevalence and Characteristics of Gastrointestinal Symptoms in Patients With Severe Acute Respiratory Syndrome Coronavirus 2 Infection in the United States: A Multicenter Cohort Study. Gastroenterology 2020;159:765-767.e2. [Crossref] [PubMed]
- Galván Casas C, Català A, Carretero Hernández G, et al. Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol 2020;183:71-7. [Crossref] [PubMed]
- Inciardi RM, Lupi L, Zaccone G, et al. Cardiac Involvement in a Patient With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol 2020;5:819-24. [Crossref] [PubMed]
- Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 2020;395:507-13. [Crossref] [PubMed]
- Tian Y, Rong L, Nian W, et al. Review article: gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther 2020;51:843-51. [Crossref] [PubMed]
- Cheung KS, Hung IFN, Chan PPY, et al. Gastrointestinal Manifestations of SARS-CoV-2 Infection and Virus Load in Fecal Samples From a Hong Kong Cohort: Systematic Review and Meta-analysis. Gastroenterology 2020;159:81-95. [Crossref] [PubMed]
- Suchonwanit P, Leerunyakul K, Kositkuljorn C. Cutaneous manifestations in COVID-19: Lessons learned from current evidence. J Am Acad Dermatol 2020;83:e57-60. [Crossref] [PubMed]
- Bansal M. Cardiovascular disease and COVID-19. Diabetes Metab Syndr 2020;14:247-50. [Crossref] [PubMed]
- Shi S, Qin M, Shen B, et al. Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol 2020;5:802-10. [Crossref] [PubMed]
- Guo T, Fan Y, Chen M, et al. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol 2020;5:811-8. [Crossref] [PubMed]
- Fang Y, Zhang H, Xie J, et al. Sensitivity of Chest CT for COVID-19: Comparison to RT-PCR. Radiology 2020;296:E115-7. [Crossref] [PubMed]
- Yang Y, Yang M, Shen C, et al. Evaluating the accuracy of different respiratory specimens in the laboratory diagnosis and monitoring the viral shedding of 2019-nCoV infections. medRxiv 2020. doi: 10.1101/2020.02.11.20021493. [Crossref]
Cite this article as: Sokhi J, Khera J, El-Hibri F, Palfreeman C, Perera G, Alwan S, Rosenberg A, Christie LE. Non-pulmonary manifestations of coronavirus disease 2019 (COVID-19): case report. J Emerg Crit Care Med 2021;5:9.


