
Inotrope dose may replace serum lactate in the definition of septic shock in resource-limited settings
Introduction
The third international consensus definitions for sepsis and septic shock (Sepsis-3) define septic shock as “a subset of sepsis in which particularly profound circulatory, cellular, and metabolic abnormalities are associated with a greater risk of mortality than with sepsis alone” (1). Operationally patients with septic shock are defined as requiring vasopressor support to maintain a mean arterial pressure ≥65 mmHg, with a serum lactate of >2.0 mmol/L after adequate fluid resuscitation.
The Sepsis-3 definitions are an attempt to better risk stratify patients with infection, in the hope of better directing appropriate treatment strategies. In particular, the addition of hyperlactataemia to the definition of septic shock, identifies a cohort of patients at higher risk of mortality than patients who require vasopressor support but do not have hyperlactataemia. This addition has not been without criticism, most notably because the cohorts from which the data was derived for the Sepsis-3 definitions were predominantly from high-income countries. Subsequent studies from Brazil and from South Africa (from the same cohort of patients presented in this study) have however confirmed that hyperlactataemia does identify a group of patients at higher risk of death in both Latin America and sub-Saharan Africa (2,3). The optimal lactate cut-off was however greater than the 2.0 mmol/L cut-off used in Sepsis-3 in both these studies (4.0 mmol/L in Brazil and 4.5 mmol/L in South Africa).
While the above results broadly validate the concept that hyperlactataemia identifies a subset of patients at higher risk of death, the concern remains that in lower- and middle-income countries serum lactate measurements may not be freely available or may add unnecessary cost to the treatment of patients in resource-limited settings. We thus hypothesised that other organ dysfunction or demographic variables may be able to similarly discriminate between higher-risk and lower-risk subgroups of patients with sepsis and hypotension requiring vasopressor support. We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/jeccm-20-54).
Methods
Study design/setting
This study was a further analysis of a study designed to evaluate the utility of the addition of hyperlactataemia to the definition of septic shock (as per Sepsis-3) (3). It was retrospective observational study of patients with sepsis and hypotension requiring vasopressor support, admitted to a 12-bedded multidisciplinary ICU in Durban, KwaZulu-Natal, South Africa.
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study protocol was approved by the University of KwaZulu-Natal Biomedical Research Ethics Committee BREC (BE-457/17) and the KwaZulu-Natal Department of Health under (HRKM 475/17) and individual consent for this retrospective analysis was waived.
Participants/data size
The methodology has been detailed previously (3). All adult patients (18 years and above) with sepsis and hypotension requiring inotropic support were included in the study. Patients were excluded if they did not have a serum lactate measured. Data collection proceeded retrospectively from the date of ethics approval until the power calculated sample size of 170 patients had been achieved (February 2018 to March 2017). Biochemical and clinical data presented is admission data unless otherwise specified. Lactate was measured by the GEM Premier 3000 blood gas machine (Instrumentation Laboratory, Werfen Group, Spain). Unless otherwise specified the terms inotropic and vasopressor support refer to the use of the “inopressors” adrenaline and noradrenaline as “pure” vasopressors (phenylephrine or vasopressin) or inotropes (dobutamine) were not used in the management of septic shock in the study ICU during the study period. Adrenaline and noradrenaline were treated as equipotent for the purpose of calculating “inotrope” dose.
Statistical methods/variables/measurement
Statistical analysis was performed using IBM SPSS Statistics for Windows, version 25.0 and R, version 3.5.1. Categorical variables were described as percentages and compared using the Chi-square test or Fisher’s exact test, where appropriate. Continuous data were described using median and interquartile range (IQR) and compared using the Mann-Whitney U test. Receiver operating characteristic (ROC) curves were constructed for lactate and the 6 predictor variables comprising the components of the SOFA score. Optimal cut-off points were established for each of the variables by determining the point closest to the (0,1) corner in the ROC plane. The areas under the ROC curve (AUC) were compared using DeLong’s test. As previously described the optimal cut-off point for lactate was 4.5 mmol/L in the study cohort (3). This was used as the reference cut-off for serum lactate for the analyses presented in this paper. However, where informative, comparisons with the serum lactate cut-off of 2.0 mmol/L, as used in Sepsis-3, were also reported.
Multivariable logistic regression analysis was performed using demographic variables that were significant (P<0.05) on univariate analysis, serum lactate and all 6 comparator organ dysfunction variables, with lactate and the organ dysfunction variables initially being evaluated as continuous variables. As it is difficult to clinically utilise the results of such an analysis that uses continuous variables, a subsequent multivariable logistic regression analysis was conducted with the lactate and organ dysfunction variables dichotomised using the optimal cut-off points calculated above.
The net reclassification improvement for prediction of ICU mortality was calculated using the optimal inotropic cut-off compared to both a lactate cut-off of 2.0 and 4.5 mmol/L. Mortality risk was stratified using the following categories: <25.0%, 25.0–49.9%, 50.0–74.9% and ≥75.0%. Predicted mortality was derived from the final multivariable logistic regression analysis described above.
The correlation between serum lactate and inotrope dose was evaluated using Pearson and Spearman correlation.
Results
Of the 170 patients included in the study, 84 (49.5%) died in ICU. The median age of the cohort was 42 years (IQR 30–58), with the median age of survivors (38 years, IQR 26–53) being significantly lower than that of non-survivors (48 years, IQR 34–62, P<0.001). Ninety-four (55.3%) of patients were female, with females having a significantly higher mortality than males (59.5% vs. 36.8%, P=0.003). The majority of patients (122, 71.8%) were referred from a surgical discipline, with the remainder being medical patients. Abdominal sepsis was the most frequent source of sepsis (95 patients, 55.9%), followed by pneumonia (44 patients, 25.9%), and skin and soft tissue infection (24 patients, 14.1%). The median SOFA score on admission was 10 (IQR 8–12), with 167 (98.2%) patients requiring mechanical ventilation and 24 (14.1%) requiring renal replacement therapy. The median ICU length of stay was 5 days (IQR 3–7). Adrenaline was the most frequently used vasopressor/inotrope (162 patients, 95.3%), with the remaining patients receiving noradrenaline.
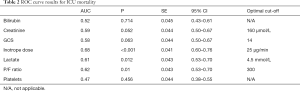
Table 1 provides univariate analyses for serum lactate and comparator organ dysfunction variables for ICU mortality.

Full table
ROC curve analyses were performed for all the variables in Table 1. The results are shown in Table 2. Inotropic dose had the highest AUC of 0.68, which was 0.07 greater than the AUC for lactate. This difference was however not statistically significant when compared using DeLong’s test (P=0.134). Optimal cut-off points were established for each of the variables by determining the point closest to the (0,1) corner in the ROC plane. These are also listed in Table 2. Due the shape of the ROC curve and poor performance of bilirubin and platelet count, optimal cut-off points were not determined, and these variables were not included in further analysis. The ROC curves for serum lactate and inotropic dose are shown in Figure 1.
Full table
Table 3 shows the categorical univariate analyses for lactate and the comparator organ dysfunction variables for ICU mortality. ICU mortality was 39.3% in patients with a serum lactate of ≤4.5 mmol/L and 36.0% in those requiring and inotropic dose of ≤25 µg/min, in contrast, for those with a lactate of >4.5 mmol/L ICU mortality was 59.3% and was 64.2% with an inotropic dose >25 µg/min.

Full table
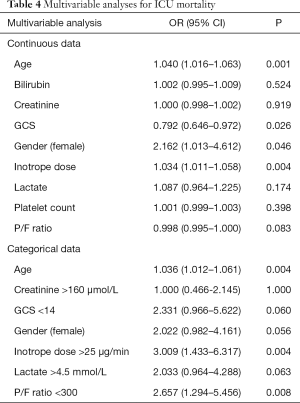
The results of the multivariable logistic regression analyses for mortality are shown in Table 4. Analysis (A) utilises continuous data and analysis (B) represents the data dichotomised using the calculated optimal cut-offs.
Full table
The NRI for those with events (ICU mortality) was 8.5% when inotropic dose (at a cut-off of 25 µg/min) was compared to lactate at threshold of 2.0 mmol/L, while the NRI for those without events (ICU survival) was 5.9%, giving a total NRI of 14.4% (P=0.183). When comparing inotropic dose and lactate at a threshold of 4.5 mmol/L the NRI for those with events was 4.9% and for those without events was 1.2%, giving a total NRI of 6.1% (P=0.518).
The Pearson correlation for serum lactate and inotropic dose was 0.476 (P<0.001) and Spearman’s rho was 0.409 (P<0.001), suggesting a moderate association between the 2 variables.
Discussion
The results of this study suggest that inotrope dose (in µg/min) is at least equivalent to hyperlactataemia in risk-stratifying patients with sepsis and hypotension requiring inotropic support, in terms of ICU mortality. Both serum lactate and inotrope dose (as continuous variables) were significantly associated with ICU mortality on univariate analysis, as was a serum lactate >4.5 mmol/L and an inotrope dose >25 µg/min. Using the abovementioned cut-offs, the ICU mortality was 39.3% in patients with a serum lactate below the cut-off and 59.3% above the cut-off. The corresponding figures for inotrope dose were 36.0% and 64.2% respectively. While inotrope dose had the highest AUC on ROC analysis at 0.68, this did not differ significantly from that of lactate (0.61). Similarly, while inotrope dose numerically improved the net reclassification of mortality risk compared to both a lactate cut-off of 2.0 and 4.5 mmol/L, with an improvement in reclassification of both patients with events and without events, these differences were not statistically significant. On multivariable analysis, however, inotrope dose was an independent predictor of ICU mortality both as a continuous and a categorical variable, whereas lactate was not a significant predictor on either analysis.
Critical care is dominated by syndromes: septic shock, acute respiratory distress syndrome, and acute kidney injury, to name but a few. The current scourge of negative clinical trials in critical care may be attributable to the heterogenous nature of these ICU syndromes: patients with varied disease profiles, including mortality risk, are being inappropriately grouped together and subjected to interventions that may have varying effects in different patient populations. In this regard, the efforts of the Sepsis-3 authors to differentiate patients with “septic shock” into lower- and higher-risk groups with the addition of hyperlactataemia to the diagnostic criteria for serum lactate, must be commended. Similarly, from a clinical perspective, differentiating patients into high- and low-risk subgroups may allow clinicians to appropriately utilise scarce resources, including critical care bed allocation.
In low- and middle-income countries access to serum lactate measurements may be limited or absent, due to cost or logistical factors. The use of serum lactate to differentiate between higher- and lower-risk subgroups of patients with sepsis and hypotension requiring vasopressor support, precludes or delays clinicians and researchers in resource-limited settings from accessing the benefits of appropriate risk-stratification. In these settings a simple clinical parameter that does not require any additional laboratory data and allows for equivalent risk stratification would be preferable to serum lactate. Based on the findings of this study, inotrope dose fulfils these criteria. We would thus argue that inotrope dose may be used instead of serum lactate for the diagnosis of septic shock in situations where lactate measurements are not freely available. If the findings of this study are validated in additional patient populations, including large global cohorts, we should suggest replacing serum lactate in the diagnosis of septic shock with inotrope dose.
The inotrope dose reported in this study is not a weight-based dose. The concern is that the applicability of the study findings may be limited in patients at the extremes of weight. Conversely, the strength of a non-weight-based dose lies in not requiring the patient’s weight, which is a particular advantage in situations where bed scales or detailed medical records (including weight) are unlikely to be available and precludes the flawed estimation of body weight. Further studies that utilise ideal body weight, as estimated from patient height (which is easily measured even in resource-limited settings), should be conducted to determine whether a weight-based inotrope cut-off is superior to a non-weight-based cut-off.
The study ICU utilised predominantly adrenaline as an inopressor. Only a small subset of patients received noradrenaline as this has limited availability in the study country. Due to broad dose-equivalency between adrenaline and noradrenaline in terms of µg/min or µg/kg/min, the findings are likely to be generalisable to institutions that utilise either agent (4). A possible concern, however, is the potential of adrenaline to increase lactate production (5-7). This may have confounded the performance of serum lactate as a predictor of mortality. The correlation between inotrope dose and serum lactate was, however, only moderate, suggesting that this was not a significant concern. The study ICU does not utilise dobutamine, vasopressin or phenylephrine in the management of patients with septic shock. The study findings are thus not generalisable to settings that utilise these agents and further studies are required in these settings. There may be variability in blood pressure targets, and thus inotrope doses, between physicians and patients. While this study didn’t specifically assess the appropriateness of the blood pressure targets and inotrope doses utilised, the study ICU titrates inotrope doses according to Surviving Sepsis Guidelines, thus reducing inter-physician variability. Where a physician chooses a higher target and thus a higher inotrope dose, however, this may still be an important summary indicator of the patient’s overall haemodynamic status.
The median age of the patients in the study was only 42 years, which is remarkably young compared to intensive care cohorts from other settings, especially high-income countries (1,2,8,9). There was also a predominance of abdominal sepsis, which contrasts to the usual predominance of respiratory infections in high-income cohorts (8). The study population may thus represent a subset of patients with a particular response to inotropes that may differ from other cohorts. This illustrates the need to conduct critical care trials in several settings before generalising results.
Conclusions
Both serum lactate >4.5 mmol/L and inotrope dose (adrenaline or noradrenaline) >25 µg/min are significant predictors of ICU mortality in patients with sepsis and hypotension requiring vasopressor support. We propose that the diagnostic criteria for septic shock in resource-limited settings be amended as follows: patients with septic shock are defined as requiring vasopressor support to maintain a mean arterial pressure ≥65mmHg, with either hyperlactataemia or a vasopressor dose of >25 µg/min of adrenaline/noradrenaline after adequate fluid resuscitation.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/jeccm-20-54
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jeccm-20-54
Peer Review File: Available at http://dx.doi.org/10.21037/jeccm-20-54
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jeccm-20-54). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study protocol was approved by the University of KwaZulu-Natal Biomedical Research Ethics Committee BREC (BE-457/17) and the KwaZulu-Natal Department of Health under (HRKM 475/17) and individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Singer M, Deutschman CS, Seymour C, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA 2016;315:801-10. [Crossref] [PubMed]
- Besen BAMP, Romano TG, Nassar AP, et al. Sepsis-3 definitions predict ICU mortality in a low–middle-income country. Ann Intensive Care 2016;6:107. [Crossref] [PubMed]
- Elhouni A, De Vasconcellos KJSSAMJ. The utility of hyperlactataemia in the definition of septic shock: Evaluating the Sepsis-3 definitions in a sub-Saharan African intensive care unit. S Afr Med J 2019;109:880-4. [Crossref]
- Vincent JL, Moreno R, Takala J, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 1996;22:707-10. [Crossref] [PubMed]
- Gjedsted J, Buhl M, Nielsen S, et al. Effects of adrenaline on lactate, glucose, lipid and protein metabolism in the placebo controlled bilaterally perfused human leg. Acta Physiol (Oxf) 2011;202:641-8. [Crossref] [PubMed]
- Levy B, Perez P, Perny J, et al. Comparison of norepinephrine-dobutamine to epinephrine for hemodynamics, lactate metabolism, and organ function variables in cardiogenic shock. A prospective, randomized pilot study. Crit Care Med 2011;39:450-5. [Crossref] [PubMed]
- Totaro RJ, Raper RF. Epinephrine-induced lactic acidosis following cardiopulmonary bypass. Crit Care Med 1997;25:1693-9. [Crossref] [PubMed]
- Vincent J-L, Sakr Y, Sprung CL, et al. Sepsis in European intensive care units: Results of the SOAP study. Critical Care Medicine 2006;34:344-53. [Crossref] [PubMed]
- Vincent J-L, Marshall JC, Ñamendys-Silva SA, et al. Assessment of the worldwide burden of critical illness: the Intensive Care Over Nations (ICON) audit. Lancet Respir Med 2014;2:380-6. [Crossref] [PubMed]
Cite this article as: Elhouni AA, de Vasconcellos K. Inotrope dose may replace serum lactate in the definition of septic shock in resource-limited settings. J Emerg Crit Care Med 2020;4:33.



