
Prevalence of deep venous thrombosis in ventilated COVID-19 patients: a mono-center cross-sectional study
Introduction
The spectrum of illness severity of COVID-19 is very wide: mild infection (no signs of lung involvement) is reported in approximately 80 percent, severe illness (e.g., with dyspnea or hypoxia) is reported in approximately 15 percent and critical disease (e.g., with respiratory failure, shock, or multiorgan dysfunction) is reported in 5 percent (1,2). The clinical manifestations of this disease may suggest that SARS-CoV-2 predominantly targets the respiratory tract. Consequently, respiratory failure due to severe bilateral pneumonia and/or acute respiratory distress syndrome (ARDS) has been reported as a major cause of death (1). Therefore, initial supportive therapy at our intensive care unit (ICU), Jessa Hospital, Hasselt, Belgium, focused mainly at respiratory support with (non)-invasive ventilatory support with oxygen- and positive end-expiratory pressure (PEEP).
On the 13th of March, the first COVID-19 patient was admitted to the ICU in our hospital. Within a few days the admissions at our COVID-19 ICU unit grew exponential. During the placement of a dialysis catheter in the femoral vein in one patient, we noticed a large deep vein thrombosis (DVT) proximal in both common femoral veins. This patient had no clinical signs of DVT. We hypothesised that thrombo-embolic phenomenon’s may be another (at the time of writing) less-well known deadly complication of a severe COVID 19 infection. To test this hypothesis, we decided to evaluate the prevalence of deep venous thrombosis (DVT) in our intubated and mechanically ventilated COVID-19+ patients. We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/jeccm-20-62).
Methods
This mono-center, investigator-initiated, observational, cross-sectional study is performed at the ICU of the Jessa Hospital, Hasselt, Belgium. All intubated and mechanically ventilated COVID-19+ patients treated at ICU ward C3 on March 29th were included in the study. Selection bias was countered by inclusion of all intubated and mechanically ventilated COVID-19. There is no information bias since the collected information was objective. Confounding was not excluded in our analysis. The study was approved by the ethical committee of the Jessa Hospital, Hasselt, Belgium (Chairperson Dr. Koen Magerman, registration number 20.35_LEC_retrospectief) on April 10th 2020 and is executed based on the Declaration of Helsinki (as revised in 2013). The study is registered on clinicaltrials.gov (NCT04338932). Written informed consent was waived in light of the urgent need to collect data in the ongoing pandemic.
The primary endpoint of this study was the prevalence of DVT in intubated and mechanically ventilated COVID-19+ patients. All patients were screened for the presence of DVT in femoral, popliteal, jugular and inferior caval vein with duplex ultrasound. A positive diagnosis of DVT was based on a venous duplex ultrasound of the full legs, inferior caval vein and jugular veins.
A possible correlation between the presence of DVT and demographic variables, medical history, symptoms at admission to ICU, laboratory findings, medical treatment and ventilator parameters was also assessed.
All patients included in this study, were treated according to the COVID-protocol of the Jessa Hospital. This protocol was developed at the beginning of March 2020 based on the latest insights on COVID-19 at that time point, mainly focusing on the pulmonary involvement of the disease. According to this protocol, all patients admitted to our ICU received an IV-infusion with glucose 5% at 60 mL/h as maintenance fluid, stress ulcer prophylaxis with Pantoprazole 40 mg intravenously daily and thrombosis prophylaxis with Nadroparin subcutaneously 0.3 mL (2850 IE) daily. Prophylactic antibiotic therapy was initiated for 5 days, using Amoxicillin-clavulanic acid 1 g intravenously 4 times a day or moxifloxacin 400 mg intravenously once daily in case of known allergy to penicillin. The protocol also comprised the administration of hydroxychloroquine orally starting with a loading dose of 400 mg twice on the first day, followed by 200 mg twice a day, for 4 days, with daily monitoring of the QTC-interval by ECG. This therapy was interrupted if the QTC-interval exceeded 500 ms since QTC-interval prolongation is one of the well-known important side-effects of hydroxychloroquine potentially causing severe cardiac arrythmias. Ventilatory support was initiated with a high-flow nasal canulla or non-invasive mechanical ventilation as long as the patient was cooperative to this treatment. In case of respiratory fatigue, patients were sedated and intubated and invasive mechanical ventilation was started according to the ARDS-network guidelines that included a protocol based on low-oxygen and high-PEEP. This was based on the first reports that the viral pneumonia caused by SARS-CoV-2 mimics an ARDS-like pattern. Sedation was performed by a combination of propofol, midazolam and piritramide aiming for the lowest level of sedation by which the patient would tolerate the mechanical ventilation. Adjustments were made guided by spO2-levels, which are continuously monitored and arterial blood gasses taken every 4 hours. In case of hypotension due to vasoplegia, norepinephrine was used as first choice vasopressor.
Descriptive statistics were presented as frequencies and percentages, while numerical variables were presented as mean ± SD. A one-way ANOVA with Bonferroni correction for multiple testing was used to calculate differences between the groups. A P value <0.05 was considered statistically significant. All analyses are conducted using the statistical software Graphpad Prism version 5.1.
Results
In total, 8 from all 12 intubated and ventilated COVID 19+ patients treated at ICU-C3 in the Jessa Hospital, Hasselt, Belgium on march 29th had already developed minimally one DVT (Table 1).

Full table
Demographic data, underlying medical conditions and symptoms at admission of all intubated and mechanically patients with COVID-19 admitted at ICU on 2020/03/29 are presented in Table 1, stratified according to the presence of DVT.
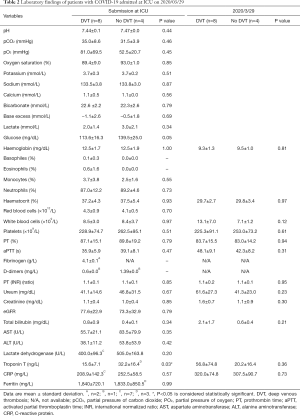
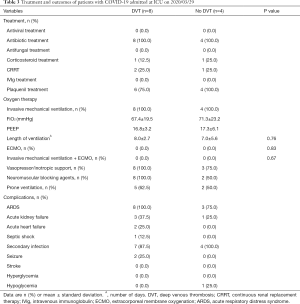
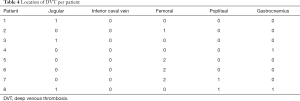
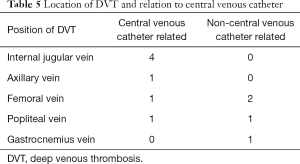
Laboratory data, stratified according to the presence of DVT, are presented in Table 2. Platelet count and coagulation test values were not significantly different. Treatment, ventilator parameters and outcomes, stratified according to the presence of DVT, are presented in Table 3. We could not detect statistically significant differences between two groups. Location of DVT per patient is shown in Table 4 and the relation of DVT with deep venous catheter is shown in Table 5.
Full table
Full table
Full table
Full table
Discussion
In this mono-centre cross-sectional study, assessment of the presence of DVT in intubated and mechanically ventilated COVID-19+ patients revealed a prevalence of DVT of more than 65%. No relevant correlations between the presence of DVT and demographic variables, medical history, symptoms at admission to ICU, laboratory findings, medical treatment and ventilator parameters could be detected. Presumably, patient groups were too small to demonstrate relevant correlations.
Virchow’s triad describes the major DVT risk factors in three categories: venous stasis, vessel injury and activation of blood coagulation. Consequently, every ICU patient is at risk for thrombo-embolic events. The incidence of DVT during ICU stay has been reported between 10% and 30% (3). Our results however suggest that COVID-19 ICU patients are at two- or even three-fold increased relative risk of developing thrombo-embolic complications.
Our results are in line with recent literature. Klok et al. found an incidence of symptomatic acute pulmonary embolism (PE), DVT, ischemic stroke, myocardial infarction or systemic arterial embolism of 31% despite systematic thrombosis prophylaxis in 184 COVID-19 patients treated admitted to ICU (4). Wang et al. reported temporary improvement in the P/F ratio (ratio of arterial oxygen partial pressure to fractional inspired oxygen concentration) after off-label usage of alteplase for COVID-19 patients suffering from ARDS and respiratory failure (5). Tang et al. found improved mortality in severe COVID-19 patients, meeting sepsis induced coagulopathy criteria or with markedly elevated D-dimer, who received anticoagulant therapy (6).
Multiple studies have proposed that SARS-CoV-2 induces a hyper-inflammatory state (7-9). It has been suggested that systemic inflammation induces endothelial injury. The injury activates the coagulation cascade and impairs fibrinolysis with disruption of endothelial barrier and loss of physiologic antithrombotic factors. This process elevates the risk for venous thrombo-embolism (VTE) significantly (10).
Application of neuromuscular blocking agents (NMBA) to facilitate oxygenation and ventilation in severe ARDS (acute respiratory distress syndrome) may be another precipitating factor for the development of DVT due to patient immobilization (11). Boddi et al. already concluded that administration of NMBA is the strongest independent predictor for DVT (12). In this study, all 8 patients who developed a DVT received intermittent NMBA boluses to optimize oxygenation and ventilation and to facilitate prone-ventilation.
Prolonged mechanical ventilation (>7 days) is another well-known risk factor for the development of DVT (13). In this study, mean length of mechanical ventilation was 8 days. Furthermore, mechanical ventilation with high PEEP reduces blood flow to the heart. This may facilitate stasis of venous blood and can promote formation of a DVT despite adequate prophylaxis (12,14,15). A high PEEP-strategy was applied in all our mechanically ventilated patients, according to the local COVID-protocol. Theoretically, these ventilator settings may have additionally increased patient’s intra-thoracic pressure and impaired venous return. However, a positive correlation between high PEEP and development of upper extremity DVT’s could not be demonstrated (16).
Central venous catheterization is another well-known risk factor for DVT due to the vessel injury and venous stasis (17). In this study, 63.6% (7/11) of the proved DVT’s were associated with an indwelling catheter (Table 5).
Obesity is another risk factor for DVT (18). This might be explained by blood stasis due to the impaired venous return caused by the chronic increased intra-abdominal pressure. Furthermore, a relative hypercoagulability state may be present within the obese population due to an elevation of pro-thrombotic factors and impaired fibrinolysis (19). In this study, 58% (7/12) of included patients were overweight and 33% (4/12) were obese.
Dehydration has also been proposed to elevate the risk of DVT since a higher viscosity of the blood may decrease venous return (20). In total, 40% of COVID-19+ patients report dehydration prior to hospitalization (1). Furthermore, according to the surviving sepsis guidelines for COVID-19 patients, we also applied a fluid restrictive policy (17). These two factors may have led to relative dehydration of our patients and an elevated risk of developing DVT’s.
Last but not least, SARS-CoV-2 itself may induce coagulopathy and an increased risk of VTE’s. Multiple viruses have been associated with thrombo-embolic events (21). Several mechanisms have been proposed to explain this association: alteration of the level of a variety of coagulation proteins, viral platelet binding with subsequent platelet activation, viral induction of endothelial injury resulting in activation of coagulation and fibrinolysis impairment, due to increased levels of plasminogen activator inhibitor-1 (21). Very recently, The American Association of Hematology also recognizes the possibility that a severe COVID-19 infection may elicit a fulminant activation of coagulation (22).
This study also contains some limitations. First, due to cross-sectional design of the study, there is a lack of temporal relationship between the development of DVT and the different risk factors that were evaluated. Second, due to the small sample size, the prevalence of DVT may be overestimated in this study. Third, for small sample sizes, it is unrealistic to look for correlations across large number of parameters. Fourth, patients in this study only received a prophylactic dose of low-molecular-weight heparin (LMWH). A follow-up study would be interesting to assess the effect of the measures taken to lower the risk of DVT’s. Finally, the generalisability of these results may be questioned due to the single-centre design.
In conclusion, our results suggest that the prevalence of DVT in critically ill ICU patients with COVID-19 treated with a prophylactic dose of LMWH is more than 60%. Consequently, the risk of VTE complications in this patient group is very high. In the light of these considerations, we suggest the use of thigh-high graduated compression (elastic) stockings (GCS) and intermediate-dose LMWH for thrombosis prophylaxis in all COVID-19 patients with daily anti-Xa-activity monitoring to provide adequate therapy since an important interpatient variability has been reported. Furthermore, we suggest to routinely screen all COVID-19 patients admitted to ICU for DVT twice weekly via venous duplex ultrasound.
Future research should focus on the mechanisms underlying these observations.
Acknowledgments
Funding: The study was funded solely by departmental funding.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/jeccm-20-62
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jeccm-20-62
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jeccm-20-62). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was approved by the ethical committee of the Jessa Hospital, Hasselt, Belgium (Chairperson Dr. Koen Magerman, registration number 20.35_LEC_retrospectief) on April 10th 2020 and is executed based on the Declaration of Helsinki (as revised in 2013). The study is registered on clinicaltrials.gov (NCT0433893). Written informed consent was waived in light of the urgent need to collect data in the ongoing pandemic.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Wang D, Hu B, Hu C, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 2020;323:1061-9. [Crossref] [PubMed]
- Chan JFW, Yuan SY, Kok KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lances 2020;395:514-23. [Crossref] [PubMed]
- Attia J, Ray JG, Cook DJ, et al. Deep vein thrombosis and its prevention in critically ill adults. Arch Intern Med 2001;161:1268-79. [Crossref] [PubMed]
- Klok FA, Kruip MJHA, Van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res 2020;191:145-7. [Crossref] [PubMed]
- Wang J, Hajizadeh N, Moore EE, et al. Tissue Plasminogen Activator (tPA) Treatment for COVID-19 Associated Acute Respiratory Distress Syndrome (ARDS): A Case Series. J Thromb Haemost 2020;18:1752-5. [Crossref] [PubMed]
- Tang N, Bai H, Chen X, et al. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost 2020;18:1094-9. [Crossref] [PubMed]
- Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020;395:497-506. [Crossref] [PubMed]
- He L, Ding Y, Zhang Q, et al. Expression of elevated levels of pro-inflammatory cytokines in SARS-CoV-infected ACE2+ cells in SARS patients: relation to the acute lung injury and pathogenesis of SARS. J Pathol 2006;210:288-97. [Crossref] [PubMed]
- Mahallawi WH, Khabour OF, Zhang Q, et al. MERS-CoV infection in humans is associated with a pro-inflammatory Th1 and Th17 cytokine profile. Cytokine 2018;104:8-13. [Crossref] [PubMed]
- Branchford BR, Carpenter SL. The role of inflammation in venous thromboembolism. Front Pediatr 2018;6:142. [Crossref] [PubMed]
- Tripathi SS, Hunter JM. Neuromuscular blocking drugs in the critically ill. Cont Educ Anaesth Crit Care Pain 2006;6:119-23. [Crossref]
- Boddi M, Barbani F, Abbate R, et al. Reduction in deep vein thrombosis incidence in intensive care after a clinician education program. J Thromb Haemost 2010;8:121-8. [Crossref] [PubMed]
- Ibrahim EH, Iregui M, Prentice D, et al. Deep vein thrombosis during prolonged mechanical ventilation despite prophylaxis. Crit Care Med 2002;30:771-4. [Crossref] [PubMed]
- Luecke T, Pelosi P. Clinical review: Positive end-expiratory pressure and cardiac output. Crit Care 2005;9:607-21. [Crossref] [PubMed]
- Luecke T, Pelosi P, Quintel M. Haemodynamic effects of mechanical ventilation. Anaesthesist 2007;56:1242-51. [Crossref] [PubMed]
- Al-Saffar F, Gupta E, Siddiqi F, et al. Is There Any Association between PEEP and Upper Extremity DVT? Crit Care Res Pract 2015;2015:614598. [Crossref] [PubMed]
- Alhazzani W, Møller MH, Arabi YM, et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Crit Care Med 2020;48:e440-e469. [Crossref] [PubMed]
- Ageno W, Becattini C, Brighton T, et al. Cardiovascular risk factors and venous thromboembolism: a meta-analysis. Circulation 2008;117:93-102. [Crossref] [PubMed]
- Rosito GA, D'Agostino RB, Massaro J, et al. Association between obesity and a prothrombotic state: the Framingham Offspring Study. Thromb Haemost 2004;91:683-9. [Crossref] [PubMed]
- Patel K, Chun LJ. Deep Venous Thrombosis (DVT). 2019. Available online: https://emedicine.medscape.com/article/1911303-overview
- Goeijenbier M, Van Wissen M, Van de Weg C, et al. Review: Viral infections and mechanisms of thrombosis and bleeding. J Med Virol 2012;84:1680-96. [Crossref] [PubMed]
- Lee AYY, Connors JM, Kreuziger LB, et al. COVID-19 and Coagulopathy: Frequently Asked Questions. 2020. Available online: https://www.hematology.org/covid-19/covid-19-and-coagulopathy?fbclid=IwAR0S0za_BmQvnWRXpRmCefSiDZVa1GUcMa53J5v32eDNSBlxpLg1EncOCo0
Cite this article as: Pellens B, Romont M, Van Tornout M, De Mey N, Dubois J, De Pauw I, Ramaekers D, Stessel B. Prevalence of deep venous thrombosis in ventilated COVID-19 patients: a mono-center cross-sectional study. J Emerg Crit Care Med 2020;4:31.


