
Challenges in procedural sedation and analgesia in the emergency department
Introduction
Procedural sedation and analgesia (PSA) constitutes a rather common practice aiming to relieve patients’ anxiety, discomfort and pain during invasive diagnostic and therapeutic procedures or diagnostic imaging (1). PSA aims to suppress the patients’ level of consciousness while maintaining purposeful response to verbal commands, a state known as moderate sedation (2). The emergency department (ED), along with the intensive care unit (ICU), are the two most common settings of PSA administration (3).
PSA administration in the ED has been gaining attention amongst the medical community as it concerns various specialties, namely Emergency Medicine, Anesthesiology, Pediatrics, Orthopedic Surgery etc. The most common procedures associated with its use include orthopedic manipulations, abscess incision and drainage, wound debridement and direct current cardioversion (1).
The ideal agent for PSA in the ED should provide anxiolysis, analgesia and amnesia in a rapid, predictable manner, with minimal side effects, and should have a quick recovery phase (1). One of the most important obstacles in the implementation of PSA in the ED is the variability of sedative drugs’ availability worldwide (4). This results in the application of local or national guidelines, regulated by the availability of sedative agents and personnel, making the implementation of universal guidelines challenging (1,5).
There is a plethora of controversies regarding PSA in emergency settings, amongst them the appropriate level of sedation, the selection of the proper pharmaceutical agent and the management of potential adverse effects. It is thus essential to manage ED patients using a multidisciplinary team approach and establish standardized protocols for PSA administration. Towards this end, the European Society of Anesthesiology (ESA) and the European Board of Anesthesiology launched guidelines regarding PSA in adults in 2018 (1), that were twice updated since (6,7).
In the present review, we discuss the assessment and monitoring necessary for PSA administration, the most commonly available and used pharmaceutical agents and the required knowledge, skills, and interventions that are necessary to manage potential complications related to PSA in the ED setting.
Assessment and monitoring for PSA
Pre-sedation assessment
In the ED, pre-sedation assessment is usually challenging due to specific, setting-related parameters, such as urgently-needed anesthesia for patients with potentially severe comorbidities. Whenever feasible, a pre-sedation assessment should be conducted, including a focused history and physical examination and a review of comorbidities, medications and allergies (8). The most widely used scheme is the American Society of Anesthesiologists (ASA) Physical Status Classification, aiming to identify patients at risk of adverse events (9).
Pre-procedural fasting
The depression of upper-airway reflexes during anesthesia is a major risk factor for the development of aspiration pneumonia (10). Thus, ASA recommends pre-procedural fasting before the administration of sedation (11). However, it is easily understandable that pre-procedural fasting cannot be considered routinely applicable in the ED. Fortunately, the moderate sedation achieved during PSA administration in the ED does not suppress protective airway reflexes (10). Moreover, no deaths from aspiration have been reported in the literature associated with PSA administration in the ED (12). Consequently, the American College of Emergency Physicians (ACEP) does not consider recent food intake as a contraindication for PSA in the ED (2).
Monitoring equipment
In line with ACEP guidelines, it is standard practice to continuously monitor the patient’s cardiac rhythm (electrocardiogram), pulse rate, oxygen saturation, and respiratory rate during PSA in the ED (2). Blood pressure is typically measured in a non-invasive manner every five minutes (13). In addition to the above, suction devices, supplemental oxygen and advanced monitoring equipment should be readily available (2). Finally, advanced airway equipment, resuscitative medications and vascular access supplies should be easily accessible (2). Moreover, core body temperature monitoring is recommended by ASA in patients who receive moderate or deep sedation, unless restricted by the patient’s status, the available equipment or the procedure itself (14).
Capnography (end-tidal carbon dioxide monitoring) is used for the detection of early signs of respiratory depression (15,16), however its routine use for PSA in the ED is debatable. In a meta-analysis conducted in 2011, Waugh et al. (17) reported an increased rate of detection of respiratory depression (17.6 times higher) when comparing capnography with standard monitoring alone. On the other hand, it has been suggested that the detection of apnea or transiently decreased ventilation by capnography could lead to unnecessary interventions, like positive pressure ventilation and may result in complications such as aspiration and gastric insufflation (15). Additionally, capnography implementation has not been proven effective in foreseeing desaturation episodes or in decreasing the incidence of clinically important adverse effects (18-23). Therefore, capnography is not considered standard of care for PSA in the ED.
Sedation scales, responsiveness monitoring
Numerous sedation/responsiveness scales are available for use during PSA, nevertheless none has been proven superior to the other in evaluating sedation efficacy (24). The most frequently cited scales are the Observer’s Assessment of Alertness/Sedation Scale, the Richmond Agitation-Sedation Scale and the Ramsay Sedation Scale (25,26). In addition to using established scales to monitor a patient’s responsiveness, patients’ distress, ventilatory adequacy and procedural recall can be used as indirect measures of PSA effectiveness (27).
Sedative agents and their adverse effects
A vast amount of sedative drugs are available worldwide, with their accessibility varying amongst different countries. As a result, the establishment of globally applicable guidelines is challenging. A review article published by Gozal and Mason in 2010 (28) addressed this specific topic and although they elaborated on pediatric sedation their conclusions may be applicable on adult populations too. Based on randomized controlled trials (RCTs) and meta-analyses, they propose that traditional sedation agents, like ketamine, midazolam, propofol, etomidate and nitrous oxide should maintain their primary role in PSA. Furthermore, they suggest that some combinations, such as ketamine-midazolam and ketamine-propofol may be beneficial due to their synergistic effect (28). Characteristics of the most commonly used anesthetic agents are shown in Table 1.
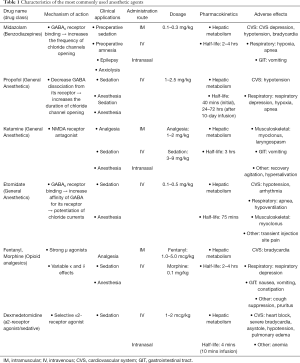
Full table
Propofol
Propofol is the most commonly used sedative drug because of its fast onset of sedation and predictability of effect duration. Hepatic clearance is high, so accumulation of the drug is negligible even after repeated administrations or continuous infusion. The initial effects of propofol include mild sedation and amnesia, leading to deep sedation when administered in high doses. Doses needed for induction of anesthesia are 1–2.5 mg/kg, whereas plasma concentration needed for sedation is 1–2 mcg/mL (29).
Propofol can be used in a variety of procedures. It has been showcased that it provides patient satisfaction, better quality of sedation and faster recovery in gastrointestinal endoscopy interventions (30). Therefore, a new position statement from the British Society for Gastroenterology and the Royal College of Anesthetists proposes propofol administration for the execution of complex gastrointestinal endoscopies (31). Moreover, in a study by Mathieu et al. in 2009 (32), a sedation regimen using propofol was more successful than the previously preferable midazolam/analgesic combination in achieving optimal conditions for dislocated hip prosthesis reductions.
Target Control Infusion (TCI) pumps allow for more precise control over the level of sedation, although there is a shortage of data concerning the use of TCI in the ED (33). A RCT, termed “The Propofol Target-Controlled Infusion in Emergency Department Procedural Sedation” (PROTEDS) is underway, aiming to assess the safety and efficacy of TCI-administered propofol, as well as the practicalities of its use in the ED for dislocated shoulder reduction (34). A similar idea, called “The Computer Assisted Propofol Sedation System” (CAPS), was implemented as a substitute of the bolus propofol injection by anesthesiologists, but due to high cost and low profitability the project was discontinued by the manufacturer. However, the experience of hospitals where CAPS was used was positive (35).
Propofol administration in the ED may result in various adverse effects, such as apnea, hypoxia, and hypotension (36). Therefore, the presence of personnel qualified in airway management and resuscitation is essential. Homfray et al. assessed the adverse effects of propofol, midazolam/morphine and other sedative agents in a population of 704 elderly patients in their safety analysis in 2018. The overall sentinel adverse effect rate (hypoxia, apnea, hypotension) was 2.6%, but no safe conclusions could be drawn when comparing propofol with other agents. It is interesting to note that in this particular population, a bolus propofol dose of 0.5 mg/kg with a subsequent bolus dose of 0.25 mg/kg, if sedation was unsatisfactory, resulted in the lowest rate of adverse effects. However, the overall rate of adverse events noted during propofol administration was high, accentuating the need for further research in sedation practices (37).
Midazolam
Midazolam is a fast-acting benzodiazepine with a short half-life (38). It has anxiolytic, sedative and anterograde amnestic effects in lower doses, while deep sedation is achieved when administered in higher doses (39). It has no proven analgesic properties, however there are limited indications that intravenous administration of midazolam prior to nasogastric tube insertion leads to reduced self-reported pain (40). Midazolam is the pharmaceutical agent most commonly used for PSA in the ED in the United Kingdom (41). The recommended dosing range is 0.1–0.3 mg/kg in divided doses of 1–2 mg until the desired level of sedation is achieved. Midazolam has an immediate onset of effect, a half-life of 2–4 hours and its effects last for roughly 20 minutes (42).
Midazolam in the ED can be administered through the intravenous or intranasal route. Intravenous administration enables precise dosing and faster onset of action, with easy titration of the divided doses. Intranasal application is gaining attention in the pediatric population, as well as in combative patients, while also being a viable option in patients with difficult venous access. Intranasal delivery of the drug is achieved with devices known as “atomizers”. The volume of midazolam administered per nostril should ideally be between 0.2–0.5 mL/kg, with the maximum dose being 1 mL/kg. Since high volumes of drug administration cannot be achieved through the intranasal route, its use is restricted to patients weighing less than 50 kg (43).
The most common adverse effects of midazolam include hypotension, hypoxia and apnea with the incidence estimated at 6.1, 51.2 and 51.4 per 1,000 patients, respectively. Vomiting is a less frequent side effect. Cardiovascular depression, presenting as hypotension or bradycardia, is rare when midazolam is given as a sole agent (44). In case of life-threatening adverse events, a selective GABAA antagonist called flumazenil should be administered. It is usually given intravenously or intranasally, in doses of 8.0–15.0 mcg/kg. Its onset of action is fast, but its effect duration brief, rendering re-sedation possible in 20 minutes (45).
Ketamine
Ketamine is a phencyclidine derivative that causes a dissociative anesthetic state which differs from “classic” sedation. It is a NMDA receptor antagonist with strong analgesic effects (46) that can be used as a first line agent for the management of agitated patients as it acts faster than benzodiazepines and haloperidol (40). In a study by Riddell et al., ketamine displayed a favorable hemodynamic profile, as no significant changes in blood pressure or pulse rate were documented. The administered dosage was 4–5 mg/kg IM with an onset of action at 5 minutes, or an IV dose of 1–2 mg/kg. This single center study was conducted in a population exhibiting a high percentage of methamphetamine abuse, thus presenting a severe limitation in generalizing its conclusions (47).
Ketamine can be used as a sole agent during arduous procedures, such as orthopedic manipulations. In a cohort study by Newton et al., including 92 ED patients, ketamine provided optimal analgesia and sedation in the majority of the patients. Initial dose was 0.5 mg/kg, repeated after 5 minutes, if sedation was deemed inadequate by the treating physician, to a maximum of 1 mg/kg (48).
In addition to intravenously and intramuscularly, ketamine can also be administered intranasally. The recommended doses are 3–9 mg/kg for sedation and 1–2 mg/kg for analgesia. Maximal concentration of ketamine is 50 mg/mL, limiting the total dose in adults to 100–200 mg. For that reason, intranasal administration of ketamine in this patient population is reserved for analgesic purposes (49).
The unique properties of ketamine allow for preservation of spontaneous ventilation and hemodynamic stability. Its administration can be invaluable in resource-poor settings, where ideal monitoring of respiratory and cardiovascular functions cannot be achieved. Ketamine was well tolerated and deemed satisfactory by patients and physicians alike in a study of 54 children and 54 adults treated for burns and orthopedic injuries in a tertiary hospital in Tanzania (50).
A crucial drawback of ketamine administration is its numerous adverse effects. Those include emergence reactions, such as agitation during recovery from anesthesia, hypersalivation, clonic movements, laryngospasm and vomiting. The most recurrent amongst them is recovery agitation which can be minimized or prevented by the administration of benzodiazepines (49,51).
Ketofol
“Ketofol” is a ketamine and propofol mixture, which gained popularity because of the theoretical advantages of adding the analgesic properties of ketamine to the hypnotic properties of propofol. Propofol also seems to have the ability to counteract some of the most important ketamine adverse effects; emergence phenomena, nausea and vomiting. Moreover, the addition of ketamine results in reduced dose of propofol needed during PSA, decreasing the possibility for respiratory depression. The recommended dosage is 0.5 mg/kg of a 1:1 mixture, followed by another dose of 0.5 mg/kg. A dose of 0.25 mg/kg is used afterwards for maintenance of sedation (52).
Two recent meta-analyses studied the safety and efficacy of ketofol in comparison with propofol as a single agent. The study by Yan et al., which included six RCTs, concluded that there was no statistically significant difference in safety between the two pharmaceutical agents. While the overall danger of developing adverse events during PSA was not reduced, less respiratory events were documented in the ketofol group compared with the propofol group. However, the authors emphasize the fact that their meta-analysis included a heterogenous population (adults and children) and a variety of ketamine/propofol ratios were used in their included studies (53). Jalili et al. in 2016, attempted to overcome the aforementioned limitation, by only analyzing studies conducted in adult populations. Even so, they reached the same conclusion; ketofol appears to be a safe alternative to propofol for PSA (36).
Ketofol was also compared to a combination of propofol and fentanyl in a cohort study of 136 trauma patients in a university hospital in Iran. The administered doses were 1 mcg/kg of fentanyl, 1.0 mg/kg of ketamine and 0.5 mg/kg of propofol. There were more apneic and hypoxemic events in the group which received the propofol/fentanyl mixture. The authors concluded that the propofol/fentanyl combination can be recommended over ketofol because of its superior sedation quality and analgesic effects (54).
The combination of ketamine and propofol appears to be safe in patients with cardiogenic shock, due to the lesser influence of ketamine on hemodynamic parameters (46). A new PPIIM protocol (Preoxygenation, Pretreatment, Induction of anesthesia and paralysis, Intubation, Mechanical ventilation) was evaluated by Chalkias et al. in 2018, including 31 patients suffering from acute myocardial infraction and presenting signs of cardiogenic shock. They compared those patients to a historical control group, matched by patient characteristics, that received standard RSI (rapid sequence induction) with either midazolam, propofol or etomidate. The protocol included the use of fentanyl as pretreatment (0.7 mcg/kg) and midazolam (0.02 mg/mg), ketamine (0.35 mg/mg) and propofol (0.5 mg/kg) for the induction of anesthesia. They concluded that the patients who received the ketamine/propofol (ketofol) combination had improved hemodynamic parameters (systolic, diastolic and mean arterial pressure) and a higher rate of survival in comparison with the control group (55).
Dexmedetomidine
Dexmedetomidine is a highly selective α2 receptor agonist. The effects of central α2 receptor potentiation include anxiolysis and sedation, without concurrent respiratory depression. Its half-time is only 4 minutes when administered in 10-minute infusions but can be prolonged through longer administration (56).
A study in patients with anterior shoulder dislocation showcased better analgesic effects and faster recovery with dexmedetomidine administration when compared with a midazolam-fentanyl combination (57). Recently, dexmedetomidine was confirmed to be safe for use in upper gastrointestinal endoscopy (58). Intranasal administration of dexmedetomidine is possible, with the proposed dosage being 1–2 mcg/kg. The onset of sedation for adults is reported to be 45 minutes, with a 90–105 minutes interval for achieving peak sedation (59).
Adverse events include heart block, severe bradycardia and asystole, due to unopposed vagal stimulation. There are minimal changes in ventilatory rate and tidal volume (56).
Etomidate
Etomidate is a sedative-hypnotic drug that potentiates the activity of GABAA receptors. It has minimal effects on cardiovascular function, but suppresses cortisol production for 4–8 hours, a factor that limits its use in continuous infusions (60). There are limited new studies addressing the use of etomidate in PSA.
In advanced endoscopic procedures etomidate has been shown to be as efficacious as propofol in achieving sedation with less cardiopulmonary adverse effects (61). Furthermore, ventilation depression is less pronounced compared with propofol, but episodes of apnea may occasionally occur. Its effects on the cardiovascular system are minimal, although caution is advised when etomidate is used in hypovolemic patients as it can lower blood pressure significantly (42). In a RCT by Miner et al., propofol was found to be more efficient than etomidate with similar adverse event rates, except for myoclonus rate which was significantly higher in the etomidate group (20% versus 1.2%). It was hypothesized that the lower success rates of etomidate were related to myoclonus events (62).
In a study for the treatment of various pediatric orthopedic injuries, etomidate combined with fentanyl showed similar sedation levels to ketamine but required administration of multiple doses because of its short duration of action. The authors concluded that they would prefer ketamine to etomidate since it provides longer sedation time (63). In another study in an adult population with large joint dislocations, no difference was found in the conditions of reduction when etomidate, in doses of 0.1 or 0.5 mg/kg until adequate sedation was achieved, was compared to ketamine. However, patients in the ketamine group experienced fewer desaturation episodes and interventions to maintain airway patency were less frequently needed (64).
Opioids
Opioid drugs act on opioid receptors throughout the central nervous system, providing an analgesic effect, as well as hypnotic and sedative effects in high doses. They are mostly used as adjuncts to sedatives that do not possess analgesic effects. The most commonly used opioids for sedation and analgesia are fentanyl and morphine.
The usual dose of opioids is 1.0–5.0 mcg/kg and 0.1 mg/kg for fentanyl and morphine respectively. Fentanyl has a faster onset and shorter duration of action, rendering it easier to titrate than morphine and a sensible choice for PSA. Furthermore, fentanyl has the considerable advantage of being available for intranasal administration in both children and adults. The adverse events associated with opioid administration are numerous, including respiratory depression, cough suppression, nausea and vomiting, bradycardia, constipation and pruritus; most of these adverse effects are dose related. It is important to mention that the addition of fentanyl to midazolam results in a 4-fold increase in unwanted respiratory events in comparison to midazolam administration alone (65).
In case of severe adverse events, a competitive opioid receptor (mu) antagonist called naloxone should be administered, usually through the intravenous or intramuscular route. Its onset of action is 2 minutes and its appropriate dose ranges between 0.4 to 1 mg in adults and 0.1 mg/kg in children. However, patients who have been injected with high doses of fentanyl may subsequently require a higher dose of naloxone administration (66).
Inhaled anesthetics
Currently, potent inhaled anesthetics are rarely used in the ED, owing to the need for anesthesia machines and trained personnel. However, inhaled agents have gained attention as sedatives in the emergency setting, as they alleviate the need for intravenous access. This fact makes inhaled anesthetics a viable option for sedation, especially in the pediatric population (67).
There are a few case cohort studies regarding the use of sevoflurane after dental trauma in the pediatric ED (68,69). Kim et al. presented their experience in managing agitated children with dental trauma using sevoflurane. The inhaled agent was administered through an intranasal cannula and the end-tidal sevoflurane was monitored in order to maintain adequate sedation. No complications were documented and all children maintained spontaneous ventilation throughout the procedure (69).
Nitrous oxide (N2O) is another inhaled sedative which can be used for PSA in the emergency setting. It is typically administered as a 50–50% or 70–30% N2O-oxygen mixture, with at least 30% oxygen to avoid hypoxemia, while a concentration of at least 30% of N2O is necessary for adequate sedation. This inhaled agent can be administered by a demand-valve mask or mouthpiece held by the patient (70). Potential adverse effects of nitrous oxide include nausea, vomiting and respiratory depression (71). Brodsky et al. (72), advise against nitrous oxide administration in patients with pneumothorax, immunosuppression or possible pregnancy. Self-administration of nitrous oxide by the patient is often preferred in order to avoid over-sedation (73). Sivaramakrisnan et al., concluded that a combination of nitrous oxide and midazolam was preferable to each individual agent alone during PSA. The lower doses of midazolam seemed to improve patient safety and enhance predictability of sedation depth (74).
Oxygen supplementation
Several publications have shown that the use of general anesthetic and sedative drugs during PSA increases the risk of cardiorespiratory depression and upper respiratory tract obstruction (2,13). Current guidelines highlight the importance of constant monitoring of respiratory function, as well as oxygen supplementation in both moderate and severe sedation in order to prevent any possible adverse effects (2,75). Supplemental oxygen should be provided to all hypoxemic patients unless there are certain contraindications (2,13,75,76). Several RCTs have concluded that hypoxemia rates are markedly reduced following the administration of supplemental oxygen in patients under moderate or severe sedation (13,75).
Although oxygen supplementation during PSA reduces hypoxemia rates, it can obscure the identification of other adverse effects such as hypoventilation and upper respiratory tract obstruction (75). Remarkably, results from studies have demonstrated that regular administration of supplemental oxygen increases the risk of cardiovascular complications (75). The most common respiratory adverse events during PSA are hypoxemia (40.2 per 1,000 sedations) and apnea (12.4 per 1,000 sedations), although they rarely require endotracheal intubation (55). Deitch et al. reported that high-flow oxygen supplementation or apneic oxygenation can significantly decrease the incidence of hypoxia during PSA (77).
Even though the beneficial effects of supplemental oxygen during PSA in the ED have been established, it still remains unclear which route of supplemental oxygen administration is the most beneficial in lowering hypoxemia risk (2).
Nasal high flow (NHF) therapy
NHF therapy is a new oxygen administration method that has been proven to possess many advantages over conventional oxygen treatments (78,79). It was introduced in neonates and infants, but it has been increasingly used in adult populations in recent years (78,80). The oxygen device delivers a combination of heated, humidified air and oxygen at a fraction of inspired oxygen (FiO2) ranging from 21% to 100% with a flow rate up to 60 L/min (78,79,81).
Several studies have reported that NHF therapy creates a positive end-expiratory pressure (PEEP) and enhances respiratory function, while improving patients’ tolerance. Furthermore, it decreases anatomical dead space and facilitates the function of the mucociliary clearance mechanism (16,78,79).
NHF therapy can be used in patients presenting in the ED with various diseases (79,80). Currently, it is mainly used as the initial treatment in patients presenting with type I and type II acute hypoxemic respiratory failure (80). Further clinical indications include acute heart failure, acute respiratory failure in immunocompromised patients, preoxygenation for intubation and PSA (80). Interestingly, NHF is recommended as the primary method of oxygen supplementation during PSA but no guidelines have been made available regarding its application in clinical practice (78,80).
Apneic oxygenation
Apneic oxygenation is a technique used for airway management in emergency settings (82). Hypoxemia, brain anoxia and cardiovascular collapse consist the main unfavorable outcomes of patients who need urgent airway intubation as a result of a primary pulmonary pathology (82-84). Apneic oxygenation was designed to hinder and reduce the development of such significant adverse non-rebreather events especially during anesthesia induction (83). The results of several published data have shown that the use of apneic oxygenation during tracheal intubation minimizes the risk of arterial oxygen desaturation and extends the interval prior to apnea initiation (84). It is important to emphasize that a patent airway is recommended for the induction of apneic oxygenation technique (83).
Various techniques are used for the induction of apneic oxygenation including high flow nasal cannula oxygenation, Venturi masks, nasotracheal tubes and face masks. Published data have established the superiority of the high flow nasal canula method over a non-rebreather facemask in improving arterial oxygen saturation (82).
Apneic oxygenation is not recommended in patients presenting with hyperkalemia, pulmonary hypertension, increased intracranial pressure and metabolic acidosis (84). Finally, it is contraindicated in cases of facial trauma and airway obstruction (83).
Rapid sequence induction (RSI)
RSI of anesthesia is a method used for tracheal intubation in high risk patients (85). The principal elements of RSI include anesthesia induction followed by neuromuscular blocking agents administration and intubation (86). The main goals of the RSI technique are to reach a standard level of anesthesia while simultaneously preventing aspiration by immediate intubation (86). Specifically, it is considered highly important to initiate the intubation within a minute following the administration of anesthesia (86).
Pre-oxygenation is regarded a vital step in the RSI technique, as it has been proven to be beneficial on apnea tolerance (85). The use of ventilation masks has been suggested in cases of obese and critically ill patients in order to avoid the development of hypoxemia throughout the apneic phase (87). The results of various publications have emphasized the significance of this method, particularly for patients with respiratory decompensation, pregnant women and children who have difficulty in tolerating apnea (86).
Application of cricothyroid pressure is considered important in order to reduce the risk of gastric content regurgitation (86). Interestingly, several studies have highlighted the questionable efficacy of applying cricoid pressure (87,88).
The major induction drugs used in RSI method include propofol, etomidate, ketamine and thiopental. Propofol and thiopental are first-line choices whereas ketamine and etomidate are considered alternatives (86). Ketamine is recommended as first line therapy in patients with shock; etomidate is preferred in heart failure patients (86). However, the selection of the induction drug, the proper dosage and the preferred route of administration remain controversial (86).
The administration of sedative agents should be followed by infusion of a muscle relaxant agent, such as succinylcholine or rocuronium, to facilitate tracheal intubation (86,89). Although succinylcholine had been historically considered the agent of choice, currently it has been substituted by rocuronium (86).
Recently, several alternatives of the RSI method have been introduced, making the establishment of official guidelines challenging, although it is still the preferred method in emergency situations (86,90).
Resources, quality and training
As previously mentioned, procedural sedation is associated with a number of adverse effects and complications. Therefore, clinicians involved should be adequately skilled in sedation induction, patient monitoring and management of adverse events (76).
Various reporting protocols, aiming to optimize PSA practice, are used in order to record procedural outcomes, adverse effects and physicians’ efficacy. The International Committee for the Advancement of Procedural Sedation recommended the use of a new tool for monitoring sedation outcomes in 2018 titled “Tracking and Reporting Outcomes of Procedural Sedation” (TROOPS). TROOPS may be adopted by any interested physician in order to monitor their sedation practices or as a research tool (91).
Respiratory depression and hemodynamic instability are considered the most common and important adverse events of PSA. A meta-analysis including 9,652 PSAs in the ED, reported that apnea, hypotension and bradycardia may occur in 12.4, 15.2 and 6.5 out of 1,000 sedations respectively (44). In rare cases, cardiac arrest may succeed these complications (92). Consequently, PSA providers must be well trained in recognizing and treating life threatening complications that may arise during sedation. That implies that they should be proficient in antidote administration (flumazenil, naloxone), advanced airway management, IV cannulation and cardiac arrest management (Advanced Life Support) (76,93). Finally, physicians should be able to perform pre-procedural equipment checks and accurately assess the patient’s medical condition.
ESA recommended the use of a written test as well as a practical course on mannequins to ascertain the sufficiency of trained physicians before their exposure in real-life scenarios (76). Likewise, ASA advises that deep sedation should be restrictedly administered by “qualified anesthesia professionals” or non-anesthesiologist physicians who have been officially trained (94).
The guidelines by ESA also indicate the groups of patients that need to be taken care of by an anesthesiologist, should they undergo PSA, according to their age (>70 years old), their physical status (ASA 3 or 4), or their comorbidities, including severe cardiovascular disease, presence of obstructive sleep apnea, severe renal or hepatic disease and morbid obesity (9).
Conclusions
In conclusion, a variety of logistic and practical difficulties, such as drug availability and appropriate personnel training, prevent the implementation of global guidelines regarding PSA in the ED. Further multicentric studies with large datasets might contribute in establishing protocols that can be effectively applied in an emergency setting. Non-anesthesiologist physicians’ training on sedation should be prioritized in order to administer PSA in a safe and efficient manner.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Athanasios Chalkias) for the series “Recent Advances in Critical Emergency Medicine: From Pathophysiology to Clinical Practice” published in Journal of Emergency and Critical Care Medicine. The article was sent for external peer review organized by the Guest Editor and the editorial office.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jeccm-19-212). The series “Recent Advances in Critical Emergency Medicine: From Pathophysiology to Clinical Practice” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Hinkelbein J, Lamperti M, Akeson J, et al. European Society of Anaesthesiology and European Board of Anaesthesiology guidelines for procedural sedation and analgesia in adults. Eur J Anaesthesiol 2018;35:6-24. [Crossref] [PubMed]
- Practice Guidelines for Moderate Procedural Sedation and Analgesia. 2018: A Report by the American Society of Anesthesiologists Task Force on Moderate Procedural Sedation and Analgesia, the American Association of Oral and Maxillofacial Surgeons, American College of Radiology, American Dental Association, American Society of Dentist Anesthesiologists, and Society of Interventional Radiology. Anesthesiology 2018;128:437-79. [Crossref] [PubMed]
- Chawla N, Boateng A, Deshpande R. Procedural sedation in the ICU and emergency department. Curr Opin Anaesthesiol 2017;30:507-12. [Crossref] [PubMed]
- Report of the International Narcotics Control Board on the Availability of Internationally Controlled Drugs: Ensuring Adequate Access for Medical and Scientific Purposes [Internet]. [cited 2020 Apr 7]. Available online: https://apps.who.int/medicinedocs/en/m/abstract/Js21969en/
- Mason KP, editor. Pediatric Sedation Outside of the Operating Room: A Multispecialty International Collaboration [Internet]. 2nd ed. New York: Springer-Verlag; 2015 [cited 2020 Apr 7]. Available online: https://www.springer.com/gp/book/9781493913893
- Barends CRM, Absalom AR, Struys MMRF. Drug selection for ambulatory procedural sedation. Curr Opin Anaesthesiol 2018;31:673-8. [Crossref] [PubMed]
- Raeder J. Procedural sedation in ambulatory anaesthesia: what’s new? Curr Opin Anaesthesiol 2019;32:743-8. [Crossref] [PubMed]
- Sutherland JR, Conway A, Sanderson EL. Preprocedural Assessment for Patients Anticipating Sedation. Curr Anesthesiol Rep 2020;10:35-42. [Crossref]
- Doyle DJ, Garmon EH. American Society of Anesthesiologists Classification (ASA Class). In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 [cited 2020 Apr 7]. Available online: http://www.ncbi.nlm.nih.gov/books/NBK441940/
- Molina JAD, Lobo CA, Goh HK, et al. Review of studies and guidelines on fasting and procedural sedation at the emergency department. Int J Evid Based Healthc 2010;8:75-8. [PubMed]
- Practice Guidelines for Preoperative Fasting and the Use of Pharmacologic Agents to Reduce the Risk of Pulmonary Aspiration: Application to Healthy Patients Undergoing Elective Procedures: An Updated Report by the American Society of Anesthesiologists Task Force on Preoperative Fasting and the Use of Pharmacologic Agents to Reduce the Risk of Pulmonary Aspiration. Anesthesiology 2017;126:376-93. [Crossref] [PubMed]
- Bahn EL, Holt KR. Procedural sedation and analgesia: a review and new concepts. Emerg Med Clin North Am 2005;23:503-17. [Crossref] [PubMed]
- Tobias JD, Leder M. Procedural sedation: A review of sedative agents, monitoring, and management of complications. Saudi J Anaesth 2011;5:395-410. [Crossref] [PubMed]
- Standards for Basic Anesthetic Monitoring | American Society of Anesthesiologists (ASA) [Internet]. [cited 2020 Apr 7]. Available online: https://www.asahq.org/standards-and-guidelines/standards-for-basic-anesthetic-monitoring
- Green SM, Pershad J. Should Capnographic Monitoring Be Standard Practice During Emergency Department Procedural Sedation and Analgesia? Pro and Con. Ann Emerg Med 2010;55:265-7. [Crossref] [PubMed]
- Helviz Y, Einav S. A Systematic Review of the High-flow Nasal Cannula for Adult Patients. Crit Care [Internet]. 2018 Mar 20 [cited 2020 Apr 7];22. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5861611/
- Waugh JB, Epps CA, Khodneva YA. Capnography enhances surveillance of respiratory events during procedural sedation: a meta-analysis. J Clin Anesth 2011;23:189-96. [Crossref] [PubMed]
- Dewdney C, MacDougall M, Blackburn R, et al. Capnography for procedural sedation in the ED: a systematic review. Emerg Med J 2017;34:476-84. [Crossref] [PubMed]
- Deitch K, Miner J, Chudnofsky CR, et al. Does end tidal CO2 monitoring during emergency department procedural sedation and analgesia with propofol decrease the incidence of hypoxic events? A randomized, controlled trial. Ann Emerg Med 2010;55:258-64. [Crossref] [PubMed]
- Mohr NM, Stoltze A, Ahmed A, et al. Using continuous quantitative capnography for emergency department procedural sedation: a systematic review and cost-effectiveness analysis. Intern Emerg Med 2018;13:75-85. [Crossref] [PubMed]
- Sivilotti MLA, Messenger DW, van Vlymen J, et al. A comparative evaluation of capnometry versus pulse oximetry during procedural sedation and analgesia on room air. CJEM 2010;12:397-404. [Crossref] [PubMed]
- Smally AJ, Nowicki TA, Simelton BH. Procedural sedation and analgesia in the emergency department. Curr Opin Crit Care 2011;17:317-22. [Crossref] [PubMed]
- Wall BF, Magee K, Campbell SG, et al. Capnography versus standard monitoring for emergency department procedural sedation and analgesia. Cochrane Database Syst Rev 2017;3:CD010698. [PubMed]
- Williams MR, McKeown A, Dexter F, et al. Efficacy Outcome Measures for Procedural Sedation Clinical Trials in Adults: An ACTTION Systematic Review. Anesth Analg 2016;122:152-70. [Crossref] [PubMed]
- Chernik DA, Gillings D, Laine H, et al. Validity and reliability of the Observer’s Assessment of Alertness/Sedation Scale: study with intravenous midazolam. J Clin Psychopharmacol 1990;10:244-51. [PubMed]
- Frenzel D, Greim C-A, Sommer C, et al. Is the bispectral index appropriate for monitoring the sedation level of mechanically ventilated surgical ICU patients? Intensive Care Med 2002;28:178-83. [Crossref] [PubMed]
- ACEP // Unscheduled Procedural Sedation: A Multidisciplinary Consensus Practice Guideline [Internet]. [cited 2020 Apr 7]. Available online: https://www.acep.org/patient-care/policy-statements/unscheduled-procedural-sedation-a-multidisciplinary-consensus-practice-guideline/
- Gozal D, Mason KP. Pediatric Sedation: A Global Challenge [Internet]. Vol. 2010, International Journal of Pediatrics. Hindawi; 2010 [cited 2020 Apr 7]. p. e701257. Available online: https://www.hindawi.com/journals/ijpedi/2010/701257/
- Sahinovic MM, Struys MMRF, Absalom AR. Clinical Pharmacokinetics and Pharmacodynamics of Propofol. Clin Pharmacokinet 2018;57:1539-58. [Crossref] [PubMed]
- McQuaid KR, Laine L. A systematic review and meta-analysis of randomized, controlled trials of moderate sedation for routine endoscopic procedures. Gastrointest Endosc 2008;67:910-23. [Crossref] [PubMed]
- Sidhu R, Turnbull D, Newton M, et al. Deep sedation and anaesthesia in complex gastrointestinal endoscopy: a joint position statement endorsed by the British Society of Gastroenterology (BSG), Joint Advisory Group (JAG) and Royal College of Anaesthetists (RCoA). Frontline Gastroenterol 2019;10:141-7. [Crossref] [PubMed]
- Mathieu N, Jones L, Harris A, et al. Is propofol a safe and effective sedative for relocating hip prostheses? Emerg Med J 2009;26:37-8. [Crossref] [PubMed]
- Guarracino F, Lapolla F, Cariello C, et al. Target controlled infusion: TCI. Minerva Anestesiol 2005;71:335-7. [PubMed]
- Burton FM, Lowe DJ, Millar J, et al. A study protocol for a feasibility study: Propofol Target-Controlled Infusion in Emergency Department Sedation (ProTEDS)—a multi-centre feasibility study protocol. Pilot Feasibility Stud 2019;5:27. [Crossref] [PubMed]
- Lin OS, La Selva D, Kozarek RA, et al. One year experience with computer-assisted propofol sedation for colonoscopy. World J Gastroenterol 2017;23:2964-71. [Crossref] [PubMed]
- Jalili M, Bahreini M, Doosti-Irani A, et al. Ketamine-propofol combination (ketofol) vs propofol for procedural sedation and analgesia: systematic review and meta-analysis. Am J Emerg Med 2016;34:558-69. [Crossref] [PubMed]
- Homfray G, Palmer A, Grimsmo-Powney H, et al. Procedural sedation of elderly patients by emergency physicians: a safety analysis of 740 patients. Br J Anaesth 2018;121:1236-41. [Crossref] [PubMed]
- Heizmann P, Eckert M, Ziegler WH. Pharmacokinetics and bioavailability of midazolam in man. Br J Clin Pharmacol 1983;16:43S-49S. [Crossref] [PubMed]
- Basic & Clinical Pharmacology, 14e | AccessMedicine | McGraw-Hill Medical [Internet]. [cited 2020 Apr 7]. Available online: https://accessmedicine.mhmedical.com/book.aspx?bookID=2249
- Manning CT, Buinewicz JD, Sewatsky TP, et al. Does Routine Midazolam Administration Prior to Nasogastric Tube Insertion in the Emergency Department Decrease Patients’ Pain? (A Pilot Study). Acad Emerg Med 2016;23:766-71. [Crossref] [PubMed]
- Elkhodair SM, Baker EC, Glasebrook WR, et al. Emergency department procedural sedation: the London experience. Eur J Emerg Med 2015;22:407-12. [Crossref] [PubMed]
- Chalkias A, Xanthos T, Papageorgiou E, et al. Intraoperative initiation of a modified ARDSNet protocol increases survival of septic patients with severe acute respiratory distress syndrome. Heart Lung 2018;47:616-21. [Crossref] [PubMed]
- Rech MA, Barbas B, Chaney W, et al. When to Pick the Nose: Out-of-Hospital and Emergency Department Intranasal Administration of Medications. Ann Emerg Med 2017;70:203-11. [Crossref] [PubMed]
- Bellolio MF, Gilani WI, Barrionuevo P, et al. Incidence of Adverse Events in Adults Undergoing Procedural Sedation in the Emergency Department: A Systematic Review and Meta-analysis. Acad Emerg Med 2016;23:119-34. [Crossref] [PubMed]
- Chalkias A, Georgiou M, Böttiger B, et al. Recommendations for resuscitation after ascent to high altitude and in aircrafts. Int J Cardiol 2013;167:1703-11. [Crossref] [PubMed]
- Li L, Vlisides PE. Ketamine: 50 Years of Modulating the Mind. Front Hum Neurosci [Internet]. 2016 Nov 29 [cited 2020 Apr 9];10. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5126726/
- Riddell J, Tran A, Bengiamin R, et al. Ketamine as a first-line treatment for severely agitated emergency department patients. Am J Emerg Med 2017;35:1000-4. [Crossref] [PubMed]
- Newton A, Fitton L. Intravenous ketamine for adult procedural sedation in the emergency department: a prospective cohort study. Emerg Med J 2008;25:498-501. [Crossref] [PubMed]
- Tzimas P, Samara E, Petrou A, et al. The influence of anesthetic techniques on postoperative cognitive function in elderly patients undergoing hip fracture surgery: General vs spinal anesthesia. Injury 2018;49:2221-6. [Crossref] [PubMed]
- Coralic Z, Sawe HR, Mfinanga JA, et al. Ketamine procedural sedation in the emergency department of an urban tertiary hospital in Dar es Salaam, Tanzania. Emerg Med J 2018;35:214-9. [Crossref] [PubMed]
- Vasileiou PVS, Chalkias A, Brozou V, et al. Interleukin-6 as a marker of inflammation secondary to endotracheal intubation in pediatric patients. Inflammation 2013;36:1533-8. [Crossref] [PubMed]
- Lamperti M. Adult procedural sedation: an update. Curr Opin Anaesthesiol 2015;28:662-7. [Crossref] [PubMed]
- Yan JW, McLeod SL, Iansavitchene A. Ketamine-Propofol Versus Propofol Alone for Procedural Sedation in the Emergency Department: A Systematic Review and Meta-analysis. Acad Emerg Med 2015;22:1003-13. [Crossref] [PubMed]
- Aminiahidashti H, Shafiee S, Hosseininejad SM, et al. Propofol-fentanyl versus propofol-ketamine for procedural sedation and analgesia in patients with trauma. Am J Emerg Med 2018;36:1766-70. [Crossref] [PubMed]
- Chalkias A, Pavlopoulos F, Papageorgiou E, et al. Development and Testing of a Novel Anaesthesia Induction/Ventilation Protocol for Patients With Cardiogenic Shock Complicating Acute Myocardial Infarction. Can J Cardiol 2018;34:1048-58. [Crossref] [PubMed]
- Davy A, Fessler J, Fischler M, et al. Dexmedetomidine and general anesthesia: a narrative literature review of its major indications for use in adults undergoing non-cardiac surgery. Minerva Anestesiol 2017;83:1294-308. [PubMed]
- Masoumi K, Maleki SJ, Forouzan A, et al. Dexmedetomidine versus Midazolam-Fentanyl in Procedural Analgesia Sedation for Reduction of Anterior Shoulder Dislocation: A Randomized Clinical Trial. Rev Recent Clin Trials 2019;14:269-74. [Crossref] [PubMed]
- Nishizawa T, Suzuki H, Sagara S, et al. Dexmedetomidine versus midazolam for gastrointestinal endoscopy: a meta-analysis. Dig Endosc 2015;27:8-15. [Crossref] [PubMed]
- Gallego-Ligorit L, Vives M, Vallés-Torres J, et al. Use of Dexmedetomidine in Cardiothoracic and Vascular Anesthesia. J Cardiothorac Vasc Anesth 2018;32:1426-38. [Crossref] [PubMed]
- Konstantogianni E, Tsalkidou V, Dimou P, et al. Comparative study of Supreme, Cobra, and i-gel during spontaneous and controlled mechanical ventilation: a case series. Am J Emerg Med 2015;33:1524-5. [Crossref] [PubMed]
- Han SJ, Lee TH, Yang JK, et al. Etomidate Sedation for Advanced Endoscopic Procedures. Dig Dis Sci 2019;64:144-51. [Crossref] [PubMed]
- Miner JR, Danahy M, Moch A, et al. Randomized clinical trial of etomidate versus propofol for procedural sedation in the emergency department. Ann Emerg Med 2007;49:15-22. [Crossref] [PubMed]
- Dişel NR, Yilmaz HL, Sertdemir Y, et al. Etomidate Versus Ketamine: Effective Use in Emergency Procedural Sedation for Pediatric Orthopedic Injuries. Pediatr Emerg Care 2016;32:830-4. [Crossref] [PubMed]
- Salen P, Grossman M, Grossman M, et al. A comparison of ketamine versus etomidate for procedural sedation for the reduction of large joint dislocations. Int J Crit Illn Inj Sci 2016;6:79-84. [Crossref] [PubMed]
- Roback MG, Wathen JE, Bajaj L, et al. Adverse events associated with procedural sedation and analgesia in a pediatric emergency department: a comparison of common parenteral drugs. Acad Emerg Med 2005;12:508-13. [Crossref] [PubMed]
- Schiller EY, Mechanic OJ. Opioid Overdose. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 [cited 2020 Apr 9]. Available online: http://www.ncbi.nlm.nih.gov/books/NBK470415/
- Gamis AS, Knapp JF, Glenski JA. Nitrous oxide analgesia in a pediatric emergency department. Ann Emerg Med 1989;18:177-81. [Crossref] [PubMed]
- Park CH, Kim S. Survey of the sevoflurane sedation status in one provincial dental clinic center for the disabled. J Dent Anesth Pain Med 2016;16:283-8. [Crossref] [PubMed]
- Kim J, Yoo S, Kim J, et al. Using nasal cannula for sevoflurane deep sedation in emergency dental treatment. J Dent Anesth Pain Med 2015;15:11-5. [Crossref] [PubMed]
- Huang C, Johnson N. Nitrous Oxide, From the Operating Room to the Emergency Department. Curr Emerg Hosp Med Rep 2016;4:11-8. [Crossref] [PubMed]
- Luhmann JD, Schootman M, Luhmann SJ, et al. A randomized comparison of nitrous oxide plus hematoma block versus ketamine plus midazolam for emergency department forearm fracture reduction in children. Pediatrics 2006;118:e1078-86. [Crossref] [PubMed]
- Brodsky JB, Cohen EN. Adverse Effects of Nitrous Oxide. Med Toxicol 1986;1:362-74. [Crossref] [PubMed]
- Heinrich M, Menzel C, Hoffmann F, et al. Self-administered procedural analgesia using nitrous oxide/oxygen (50:50) in the pediatric surgery emergency room: effectiveness and limitations. Eur J Pediatr Surg 2015;25:250-6. [PubMed]
- Sivaramakrishnan G, Sridharan K. Nitrous Oxide and Midazolam Sedation: A Systematic Review and Meta-Analysis. Anesth Prog 2017;64:59-65. [Crossref] [PubMed]
- Maurer WG, Walsh M, Viazis N. Basic Requirements for Monitoring Sedated Patients: Blood Pressure, Pulse Oximetry, and EKG. Digestion 2010;82:87-9. [Crossref] [PubMed]
- American Society of Anesthesiologists Task Force on Sedation and Analgesia by Non-Anesthesiologists. Practice guidelines for sedation and analgesia by non-anesthesiologists. Anesthesiology 2002;96:1004-17. [Crossref] [PubMed]
- Deitch K, Chudnofsky CR, Dominici P, et al. The utility of high-flow oxygen during emergency department procedural sedation and analgesia with propofol: a randomized, controlled trial. Ann Emerg Med 2011;58:360-364.e3. [Crossref] [PubMed]
- Ischaki E, Pantazopoulos I, Zakynthinos S. Nasal high flow therapy: a novel treatment rather than a more expensive oxygen device. Eur Respir Rev 2017;26:170028. [Crossref] [PubMed]
- Mauri T, Wang YM, Dalla Corte F, et al. Nasal high flow: physiology, efficacy and safety in the acute care setting, a narrative review. Open Access Emerg Med 2019;11:109-20. [Crossref] [PubMed]
- Gotera C, Díaz Lobato S, Pinto T, et al. Clinical evidence on high flow oxygen therapy and active humidification in adults. Rev Port Pneumol 2013;19:217-27. [Crossref] [PubMed]
- Pantazopoulos I, Adamos G, Sotiriou A, et al. Nasal high flow application for perioperative support of respiratory system in adult patients. J Emerg Crit Care Med [Internet]. 2019 Nov 27 [cited 2020 Apr 7];0(0). Available online: http://jeccm.amegroups.com/article/view/5521
- Gleason JM, Christian BR, Barton ED. Nasal Cannula Apneic Oxygenation Prevents Desaturation During Endotracheal Intubation: An Integrative Literature Review. West J Emerg Med 2018;19:403-11. [Crossref] [PubMed]
- Ayten S, Halime Hanim P, Mehmet Y, et al. Apneic Oxygenation and High Flow. Int J Anesth Anesthesiol 2018;5:081.
- Wong DT, Yee AJ, Leong SM, et al. The effectiveness of apneic oxygenation during tracheal intubation in various clinical settings: a narrative review. Can J Anesth 2017;64:416-27. [Crossref] [PubMed]
- Woloszczuk-Gebicka B. RSII rapid-sequence induction of anaesthesia and intubation of the trachea. Disaster Emerg Med J 2017;2:33-8. [Crossref]
- Lauren Berkow. Rapid sequence induction and intubation (RSII) for anesthesia [Internet]. UpToDate. [cited 2020 Apr 7]. Available online: https://www.uptodate.com/contents/rapid-sequence-induction-and-intubation-rsii-for-anesthesia
- Sajayan A, Wicker J, Ungureanu N, et al. Current practice of rapid sequence induction of anaesthesia in the UK - a national survey. Br J Anaesth 2016;117 Suppl 1:i69-74. [Crossref] [PubMed]
- El-Orbany M, Connolly LA. Rapid sequence induction and intubation: current controversy. Anesth Analg 2010;110:1318-25. [Crossref] [PubMed]
- Succinylcholine vs. Rocuronium: Battle of the RSI Paralytics [Internet]. JEMS. 2019. Available online: https://www.jems.com/2019/05/13/succinylcholine-vs-rocuronium-battle-of-the-rsi-paralytics/
- Mason MA, Weant KA, Baker SN. Rapid sequence intubation medication therapies: a review in light of recent drug shortages. Adv Emerg Nurs J 2013;35:16-25. [Crossref] [PubMed]
- Roback MG, Green SM, Andolfatto G, et al. Tracking and Reporting Outcomes Of Procedural Sedation (TROOPS): Standardized Quality Improvement and Research Tools from the International Committee for the Advancement of Procedural Sedation. Br J Anaesth 2018;120:164-72. [Crossref] [PubMed]
- Mason KP, Roback MG, Chrisp D, et al. Results from the Adverse Event Sedation Reporting Tool: A Global Anthology of 7952 Records Derived from >160,000 Procedural Sedation Encounters. J Clin Med 2019;8:2087. [Crossref] [PubMed]
- Godwin SA, Burton JH, Gerardo CJ, et al. Clinical policy: procedural sedation and analgesia in the emergency department. Ann Emerg Med 2014;63:247-258.e18. [Crossref] [PubMed]
- Advisory on Granting Privileges for Deep Sedation to Non-Anesthesiologist Physicians [Internet]. [cited 2020 Apr 7]. Available online: https://www.asahq.org/standards-and-guidelines/advisory-on-granting-privileges-for-deep-sedation-to-non-anesthesiologist-physicians
Cite this article as: Raffay V, Fišer Z, Samara E, Magounaki K, Chatzis D, Mavrovounis G, Mermiri M, Žunić F, Pantazopoulos I. Challenges in procedural sedation and analgesia in the emergency department. J Emerg Crit Care Med 2020;4:27.


