
Bedside assessment and clinical utility of mean systemic filling pressure in acute care
Introduction
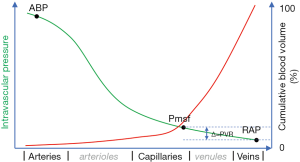
The bedside assessment of cardiovascular function in acute care has evolved substantially in recent years owing to the transition from invasive to less invasive monitoring modalities. Still, characterizing and guiding hemodynamic management remains troublesome in a substantial number of patients, especially in those in whom the origin of cardiovascular instability is not evident at first sight. Here, clinicians often tend to rely on measurement of systemic hemodynamic variables such as heart rate, arterial blood pressure (ABP), and sometimes cardiac output (CO), for assessing the adequacy of the circulation. While ABP and CO do reliably reflect the “delivery side” of the circulation that provides O2 and nutrients to the various organs and tissues (microcirculation) of the patient, it does not take the venous side of circulation into consideration which, under normal circumstances, contains the majority (about 70%) of the total blood volume (Figure 1) (1). Likewise, while the highly compliant venous side of the circulation plays a crucial role as a blood reservoir and thereby determines the venous return of blood to the heart (and thus cardiac output), it is often neglected clinically, or inappropriately reduced to the fluid balance. The mean systemic filling pressure (Pmsf) is a quantitative index of intravascular blood volume and is modifiable by vascular tone. Pmsf can nowadays be estimated at the bedside using three methods (see below). Pmsf is the upstream pressure for venous return. Hence, Pmsf may characterize the hemodynamic state and function as well as the hemodynamic response to treatment modalities, such as the administration of fluids and vasopressors. This review focusses on the theory behind Pmsf, its estimation, and probably most importantly: its current role in characterizing and monitoring hemodynamic performance in acutely ill patients.
Pmsf and the importance of the venous side of the circulation
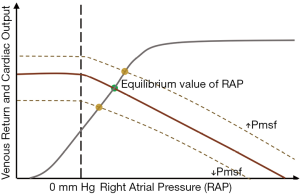
While the generation of CO itself is important for maintaining cardiovascular homeostasis, the role of the heart is often inaccurately portrayed, as it is not only the heart itself that functions as the primary “pump” of the circulation and is the sole, direct cause of the flow of blood. Instead, together with cardiac contractility and afterload (i.e., the resistance to blood flow in the peripheral circulation), preload is essential for generating flow—and thus CO. Here, the venous compartment comes into play, as cardiac preload depends on venous return (VR) of blood that flows back to the heart, and it is essentially VR that is regulated by the venous compartment (2). The important relationship between VR and CO has been elucidated by the work of Guyton in the first part of the 20th century (3), at a time when it was already known that increased ventricular stretch increases cardiac contractility (O. Frank) (4), and that increases in VR increase stroke volume (E. Starling) (5). Guyton, in a series of experiments in mechanically instrumented dogs (using right heart bypass, instrumentation of the dogs with a pump and a Starling resistor), measured right atrial pressures (RAP), as well as VR. This enabled him to plot the now well-known “venous return plots” (Figure 2) by increasing and decreasing RAP by controlling the pump speed and the height of the Starling resistor. Using this set-up, he could demonstrate that an increase in RAP was associated with a concomitant decrease in VR (and CO), and vice versa. Importantly, he revealed that the RAP functioned as a “backpressure” for venous return—an observation that was recently replicated in an elegant porcine model (6). Details on this work are excellently summarized in full detail elsewhere for the interested reader (7,8). In the original experiments by Guyton, not only RAP was altered, but another variable—the mean circulatory filling pressure (Pmcf)—was changed too, mainly by fluid administration. Here, Pmcf was defined as the “mean integrated filling pressure throughout the circulatory system when one appropriately weighs the volumes and degrees of elasticity of the different portions of the circulatory system”, and can be measured only after cessation of flow (i.e., cardiocirculatory arrest). Of note, Pmsf resembles Pmcf under normal circumstances, but given that Pmsf is related to the systemic part of the circulation only, Pmsf and Pmcf can diverge in case of specific alterations in intrathoracic or pulmonary blood volume or tone. For clarity, only the term Pmsf is used later on in this review.
To explain Pmsf in more detail, let us consider the following hypothetical situation: If the intravascular pressure were measured invasively under zero-flow conditions after complete exsanguination, it would be clear that the resulting pressure is 0 mmHg, wherever in the circulation it was measured. Subsequently, consider that blood would gradually be re-infused into the intravascular space: initially, a certain volume of blood would be necessary to fill up the vessels without exerting pressure on the vessel wall (i.e., intravascular pressure equals 0 mmHg). The blood volume required until intravascular pressure starts to increase is termed the unstressed blood volume. Importantly, different vascular compartments have different individual compliances, and the volume of blood will be distributed according to these differences. Subsequently, when more blood is re-infused, it will generate pressure on the vascular wall. This additional volume is termed the stressed blood volume and normally comprises about 25–30% of the predicted blood volume in humans (9). The mean systemic filling pressure (Pmsf) is directly proportional to the stressed blood volume and is a quantification of the “blood reservoir” of the circulation. An important factor here is the capacitance of the vascular system, which is dependent on the compliance of the overall vascular system and the different compartments, of which each compartment has its own relative compliance and vascular tone (see later). Here, the venous system is—in contrast to the arterial system—very compliant, and takes up a substantial proportion of the blood volume (≈70%). As a result, Pmsf is the pressure “representative” as a function of both the amount of blood volume and the capacitance. It may be seen as the potential energy stored in the wall of the systemic (Pmsf), or the entire (Pmcf) vascular compartment (7). Pmsf is thus the upstream pressure for venous return. As a consequence—as already suggested by Guyton (10)—Pmsf must always be higher than RAP to generate flow, i.e., to result in a “driving pressure” for venous return (PVR; PVR = Pmsf – RAP, Figure 1). Here, the role of the right heart is to keep RAP low by pumping blood to the pulmonary circulation. If RAP is kept low, VR can be maintained and thus, CO can be preserved. This view partially contradicts popular guidelines such as the Surviving Sepsis Guidelines, which until very recently advocated the universal use of rather high CVP targets (i.e., 10–15 mmHg) for the management of patients with septic shock (11).
It may be clear that Pmsf is influenced both by vascular filling—i.e., the amount of intravascular blood volume—and vascular compliance. The latter can be influenced by several factors, e.g., sympathetic and parasympathetic activity, and administration of vasoactive drugs. In particular, vasoactive drugs that influence venous tone by vasoconstriction or—dilation, may increase or decrease venous return, respectively, as Pmsf is in- or decreased in these instances. The effect of in- or decreasing Pmsf on the venous return (plot), in combination with the cardiac function curve, is given in Figure 2.
It must be stressed that the above-mentioned reasoning has been debated vehemently by different authors (12-17), mainly regarding the role of RAP either as a passive consequence or active driver of blood flow. Despite this (interesting) debate, we still lack a broad and well fitted hemodynamic model that can describe all different aspects of flow—and can still be replicated using modern experimental settings (6). Hence, until such improved models become available, the “Guytonian” model may still be the best for describing circulatory physiology—in which the role of RAP is mainly a consequence of blood flow.
How can Pmsf be measured?
In the hypothetical example above in which the circulation was stopped, it would have been relatively easy to measure Pmsf as it would only require measuring intravascular pressure anywhere within the vascular bed after equilibration. In patients with an intact circulation, it is however not straightforward to directly measure Pmsf since temporary cessation of flow can only be induced by complex and highly invasive procedures, such as cardiac fibrillation/defibrillation sequence(s) (18).
Fortunately, Pmsf can now be estimated by three indirect methods:
- Inspiratory hold maneuvers (Pmsfi). In mechanically ventilated patients, Pmsf can be assessed by application of a number of inspiratory hold maneuvers at different (increasing) airway pressures (e.g., 5, 15, 25 and 35 cmH2O) and simultaneously measuring CVP and CO (19,20). Extrapolation of the curve, and using the intercept of CVP/CO measurement pairs at a CO =0 L·min−1 results in an estimation of the Pmsf (Pmsfi), equivalent to the situation when there would be no flow. The particular disadvantage of using this method is the requirement of the patient being mechanically ventilated, sedated and paralyzed.
- Arm stop-flow measurements (Pmsfarm). Alternatively, Pmsf can be estimated by measuring intravascular pressure (e.g., using an indwelling radial arterial catheter) after rapid insufflation (<0.3 s) of a proximal arm cuff well above systolic blood pressure (20). About 30 seconds after the arterial flow to the extremity has stopped, the pressure within the arterial and venous side has equilibrated. This allows a more “direct” intravascular measurement of Pmsf: the Pmsfarm. Hence, without the need for sedation and mechanical ventilation, this method might be more feasible for assessing Pmsf.
- Calculation of an analogue of Pmsf (Pmsfa). Finally, an estimation of Pmsf can also be derived mathematically. In the 1990’s, Parkin described an algorithm that incorporates CVP, MAP and CO for the calculation of an analogue of Pmsf: Pmsfa = a(CVP) + b(MAP) + c(CO), where a and b are dimensionless variables based on the assumption that the ratio of venous to arterial compliance is about 24:1 (i.e., a=0.96, b=0.04). The variable (c) reflects the arteriovenous resistance, and its exact value is determined by demographic factors such as age, weight and height (21). Using this algorithm in a closed-loop control of fluid replacement in critically ill patients on continuous haemodiafiltration, Parkin was able to maintain hemodynamic stability for an extended period (>600 hours) despite large fluid shifts (22).
The algorithm was implemented in the NavigatorTM hemodynamic monitor for “continuous” bed-side monitoring. Unfortunately, this device is no longer commercially available. Still, despite the fact that Pmsfa requires the measurement of other hemodynamic variables (CO, CVP and MAP, which will be available in case of more complex hemodynamic disturbances), its potential bed-side monitoring ability may favor its use in future clinical practice.
There is only one method-comparison study that has evaluated the agreement between the three methods for assessing Pmsf in humans with an intact circulation (20). In that study, conducted in 11 mechanically ventilated ICU patients after cardiac surgery, fluid (500 mL colloid) was administered, and positional changes were performed in a standardized 60-minute study period following initial hemodynamic stabilization upon ICU arrival. Following these interventions, changes in Pmsfi were concordant with Pmsfarm and with Pmsfa, suggesting that changes in circulatory volume can be tracked interchangeably well by all methods. With respect to the absolute values of Pmsf, there was a non-significant bias between Pmsfi and Pmsfarm (−1.0±3.1 mmHg), and comparable mean values (20.9±5.6 vs. 19.8±5.7 mmHg, respectively). Pmsfa however, showed some difference in absolute value (mean of 14.9±4.0 mmHg), and a significant bias of −6.0±3.1 mmHg in comparison with Pmsfi. Interestingly though, the difference with Pmsfa was strictly linear, and a calibration factor of 1.42 appeared to reduce this bias to zero. This calibration factor, however, was never validated in subsequent studies. It is important to note that the validity of Pmsfi, which was set as reference Pmsf estimate in this study, was questioned recently as it was suggested that rather high estimates of absolute Pmsf values were found (19,20). Moreover, as demonstrated experimentally after right atrial occlusion experiments in pigs with different volume states, the estimated value of Pmsf depends on the exact individual volume status (23). The latter was confirmed in a similar experimental study for Pmsfarm and Pmsfa (24), and the authors suggested that none of the indirectly derived estimates of absolute Pmsf values can be clinically used. The main reason for this may be that a relatively small error in absolute estimate of Pmsf, may have a substantial influence on variables such as PVR, which is usually low and does not exceed 5 mmHg. Hence, inconsistent Pmsf values may inappropriately guide hemodynamic management (18). Importantly though, all three indirect Pmsf estimates tracked changes after interventions well with a low bias, especially Pmsfa.
The clinical use of Pmsf, or “what can Pmsf tell us?”
Despite the vital contribution of the venous compartment in maintaining hemodynamic stability, there have been no recent studies that have investigated the impact of targeting specific Pmsf values (or any of its related variables) on other relevant hemodynamic variables, nor on outcome. Therefore, its current role may still be seen as academical. The question whether hemodynamic management guided by Pmsf and its related variables impacts outcome measures (e.g., the incidence of organ-injury, composite endpoints of postoperative complications, etc.), may therefore be a relevant goal for future prospective research. Importantly though, Pmsf—and its related variables such as the PVR—can help understanding cardiovascular physiology—and yield a further characterization of the effects of therapeutic interventions, as exemplified below.
Pmsf and the effects of fluids
Pmsf may help in elucidating if—and why—an individual patient may or may not respond to fluid administration.
Traditionally, CVP was regarded as a preload variable and was used for titration of fluids aimed to “optimize” or maximize cardiac output. Knowledge of Pmsf, however, may reveal whether a high CVP is secondary to either a poor cardiac function (i.e., the heart is unable to effectively lower RAP—and thus CVP—in response to increased preload) or secondary to a high(er) Pmsf—suggestive of a high venous vascular filling and/or tone.
In a study in 39 post-surgical ICU patients (25), fluid challenges (n=101) were performed and patients were divided into responders or non-responders to fluid administration, based on an increase in stroke volume or CO of ≥10%. As may be expected, Pmsfa increased in both responders (n=43) and non-responders after fluid administration. In responders though, PVR also increased—while in non-responders, CVP increased along with Pmsfa, so that the gradient between the two (or the driving pressure for venous return) remained unchanged. This finding was confirmed later in a different setting in patients undergoing liver surgery (26). In another study in 61 post-cardiac surgery ICU patients, a total of 107 fluid boluses were administered and responders and non-responders were defined similarly (27). The differential effects of fluids on Pmsfa and PVR between responders and non-responders were again similar. In the latter two studies, the authors additionally calculated heart efficiency (Eh), which represents the ratio between the change in PVR and the change in Pmsfa, and is calculated as: (Pmsfa – CVP)/Pmsfa. In both studies Eh was higher in volume-responders, but interestingly, in the ICU patients in whom a second fluid bolus was administered, Eh correlated well with the degree of increase in CO. Hence, Eh reveals the ability of the heart to generate CO from the amount of VR that was offered.
It may be clear from the information above, that measuring CVP—either alone or in combination with CO—provides insufficient information for the characterization of the effects of fluid administration on venous return and CO.
Pmsf and the effects of vasopressors
Several very recent studies have helped in a better understanding of the (often complex) effects of vasopressors on the systemic circulation. Vasopressors, e.g., phenylephrine and norepinephrine, are frequently used to treat arterial hypotension, for example in patients suffering from septic shock or in patients in whom general and/or neuraxial anesthesia is performed. Since these drugs primarily act on α-adrenergic receptors and increase arterial tone and thus systemic vascular resistance (subsequently increasing MAP and thus cardiac afterload), in particular for phenylephrine, it is often presumed to have a negative effect on CO—or at best no CO effect at all (28). Yet, in patients undergoing abdominal surgery, it was found recently that the effect of phenylephrine infusion on the CO was dependent on their volume state, and that in “fluid responsive” patients, CO increased following phenylephrine infusion, confirming previous observations in animal studies (29,30). In these patients, both Pmsfa, CVP and CO increased following phenylephrine administration, suggesting an increase in venous return due to the constrictive effects of phenylephrine on the venous capacitance vessels, increasing Pmsf by recruiting unstressed blood volume into stressed blood volume. It is worth reminding that Pmsf is a function of both intravascular filling and the actual systemic vascular compliance, which can be influenced by vasoactive drugs. In volume-responders, the heart was able to handle the increase in VR since CO increased by virtue of an increase in PVR. Of note, this effect of phenylephrine only accounts for fluid-responsive patients in whom increasing VR results in an improved position of the heart on the Frank-Starling curve.
In a subsequent study in pigs, phenylephrine was administered continuously after the initial bolus, and a similar increase in CO was found, together with increasing values of PVR. This again demonstrates that the VR increases secondary to constriction-effects of phenylephrine on capacitance vessels (31). While not yet confirmed, it is likely that norepinephrine might affect preload in a similar way (32), although probably to a lesser extent owing to a much shorter biological half-life of 2–6 minutes (33), as compared to phenylephrine. This difference in biological half-life may be associated with lower concentrations of norepinephrine in the capacitance vessels than with phenylephrine, and subsequently with less pronounced effects on VR. Future research should substantiate this assumption. In a very different setting—i.e., in patients after cardiac surgery (n=16)—it was shown that norepinephrine administration could either increase (n=6) or decrease CO (n=10) (34). In patients in whom CO decreased, the increase in VR following an increase of Pmsf could probably not be handled by the heart, emphasizing the importance of considering both venous return and cardiac function when interpreting Pmsf and its related variables, which is also reflected in the position of the heart on its (individual) Frank-Starling curve.
These studies elegantly show that Pmsf and its related variables can help in characterizing the effects of vasopressors, and may in the future help to optimize not only the blood pressure itself, but also to assess vascular capacitance, and modify it accordingly by titrating fluids and appropriate vasopressors. On the contrary, the effect(s) of directly vasodilating agents on Pmsf has—at least to our knowledge—not yet been the subject of investigation, while an in-depth (physiological) characterization of such drugs might be very worthwhile for a further understanding of their hemodynamic impact.
Other instances in which measurement of Pmsf might be useful
There are many instances in which Pmsf may aid in, at least, characterizing the hemodynamic situation of a patient, and may play a future role in hemodynamic management. E.g., in veno-arterial extracorporeal membrane oxygenation it may help in determining the appropriate amount of fluids and vasopressors (35), it may help in characterizing hemodynamic physiology during pregnancy, and during obstetric pathology (pre-eclampsia), which is of high relevance to the attending physician at the ED or ICU (36). Furthermore, it may elucidate the complex hemodynamic effects of sedative drugs frequently used in the ICU, such as midazolam (37) or propofol (38).
Conclusions
The venous system plays a crucial role in maintaining cardiac output by preservation of venous return of blood to the heart. The Pmsf is the upstream pressure for venous return, and can nowadays be estimated indirectly by three methods. Even though absolute estimates of the Pmsf should be used with caution, changes in Pmsf estimates can reliably reflect the effects of hemodynamic interventions such as the administration of fluids and vasoactive agents. Hence, the Pmsf provides—together with an assessment of cardiac function—an integrative picture of the hemodynamic performance, and may in the future help to guide hemodynamic management in acutely ill patients.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jeccm.2020.03.04). TWLS received research grants and honoraria from Edwards Lifesciences (Irvine, CA, USA) and Masimo Inc. (Irvine, CA, USA) for consulting and lecturing and from Pulsion Medical Systems SE (Feldkirchen, Germany) for lecturing. JJV and AFK report no conflicts of interest.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Rothe CF. Reflex control of veins and vascular capacitance. Physiol Rev 1983;63:1281-342. [Crossref] [PubMed]
- Guyton AC. Regulation of cardiac output. Anesthesiology 1968;29:314-26. [Crossref] [PubMed]
- Guyton AC. Determination of cardiac output by equating venous return curves with cardiac response curves. Physiol Rev 1955;35:123-9. [Crossref] [PubMed]
- Frank O. On the dynamics of cardiac muscle. J Biol 1895;32:370-447.
- Patterson SW, Piper H, Starling EH. The regulation of the ventricles. J Physiol 1914;48:465-513. [Crossref] [PubMed]
- Moller PW, Winkler B, Hurni S, et al. Right atrial pressure and venous return during cardiopulmonary bypass. Am J Physiol Heart Circ Physiol 2017;313:H408-20. [Crossref] [PubMed]
- Henderson WR, Griesdale DE, Walley KR, et al. Clinical review: Guyton--the role of mean circulatory filling pressure and right atrial pressure in controlling cardiac output. Crit Care. 2010;14:243. [Crossref] [PubMed]
- Magder S. Bench-to-bedside review: An approach to hemodynamic monitoring - Guyton at the bedside. Crit Care 2012;16:236. [Crossref] [PubMed]
- Magder S, De Varennes B. Clinical death and the measurement of stressed vascular volume. Crit Care Med 1998;26:1061-4. [Crossref] [PubMed]
- Guyton AC, Lindsey AW, Kaufmann BN. Effect of mean circulatory filling pressure and other peripheral circulatory factors on cardiac output. Am J Physiol 1955;180:463-8. [Crossref] [PubMed]
- Dellinger RP, Levy MM, Rhodes A, et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med 2013;39:165-228. [Crossref] [PubMed]
- Levy MN. The cardiac and vascular factors that determine systemic blood flow. Circ Res 1979;44:739-47. [Crossref] [PubMed]
- Mitzner W. The classical Guyton view that mean systemic pressure, right atrial pressure, and venous resistance govern venous return is/is not correct. J Appl Physiol 2006;101:1529-30. [PubMed]
- Permutt S. The classical Guyton view that mean systemic pressure, right atrial pressure, and venous resistance govern venous return is/is not correct. J Appl Physiol 2006;101:1528. [Crossref] [PubMed]
- Magder S. The classical Guyton view that mean systemic pressure, right atrial pressure, and venous resistance govern venous return is/is not correct. J Appl Physiol 2006;101:1533. [Crossref] [PubMed]
- Wang LY, Wang WK. The classical Guyton view that mean systemic pressure, right atrial pressure, and venous resistance govern venous return is/is not correct. J Appl Physiol 2006;101:1528-9. [Crossref] [PubMed]
- Brengelmann GL. The classical Guyton view that mean systemic pressure, right atrial pressure, and venous resistance govern venous return is/is not correct. J Appl Physiol 2006;101:1532. [Crossref] [PubMed]
- Schipke JD, Heusch G, Sanii AP, et al. Static filling pressure in patients during induced ventricular fibrillation. Am J Physiol Heart Circ Physiol 2003;285:H2510-5. [Crossref] [PubMed]
- Maas JJ, Geerts BF, van den Berg PC, et al. Assessment of venous return curve and mean systemic filling pressure in postoperative cardiac surgery patients. Crit Care Med 2009;37:912-8. [Crossref] [PubMed]
- Maas JJ, Pinsky MR, Geerts BF, et al. Estimation of mean systemic filling pressure in postoperative cardiac surgery patients with three methods. Intensive Care Med 2012;38:1452-60. [Crossref] [PubMed]
- Langewouters GJ, Wesseling KH, Goedhard WJ. The pressure dependent dynamic elasticity of 35 thoracic and 16 abdominal human aortas in vitro described by a five component model. J Biomech 1985;18:613-20. [Crossref] [PubMed]
- Parkin G, Wright C, Bellomo R, et al. Use of a mean systemic filling pressure analogue during the closed-loop control of fluid replacement in continuous hemodiafiltration. J Crit Care 1994;9:124-33. [Crossref] [PubMed]
- Berger D, Moller PW, Weber A, et al. Effect of PEEP, blood volume, and inspiratory hold maneuvers on venous return. Am J Physiol Heart Circ Physiol 2016;311:H794-806. [Crossref] [PubMed]
- Werner-Moller P, Sondergaard S, Jakob SM, et al. Effect of volume status on the estimation of mean systemic filling pressure. J Appl Physiol 1985;2019:1503-13. [PubMed]
- Cecconi M, Aya HD, Geisen M, et al. Changes in the mean systemic filling pressure during a fluid challenge in postsurgical intensive care patients. Intensive Care Med 2013;39:1299-305. [Crossref] [PubMed]
- Vos JJ, Kalmar AF, Hendriks HG, et al. The effect of fluid resuscitation on the effective circulating volume in patients undergoing liver surgery: a post-hoc analysis of a randomized controlled trial. J Clin Monit Comput 2018;32:73-80. [Crossref] [PubMed]
- Gupta K, Sondergaard S, Parkin G, et al. Applying mean systemic filling pressure to assess the response to fluid boluses in cardiac post-surgical patients. Intensive Care Med 2015;41:265-72. [Crossref] [PubMed]
- Zimmerman J, Cahalan M. Vasopressors and inotropes. In: Hemmings HC, Egan TD, editors. Pharmacology and physiology for anaesthesia—foundation and clinical application. Philadelphia: Elsevier Saunders; 2013. p. 390-404.
- Kalmar AF, Allaert S, Pletinckx P, et al. Phenylephrine increases cardiac output by raising cardiac preload in patients with anesthesia induced hypotension. J Clin Monit Comput 2018;32:969-76. [Crossref] [PubMed]
- Cannesson M, Jian Z, Chen G, et al. Effects of phenylephrine on cardiac output and venous return depend on the position of the heart on the Frank-Starling relationship. J Appl Physiol 1985;2012:281-9. [PubMed]
- Wodack KH, Graessler MF, Nishimoto SA, et al. Assessment of central hemodynamic effects of phenylephrine: an animal experiment. J Clin Monit Comput 2019;33:377-84. [Crossref] [PubMed]
- Monnet X, Jabot J, Maizel J, et al. Norepinephrine increases cardiac preload and reduces preload dependency assessed by passive leg raising in septic shock patients. Crit Care Med 2011;39:689-94. [Crossref] [PubMed]
- Beloeil H, Mazoit JX, Benhamou D, et al. Norepinephrine kinetics and dynamics in septic shock and trauma patients. Br J Anaesth 2005;95:782-8. [Crossref] [PubMed]
- Maas JJ, Pinsky MR, de Wilde RB, et al. Cardiac output response to norepinephrine in postoperative cardiac surgery patients: interpretation with venous return and cardiac function curves. Crit Care Med 2013;41:143-50. [Crossref] [PubMed]
- Moller PW, Hana A, Heinisch PP, et al. The Effects of Vasoconstriction And Volume Expansion on Veno-Arterial ECMO Flow. Shock 2019;51:650-8. [Crossref] [PubMed]
- Crozier TM, Wallace EM, Parkin WG. Haemodynamic assessment in pregnancy and pre-eclampsia: A Guytonian approach. Pregnancy Hypertens 2015;5:177-81. [Crossref] [PubMed]
- Chen J, Yu T, Longhini F, et al. Midazolam increases preload dependency during endotoxic shock in rabbits by affecting venous vascular tone. Ann Intensive Care 2018;8:59,018-0403-9.
- de Wit F, van Vliet AL, de Wilde RB, et al. The effect of propofol on haemodynamics: cardiac output, venous return, mean systemic filling pressure, and vascular resistances. Br J Anaesth 2016;116:784-9. [Crossref] [PubMed]
Cite this article as: Vos JJ, Kalmar AF, Scheeren TWL. Bedside assessment and clinical utility of mean systemic filling pressure in acute care. J Emerg Crit Care Med 2020;4:25.


