
Left ventricular global longitudinal systolic function predicts mortality in sepsis independent to the shock index
Introduction
Although sepsis is a relatively infrequent clinical syndrome (1), sepsis (2) remains a challenging issue world-wide (3), associated with high mortality rate (4,5), and elevated costs of medical management. In sepsis, fatal events may be accounted for by refractory cardiovascular failure in as many as 1 patient in 3, and myocardial injury can be found in approximately 1 patient in 2 in post-mortem necropsy data (6). In septic patients, left ventricular (LV), ejection fraction (EF) and the shock-index (SI), a metric of hemodynamic instability as defined by the ration between heart rate (HR) and systolic blood pressure (BP), are often assessed for risk stratification. However, LV EF does not represent LV myocardial contractility in intact human hearts due to its high dependency from LV loading conditions and geometry (7-10), and the relationships of LV EF to outcome in sepsis and septic shock is inconsistent literature (11-20). Moreover, the prognostic importance of the SI in sepsis has been not consistent in literature (21-29).
LV global peak systolic strain (GLS) is a measure of longitudinal myocardial contractility (30), found to be related to prognosis in sepsis in particular in short follow-ups (19,20). We hypothesized that GLS may contribute to risk stratification in sepsis or septic shock independently of SI, because in sepsis, impaired myocardial systolic function may not be a simple manifestation of unstable coronary perfusion secondary to hemodynamic instability (31-34).
Methods
In a time window going from October 2012 to June 2015, consecutive patients not in dialysis, admitted to a High-Dependency-Observation Unit of the Emergency Department, with sepsis or septic shock by standard definition (2), were evaluated (n=155) for hemodynamic instability and assessment of LV structure and function (21% excluded from the current analyses due to poor quality of imaging). Outcome was evaluated in a prospective study design. Events comprised death by any cause, assessed in-hospital by medical records, or by telephone and chart reviews after discharge. Re-hospitalizations were not accounted as events. Mortality rate was censored at day-7 (16%) as well as at day-28 (30%) from hospitalization. Time to event was defined as the difference between date of event and date of hospitalization, whereas for survivors, the observation time was set at 7 and 28 days by definition. No patient was lost at follow-up.
As reported previously (19,35), sepsis was defined by the coexistence of two or more of the following criteria: temperature >38 or <35 °C, HR >90 beats/minute, respiratory rate >20 breaths/minute or arterial partial pressure of carbon dioxide <32 mmHg (<4.3 kPa), white cell count >12,000 cells/mm3, or <4,000 cells/mm3, or presence of immature forms >10%, with further evaluation of arterial blood lactate concentration. Sepsis-related Organ Failure Assessment (SOFA) score was used to assess sepsis severity (36), and to increase the sepsis-related inflammatory syndrome as recommended more recently (37-39). Shock was defined as systolic BP below 90 mmHg, or abrupt systolic BP drop of at least 40 mmHg from initial values, unresponsive to intravenous fluids and persisting for more than 20 minutes. Coronary heart disease (CHD) was defined based on medical history, charts review, ECG and echocardiographic findings as specified previously (19,40). History of chronic obstructive pulmonary disease (COPD) was defined based on medical charts review and therapy. History of chronic kidney disease (CKD) was defined by laboratory findings (serum creatinine above 1.2 mg/dL or 106.1 mmol/L) within a year from the hospitalization. History of cancer was assessed based on medical chart review. The study protocol was approved by the “Toscana – Area vasta - Centro” inter-institutional ethic committee (registration number OSS.13.031).
Echocardiography was performed within 24 h from the admission by a standardized protocol (41-43) and settings (iE33, Philips Medical System, Andover, MA, USA) allowing acquisitions of digital loops of at least 2 cardiac cycles with frames per second (fps) of 45 or greater (mean value observed 57±5 fps). LV EF was assessed by ventricular planimetry (44). LV stroke volume was computed as the difference between end-diastolic and end-systolic volumes; stroke index was computed as stroke volume/body surface area; cardiac index was computed as stroke index X HR and converted to L/min/m2. Systemic vascular resistance index was computed as mean BP*80/cardiac index (divided by 1,000, kdyne*s/cm5*m2). The early wave of the LV filling (E wave) was sampled by pulsed Doppler at the tips of the mitral valve. Speckle-tracking analysis was performed off-line days or weeks after the admission (Philips QLAB Advanced Quantification Software version 8.1) in random sequence, and by experienced personnel (VP as final arbiter) blind to clinical data and outcome, by a methodology applied widely (19,43,45). Briefly, myocardial deformation was analyzed from two-dimensional gray-scale loops of the apical views of the left ventricle, by automatic tracking of myocardial speckles, granted a manual selection of the region of interest. GLS was calculated averaging the negative peak of longitudinal strain from ventricular segments from the apical 4-chamber, 2-chamber views and apical-long axis. Subnormal LV GLS was defined by values >−15% (46); very depressed GLS was empirically defined based on GLS >−10%. In-house test-retest reproducibility analyses for assessment of LV EF and GLS have been tested and reported previously (19).
Statistical analysis
Statistical analyses were conducted using IBM SPSS® software package (version 22). Data in table are mean values ± 1 standard deviation of the mean, or counts and percent. Cases with missing data (<1% of the study sample) were excluded analysis by analysis. Patients were stratified in three groups according to SI values <0.7 or ≥0.7 but <1, or ≥1 bpm/mmHg (26). For continuous variables, the null hypothesis was tested using the Student’s t test for independent groups, employing log-transformed variables in the case of excessively skewed distribution; Fisher’s exact test was used to compare counts in cross-tables (47). Receiver operating characteristic (ROC) curve was employed to evaluate the performance of classification of events, and the area under the curve of positive rate (sensitivity) versus false positive rate (1-specificity) for values of single variables was assumed as indicator of accuracy of variable-specific performance of classification. According to current recommendations (48,49), based on day-28 mortality rate, Cox’s proportional hazard function was employed to explore whether SI and GLS predicted time-dependent mortality accounting for age, SI, and history of CHD, CKD, COPD, SOFA ≥2, diabetes and of cancer, using a stepwise selection method with entry testing based on the significance of the score statistic (for P<0.05), and removal testing based on the probability of a likelihood-ratio statistic based on the maximum partial likelihood estimates (for P>0.1) (50). Time-varying effects of SI, GLS and EF on prognosis were tested by time-varying Cox’ proportional hazard models, generating separate interaction terms of time with SI, GLS and EF, added to the models along with primary variables. Correlates of prognosis at day-7 follow-up were also provided as pathophysiologically relevant in severe sepsis. Two-tailed P<0.05 was used to reject null hypotheses.
Results
Among the 123 patients, not mechanically ventilated, who comprised the study sample (21% excluded due to poor quality of echocardiographic imaging), 48 (39%) showed a SI below 0.7 bpm/mmHg, and 25 (20%) a SI ≥1 bpm/mmHg; hence, 50 participants had a SI comprised between 0.7 and 0.99 bpm/mmHg (Table 1, missing laboratory data <1% of the sample). In univariate analyses, age did not differ among groups of patients stratified based on SI; female gender tended to be more frequent among the patients with higher SI values; prevalence of diabetes and proportion of those with history of CKD did not differ between groups; history of CHD showed a not statistically significant trend toward lower prevalence with higher SI. History of COPD was lowest with SI comprised between 0.7 and 0.99 bpm/mmHg while history of cancer was lowest in the group of patients with SI <0.7 bpm/mmHg. BMI was comparable among the three groups. Almost by definition, mean BP was lower and HR was higher in patients with higher SI. Differences in blood lactate concentration showed a trend toward higher values with SI ≥1 vs. <0.7 while differences in troponin I did not reach the statistical significance across SI strata. Proportion of subjects with SOFA ≥2 did not differ significantly across the SI groups.
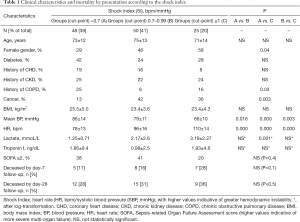
Full table
As reported in Table 2 (no missing data), LV end-diastolic volume index and E wave peak velocity were comparable among groups by SI; stroke index was slightly lower with SI ≥1 vs. SI <0.7 (P<0.01), whereas cardiac index was comparable among groups according to SI; systemic vascular resistance index tended to be lower with higher SI without reaching the statistical significance. Percent change in inferior vena cava with a single rapid deep breath did not differ among groups (35% vs. 33% vs. 30% according to SI groups as in Table 2, P>0.1).

Full table
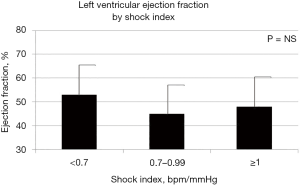
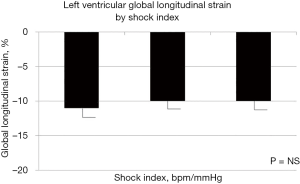
In univariate analysis, LV EF (Figure 1) and LV GSL (Figure 2) did not differ across groups of patients according to SI. Mean duration of follow-up was of 23 days (range, 1–28 days). Mortality by day-7 (n=19, 16%) showed a trend toward higher incidence with higher SI without reaching the statistical significance in univariate analyses, whereas at day-28 follow-up, the mortality (n=37, 30%) was comparable among the SI strata at admission.
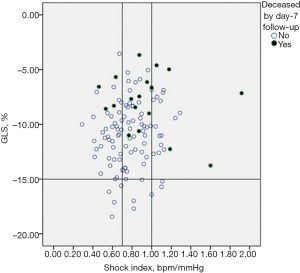
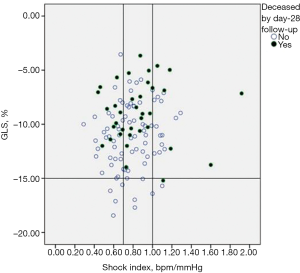
SI did not correlate with GLS (r=0.14, P=0.13) as well as with EF (r=−0.17, P=0.07). Figure 3 showed that GLS may vary widely per unit of SI; moreover, deaths by day-7 (n=18, 15%) tended to cluster according GLS >−15% more than according to SI, as demonstrated by the fact that events were recorded over a large interval of SI values, but almost never in the case of GLS <−15%, even for SI ≥1. Mortality data by day-28 follow-up (n=36, 30%) were consistent with those reported by day-7 follow-up (Figure 4).
By ROC curves, less negative GLS showed a statistically significant accuracy of the performance of classification of events by day 28 follow-up (area under the curve 0.68, 95% confidence interval 0.58–0.79, P=0.001), followed by lower LV EF (area under the curve 0.63; 95% CI, 0.51–0.75; P=0.03), whereas increasing values of SI (area under the curve 0.57; 95% CI, 0.46–0.69; P=0.1) did not appear a significant and accurate predictor events. The interaction of GLS with SI (GLS*SI, %*bpm/mmHg) showed an accuracy of the performance of classification of events by day 28 follow-up not superior to GLS alone (area under the curve 0.60; 95% CI, 0.48–0.71; P=0.09). Data using day-7 mortality were consistent (data not shown). In multivariable analyses, mortality rate at day-28 follow-ups was predicted by GLS and not by the SI, independent of covariates and cofounders (Table 3) while survival analysis considering day-7 follow-up showed a consistent trend despite a smaller number of events recorded by the follow-up. Results did not change when interaction terms of time and GLS, EF as well as SI were generated and added to the Cox’s proportional hazard models (data not shown).

Full table
Discussion
In patients with sepsis or septic shock, we provided novel information, as we showed that LV GLS is prognostically relevant at day-28 follow-up beyond the SI independently of a number of clinical confounders including history of cancer and of ischemic heart disease, and independently of SOFA score ≥2. Furthermore, we showed that in sepsis or septic shock, LV systolic function, either assessed by EF or GLS, cannot be inferred by assessing SI, which is consistent with the notion that LV dysfunction in sepsis is not the simple result of myocardial ischemia (31,32). Therefore, assessment of LV GLS may add useful short-term prognostic information in the early phase of the risk stratification in sepsis/septic shock independent to the assessment of SI, and may be useful for triaging patients in the Emergency Department. Nevertheless, further studies are required to assessment whether LV GLS, and its change over time, may guide or influence treatment options, and contribute to change outcomes in sepsis as to date vasopressors/inotropes have been unable to change prognosis in septic shock (28,51-53).
SI has been indicated as useful tool for triaging patients with potentially critical clinical conditions requiring intensive surveillance (21). Current guidelines suggest the use of SOFA to increase specificity of sepsis-related inflammation and prognosis (37). Adding a SOFA score equal or greater than 2 to the survival models did not impact the correlation of GLS with prognosis. SI and SOFA did not show strong inter-correlation in our study. In a previous study in a large cohort of patients with sepsis, SI ≥1 showed high specificity (80%) and negative predictive value (88%) with regard to outcome in the mid-term (28-day follow-up mortality) (27), but also showed relatively poor sensitivity (37%) and positive predictive value (23%); SI ≥0.7 had a negative predictive value as high as 95%, suggesting that such a simple, bed-side no-cost index could be used in sepsis for early patient’s characterization. Nevertheless, the most recent Protocol-Based Care for Early Septic Shock (ProCESS) study failed to reach the specific goal to change prognosis in group of patients with severe sepsis/septic shock specifically characterized by their hemodynamic conditions as expressed by the SI (28). Our study added new information by showing that the early assessment LV GLS predict all-cause death while SI was not prognostically relevant in patients with sepsis, in particular in the very short term. Interesting, LV end-diastolic volume, the peak velocity of the mitral E wave, cardiac index and systemic vascular resistance index did not differ significantly among SI-strata, suggesting that central hemodynamics was not the main player in determining the ratio between HR and systolic BP.
Although in experimental models in sepsis, cardiac output has being found to be increased and myocardial perfusion decreased (34), an increase in cardiac output may contribute to an increase in LV external myocardial work and oxygen consumption (34). Because SI tend to be higher with higher HR and with lower systolic BP, higher LV external work cannot be inferred by higher SI. In fact, in our study, cardiac index was not higher with higher SI while mean BP was lower with higher SI. Moreover, LV external work is proportional to stroke volume and to mean aortic pressure (54-56), as well as to systolic tension (or stress) applied to the left ventricle (57). Of note, systolic BP is one of the determinants of systolic wall stress and of myocardial blood flow (58). In our study, stroke index was only marginally lower with higher SI, cardiac index was comparable among SI-based groups, and mean BP was lower with higher SI. Those results may explain at least in part the reason why SI did not correlate with GLS or with EF, along with the fact that LV systolic function is not the simple manifestation of myocardial under-perfusion (32-34). Myocardial injury in sepsis is associated with myocardial edema, inflammatory cell infiltration, cytokines-mediated damage and micro-vascular disease, and is more likely associated with impaired longitudinal LV myocardial contractility (6,59-62) rather than SI. A weak relationship between SI and GLS while the latter emerges as an important predictor of events in sepsis and septic shock, may help explaining the relatively low power of SI as prognostic factor in sepsis.
The present study has a number of limitations. Initial definition of sepsis was based on criteria (2), which have been considered not sufficiently specific as they are excessively oriented to account for the inflammatory response. More recently, new criteria for definition of sepsis have focused on more infection-related life-threatening inflammation (37). Retrospective analyses of current databases in the research field on sepsis suggest that the new definition including SOFA (36) is able to increase the specificity of sepsis-related inflammatory syndrome and predict greater mortality (38,39). At least in part, we accounted for the issue of the impact of sepsis definition on findings by considering a SOFA ≥2 as covariate in the survival analyses. The study focused on patients not mechanically ventilated, which may have contributed to the high feasibility of LV function quantification in our study, and could have characterized patients with less severe sepsis and shock; nevertheless, mortality rate by day-7 and day-28 was in line with expectations from a more general source of patients (27). Moreover, the study focused on data collected at the admission in the High-Dependency Observational Unit, without accounting for their changes over-time and initial scenarios in the emergency ward at first medical contact. History of CHD and of diabetes did not impact the relationship between LV GLS and prognosis, which suggests that values of LV GLS at admission in the Emergency Department are largely determined by sepsis. We did not assess circumferential strain systematically in the study population, which could be used to characterize the disease process involving differently oriented myocardial fibers in the LV wall (63); nevertheless, in our hands, test-re-test variability was higher for circumferential than longitudinal strain, and early impairment of the LV longitudinal function compared to the relatively preserved circumferential function and LV EF in the early phase of the myocardial disease process has been reported previously (63).
In conclusion, in sepsis or septic shock, depressed LV longitudinal systolic performance, and not high SI, may predict all-cause mortality in short follow-ups independently of comorbidities including cancer, CAD, CKD and SOFA ≥2.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study protocol was approved by the “Toscana – Area vasta - Centro” inter-institutional ethic committee (registration number OSS.13.031) and was conducted in accordance with the Helsinki Declaration of 1964 (revised 2008). All patients gave informed consensus to enter the study.
References
- Hall MJ, Williams SN, DeFrances CJ. Inpatient Care for Septicemia or Sepsis: A Challenge for Patients and Hospitals. U S Department of Health and Human Services - National Center for Health Statistics 2011;62:1-7.
- Dellinger RP, Levy MM, Rhodes A, et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med 2013;39:165-228. [Crossref] [PubMed]
- Angus DC, Linde-Zwirble WT, Lidicker J, et al. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 2001;29:1303-10. [Crossref] [PubMed]
- Sakr Y, Elia C, Mascia L, et al. The influence of gender on the epidemiology of and outcome from severe sepsis. Crit Care 2013;17:R50. [Crossref] [PubMed]
- Dombrovskiy VY, Martin AA, Sunderram J, et al. Rapid increase in hospitalization and mortality rates for severe sepsis in the United States: a trend analysis from 1993 to 2003. Crit Care Med 2007;35:1244-50. [Crossref] [PubMed]
- Torgersen C, Moser P, Luckner G, et al. Macroscopic postmortem findings in 235 surgical intensive care patients with sepsis. Anesth Analg 2009;108:1841-7. [Crossref] [PubMed]
- de Simone G, Devereux RB, Roman MJ, et al. Assessment of left ventricular function by the midwall fractional shortening/end-systolic stress relation in human hypertension J Am Coll Cardiol 1994;23:1444-51. [erratum in J Am Coll Cardiol 1994 Sep;24(3):844]. [Crossref] [PubMed]
- Dumesnil JG, Shoucri RM. Effect of the geometry of the left ventricle on the calculation of ejection fraction. Circulation 1982;65:91-8. [Crossref] [PubMed]
- Borow KM, Green LH, Grossman W, et al. Left ventricular end-systolic stress-shortening and stress-length relations in human. Normal values and sensitivity to inotropic state. Am J Cardiol 1982;50:1301-8. [Crossref] [PubMed]
- Russo C, Jin Z, Homma S, et al. Relationship of multidirectional myocardial strain with radial thickening and ejection fraction and impact of left ventricular hypertrophy: a study in a community-based cohort. Echocardiography 2013;30:794-802. [Crossref] [PubMed]
- Parker MM, Shelhamer JH, Bacharach SL, et al. Profound but reversible myocardial depression in patients with septic shock. Ann Intern Med 1984;100:483-490. [Crossref] [PubMed]
- Jardin F, Fourme T, Page B, et al. Persistent preload defect in severe sepsis despite fluid loading: A longitudinal echocardiographic study in patients with septic shock. Chest 1999;116:1354-9. [Crossref] [PubMed]
- Vieillard-Baron A, Caille V, Charron C, et al. Actual incidence of global left ventricular hypokinesia in adult septic shock. Crit Care Med 2008;36:1701-6. [Crossref] [PubMed]
- Bouhemad B, Nicolas-Robin A, Arbelot C, et al. Acute left ventricular dilatation and shock-induced myocardial dysfunction. Crit Care Med 2009;37:441-7. [Crossref] [PubMed]
- Hochstadt A, Meroz Y, Landesberg G. Myocardial dysfunction in severe sepsis and septic shock: more questions than answers? J Cardiothorac Vasc Anesth 2011;25:526-35. [Crossref] [PubMed]
- Becker L, Prado K, Foppa M, et al. Endothelial dysfunction assessed by brachial artery ultrasound in severe sepsis and septic shock. J Crit Care 2012;27:316.e9-14. [Crossref] [PubMed]
- Landesberg G, Gilon D, Meroz Y, et al. Diastolic dysfunction and mortality in severe sepsis and septic shock. Eur Heart J 2012;33:895-903. [Crossref] [PubMed]
- Pulido JN, Afessa B, Masaki M, et al. Clinical spectrum, frequency, and significance of myocardial dysfunction in severe sepsis and septic shock. Mayo Clin Proc 2012;87:620-8. [Crossref] [PubMed]
- Palmieri V, Innocenti F, Guzzo A, et al. Left ventricular systolic longitudinal function as predictor of outcome in patients with sepsis. Circ Cardiovasc Imaging 2015;8:e003865. [PubMed]
- Chang WT, Lee WH, Lee WT, et al. Left ventricular global longitudinal strain is independently associated with mortality in septic shock patients. Intensive Care Med 2015;41:1791-9. [Crossref] [PubMed]
- Rady MY, Smithline HA, Blake H, et al. A comparison of the shock index and conventional vital signs to identify acute, critical illness in the emergency department. Ann Emerg Med 1994;24:685-90. [Crossref] [PubMed]
- Little RA, Kirkman E, Driscoll P, et al. Preventable deaths after injury: why are the traditional 'vital' signs poor indicators of blood loss? J Accid Emerg Med 1995;12:1-14. [Crossref] [PubMed]
- Birkhahn RH, Gaeta TJ, Terry D, et al. Shock index in diagnosing early acute hypovolemia. Am J Emerg Med 2005;23:323-6. [Crossref] [PubMed]
- Zarzaur BL, Croce MA, Fischer PE, et al. New vitals after injury: shock index for the young and age x shock index for the old. J Surg Res 2008;147:229-36. [Crossref] [PubMed]
- Cannon CM, Braxton CC, Kling-Smith M, et al. Utility of the shock index in predicting mortality in traumatically injured patients. J Trauma 2009;67:1426-30. [Crossref] [PubMed]
- Vandromme MJ, Griffin RL, Kerby JD, et al. Identifying risk for massive transfusion in the relatively normotensive patient: utility of the prehospital shock index. J Trauma 2011;70:384-8. [Crossref] [PubMed]
- Berger T, Green J, Horeczko T, et al. Shock index and early recognition of sepsis in the emergency department: pilot study. West J Emerg Med 2013;14:168-74. [Crossref] [PubMed]
- Yealy DM, Kellum JA, Huang DT, et al. A randomized trial of protocol-based care for early septic shock. N Engl J Med 2014;370:1683-93. [Crossref] [PubMed]
- Tseng J, Nugent K. Utility of the shock index in patients with sepsis. Am J Med Sci 2015;349:531-535. [Crossref] [PubMed]
- Burns AT, La Gerche A, D'hooge J, et al. Left ventricular strain and strain rate: characterization of the effect of load in human subjects. Eur J Echocardiogr 2010;11:283-9. [Crossref] [PubMed]
- Cunnion RE, Schaer GL, Parker MM, et al. The coronary circulation in human septic shock. Circulation 1986;73:637-44. [Crossref] [PubMed]
- Zhou M, Wang P, Chaudry IH. Cardiac contractility and structure are not significantly compromised even during the late, hypodynamic stage of sepsis. Shock 1998;9:352-8. [Crossref] [PubMed]
- Solomon MA, Correa R, Alexander HR, et al. Myocardial energy metabolism and morphology in a canine model of sepsis. Am J Physiol 1994;266:H757-68. [PubMed]
- Chagnon F, Bentourkia M, Lecomte R, et al. Endotoxin-induced heart dysfunction in rats: assessment of myocardial perfusion and permeability and the role of fluid resuscitation. Crit Care Med 2006;34:127-33. [Crossref] [PubMed]
- Innocenti F, Bianchi S, Guerrini E, et al. Prognostic scores for early stratification of septic patients admitted to an emergency department-high dependency unit. Eur J Emerg Med 2014;21:254-9. [Crossref] [PubMed]
- Vincent JL, Moreno R, Takala J, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 1996;22:707-10. [Crossref] [PubMed]
- Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016;315:801-10. [Crossref] [PubMed]
- Seymour CW, Liu VX, Iwashyna TJ, et al. Assessment of Clinical Criteria for Sepsis: For the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016;315:762-74. [Crossref] [PubMed]
- Donnelly JP, Safford MM, Shapiro NI, et al. Application of the Third International Consensus Definitions for Sepsis (Sepsis-3) Classification: a retrospective population-based cohort study. Lancet Infect Dis 2017;17:661-70. [Crossref] [PubMed]
- Innocenti F, Palmieri V, Guzzo A, et al. SOFA score and left ventricular systolic function as predictors of short-term outcome in patients with sepsis. Intern Emerg Med 2018;13:51-8. [Crossref] [PubMed]
- Lancellotti P, Tribouilloy C, Hagendorff A, et al. Recommendations for the echocardiographic assessment of native valvular regurgitation: an executive summary from the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging 2013;14:611-44. [Crossref] [PubMed]
- Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the american society of echocardiography and the European association of cardiovascular imaging. J Am Soc Echocardiogr 2015;28:1-39.e14. [Crossref] [PubMed]
- Voigt JU, Pedrizzetti G, Lysyansky P, et al. Definitions for a common standard for 2D speckle tracking echocardiography: consensus document of the EACVI/ASE/Industry Task Force to standardize deformation imaging. Eur Heart J Cardiovasc Imaging 2015;16:1-11. [Crossref] [PubMed]
- Lang RM, Bierig M, Devereux RB, et al. Recommendations for chamber quantification. Eur J Echocardiogr 2006;7:79-108. [Crossref] [PubMed]
- Palmieri V, Russo C, Buonomo A, et al. Novel wall motion score-based method for estimating global left ventricular ejection fraction: validation by real-time 3D echocardiography and global longitudinal strain. Eur J Echocardiogr 2010;11:125-30. [Crossref] [PubMed]
- Yingchoncharoen T, Agarwal S, Popovic ZB, et al. Normal ranges of left ventricular strain: a meta-analysis. J Am Soc Echocardiogr 2013;26:185-91. [Crossref] [PubMed]
- Zhang Z. Univariate description and bivariate statistical inference: the first step delving into data. Ann Transl Med 2016;4:91. [Crossref] [PubMed]
- Peduzzi P, Concato J, Feinstein AR, et al. Importance of events per independent variable in proportional hazards regression analysis. II. Accuracy and precision of regression estimates. J Clin Epidemiol 1995;48:1503-10. [Crossref] [PubMed]
- Long GS. Regression models for categorical and limited dependent variables. Thousand Oaks, CA: Sage Publications, 1997.
- Zhang Z. Semi-parametric regression model for survival data: graphical visualization with R. Ann Transl Med 2016;4:461-9. [Crossref] [PubMed]
- Asfar P, Meziani F, Hamel JF, et al. High versus low blood-pressure target in patients with septic shock. N Engl J Med 2014;370:1583-93. [Crossref] [PubMed]
- Schmittinger CA, Wurzinger B, Deutinger M, et al. How to protect the heart in septic shock: a hypothesis on the pathophysiology and treatment of septic heart failure. Med Hypotheses 2010;74:460-5. [Crossref] [PubMed]
- Dünser MW, Ruokonen E, Pettila V, et al. Association of arterial blood pressure and vasopressor load with septic shock mortality: a post hoc analysis of a multicenter trial. Crit Care 2009;13:R181. [Crossref] [PubMed]
- Starling EH, Evans LL. The respiratory exchanges of the heart in the diabetic animal. J Physiol 1914;49:67. [Crossref] [PubMed]
- Starling EH, Visscher MB. The regulation of energy output of the heart. J Physiol 1927;62:243. [Crossref] [PubMed]
- Bing RJ, Hammond MM, Handelsman JC, et al. The measurement of coronary blood flow, oxygen consumption, and efficiency of the left ventricle in man. Am Heart J 1949;38:1-24. [Crossref] [PubMed]
- Sarnoff SJ, Braunwald E, Welch GH Jr, et al. Hemodynamic determinants of oxygen consumption of the heart with special reference to the tension-time index. Am J Physiol 1958;192:148. [Crossref] [PubMed]
- Weiss MB, Ellis K, Sciacca RR, et al. Myocardial blood flow in congestive and hypertrophic cardiomyopathy: relationship to peak wall stress and mean velocity of circumferential fiber shortening. Circulation 1976;54:484-94. [Crossref] [PubMed]
- Di Bella G, Gaeta M, Pingitore A, et al. Myocardial deformation in acute myocarditis with normal left ventricular wall motion--a cardiac magnetic resonance and 2-dimensional strain echocardiographic study. Circ J 2010;74:1205-13. [Crossref] [PubMed]
- Leitman M, Tyomkin V, Peleg E, et al. Left ventricular function in acute inflammatory peri-myocardial diseases - new insights and long-term follow-up. Cardiovasc Ultrasound 2012;10:42. [Crossref] [PubMed]
- Neil CJ, Nguyen TH, Singh K, et al. Relation of delayed recovery of myocardial function after takotsubo cardiomyopathy to subsequent quality of life. Am J Cardiol 2015;115:1085-9. [Crossref] [PubMed]
- Escher F, Kasner M, Kuhl U, et al. New echocardiographic findings correlate with intramyocardial inflammation in endomyocardial biopsies of patients with acute myocarditis and inflammatory cardiomyopathy. Mediators Inflamm 2013;2013:875420.
- Mizuguchi Y, Oishi Y, Miyoshi H, et al. The functional role of longitudinal, circumferential, and radial myocardial deformation for regulating the early impairment of left ventricular contraction and relaxation in patients with cardiovascular risk factors: a study with two-dimensional strain imaging. J Am Soc Echocardiogr 2008;21:1138-44. [Crossref] [PubMed]
Cite this article as: Palmieri V, Innocenti F, Guzzo A, Donnini C, Stefanone VT, Pini R. Left ventricular global longitudinal systolic function predicts mortality in sepsis independent to the shock index. J Emerg Crit Care Med 2018;2:38.


