
Assessment of the pediatric trauma patient: differences in approach
Epidemiology of pediatric trauma
Despite many advances in the care of children, traumatic injury remains the leading source of death and dismemberment within the United States. In fact, trauma is the leading source of death in patients aged 1 to 44 years (1). Yearly, more than 12,000 school and teenaged children die from unintentional injury and more than 9 million are treated in emergency departments for non-lethal injuries. Males have a higher death rate. The highest death rates include motor vehicle crashes, although mechanisms are age specific. For less than 1 year of age, two-thirds of patients succumbed to suffocation. For ages 1 to 4 years old, drowning was the leading cause of mortality. Falls are also a leading source of morbidity. Death rates are approximately equal between Caucasians and African Americans in the United States (2). Approximately one in four children seeks medical care yearly due to unintentional injury (3). The cost of injury in the United States to these patients approaches $50 billion per annum (4).
There has been improvement in the survival of pediatric trauma with a 50% reduction in mortality from 30 to 15 deaths/100,000 children over the time course from 1984 to 2014. This increased survival likely includes numerous factors, including development of a network of prehospital care as well as regionalization of care into designated trauma centers. One factor which is associated with increased survival of injured children is treatment at a pediatric trauma center (5). However, 17 million children still live more than an hour’s transport from a trauma center. Many children treated for injuries in the United States will be treated at an adult trauma center either definitively or prior to transfer to a pediatric trauma center. It is necessary that any clinician providing trauma care to children is versed in the different patterns of trauma between children and adults. Use of weight/height based equipment and information aids such as the Broselow Pediatric Emergency Tape® (Armstrong Medical Industries, Inc., Lincolnshire, IL, USA) and size-specific resuscitation equipment carts will help in choosing age-specific life-saving maneuvers for injured children, but nothing replaces education and preparation of providers. Unless specified otherwise, aspects of pediatric trauma discussed in this paper are based upon our practice in a pediatric trauma center in a rural community in the eastern United States.
Common patterns of pediatric trauma
Recognition of the common patterns of pediatric trauma can assist in proper assessment and diagnosis simply by knowing the mechanism of injury. A child is more likely than an adult to be a pedestrian struck by a motor vehicle. The recognized constellation of injuries is Waddell’s Triad: head injury, splenic laceration, and femur fracture. Child passengers in vehicles are often unrestrained or improperly restrained. Youth drivers are more likely to be involved in a crash due to inexperience and distraction. Children must be restrained in a car or booster seat appropriate to their size. The National Highway Traffic Safety Administration provides public information to guide clinicians educating parents (6). If a child is restrained by lap belt alone without shoulder strap, especially when not in a booster seat to position the belt over the iliac crest, a lap belt injury can occur. This presents with a transverse ecchymosis over the abdomen and suggests injury to colon, intestine, pancreas, and mesentery.
Participation in youth sports can result in expected patterns of injury, especially when proper safety equipment and wise adult supervision is eschewed. As an example, the most common injuries sustained by girl soccer players were lower extremity fracture, visceral hematoma, and subdural hematoma and the recognized mechanisms were collision, fall, and kick to leg or body (7). All-terrain vehicle (ATV) crashes are more common for youth drivers and are commonly multisystem injuries with extremity injuries and head injury (8). Riding without a helmet and unsafe behaviors, such as a passenger riding behind the ATV driver, predictably lead to worse injuries.
It is important to recognize the patterns of non-accidental trauma (NAT), because if a child returns to a home in which there is child abuse, there is a high likelihood of recurrence and possibly death from future episodes. In 2014, an estimated 700,000 children sustained NAT, and approximately 1,500 died. Understanding the epidemiology of NAT helps to recognize it. The highest rate is in children less than one-year-old, and one third of victims are less than three-year-old. The abuser is known to the child in 75% of cases. One third of NAT to the head is missed on initial examination. Unexplained shock or neurologic deficit in an infant prompts a head computed tomography (CT) and NAT evaluation. Other indicators are delay in presentation, mismatch between history and examination, multiple occurrences of trauma, and injuries suggesting abuse such as bite marks, burns and lacerations in the shape of household objects, and genital injuries. Some institutions will evaluate all children younger than five-years-old injured in the home for the possibility of NAT. Use of an automatic trigger for NAT evaluation helps to eliminate provider bias that may hinder detection of abuse victims. When NAT is suspected, in addition to the ongoing assessment of pediatric trauma, the evaluation should include a skeletal survey, retinal examination, consultation of a pediatric abuse specialist, and referral to the regional Child Protective Services agency. It is an obligation of all health care providers to report child abuse.
Assessment of need for treatment in a facility with pediatric surgical expertise
Injured children often present initially to a facility with limited resources or one that has adult trauma center certification. When specific injuries are identified that will require pediatric specific surgical expertise, arrangements must be made for initial resuscitation and stabilization, communication with a designated pediatric trauma center, and safe timely transfer. An example would be a small child with a serious head injury requiring pediatric intensive care and possibly operative procedure by a pediatric neurosurgeon. The Task Force for Children’s Surgical Care has worked with the American College of Surgeons and the Children’s Hospital Association to develop the Children’s Surgery Verification Quality Improvement Program to delineate the resources necessary in a facility providing surgical care to children (9). Necessary resources include 24-hour availability of a pediatric surgeon, pediatric surgical specialists, pediatric anesthesiology, pediatric intensive care, medical pediatric specialties, and the ability to provide comprehensive care to children of all ages.
Assessment of injured children for need for trauma laparotomy
The experience of non-operative management of splenic injuries in children was first reported by Ein et al. in 1978 at the Hospital for Sick Children (10). This approach spreads to other centers and for other injuries. In 2000, the Committee on Trauma of the American Pediatric Surgical Association (APSA) published their evidence based guidelines for the management of children with hemodynamically stable isolated spleen and liver injuries (11). This clinical practice is now applicable to patients of all ages. The evaluation of such patients has also morphed with the expectation that most solid organ injuries may be managed with resuscitation and observation only.
As with other injuries, the initial priorities focus on maintaining a patent airway, ensuring adequate breathing and continued assessment of circulation. Failure to establish “ABC” signifies a child who is NOT hemodynamically stable and should prompt additional emergent interventions. Signs and symptoms of blunt abdominal trauma include pain, peritoneal irritation, hemodynamic instability, aspartate aminotransferase (AST) greater than 60 U/L, alanine aminotransferase (ALA) greater than 25 U/L, white blood cell count greater than 10×10/L, lactate dehydrogenase (LDH) greater than 330 U/L, amylase greater than 100 U/L and creatinine greater than 110 µmol/L (12). For the hemodynamically stable patient with evidence of blunt abdominal trauma, the mainstay of diagnosis is CT. This is performed with intravenous contrast and at our institution and incorporates As Low as Reasonably Achievable (ALARA) protocol to minimize the dose of ionizing radiation received. Additionally, the use of CT scan allows for grading of each injury which assists in triage, length of stay and restriction of activities (Figure 1).

Focused Abdominal Sonography for Trauma (FAST) is less accurate in detection of blunt organ injury with a decreased sensitivity compared to adult counterparts. Studies have been performed to see if use of FAST would reduce radiation exposure, but since FAST has been demonstrated to have a low sensitivity in children, a negative FAST would likely provide a false sense of security (13). Although we routinely employ FAST, we only consider a positive exam useful for decision making. A recent randomized clinical trial found that the use of FAST in children with blunt torso trauma did not improve clinical care, use of resources, emergency department (ED) length of stay, missed intra-abdominal injuries, or hospital charges when compared to with standard care (14). The use of FAST in children is controversial and a study of 14 Level I Pediatric Trauma Centers showed that the rate of use ranged from 1–94% and rarely affected care (15). One study reported that pediatric trauma providers are more likely to perform FAST when there is a higher suspicion of abdominal injury, and made it less likely that abdominal CT would be performed (16).
At our facility, we employ the Arizona-Texas-Oklahoma-Memphis-Arkansas Consortium (ATOMAC) guideline as a clinical pathway for the treatment of children with suspected abdominal trauma (Figure 2) (17). This guideline focuses on the child’s physiology rather than the grade of injury. If the patient is hemodynamically stable, then admission to a non-intensive care unit (ICU) setting is appropriate as the risk of recurrent bleeding is low. Patients who are clinically bleeding require continued resuscitation. Those who respond may be admitted to a monitored ICU bed for serial assessment. Non-responders will require additional resources including abdominal exploration. We have on rare occasions employed embolization at our institution with success, thereby negating the need for operative intervention (18). However, the finding of contrast extravasation on CT should not automatically prompt embolization and or surgery in the stable child (19).
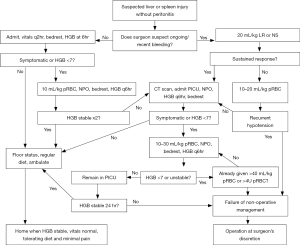
Additional findings on CT are the presence of free intraperitoneal fluid without solid organ injury. This finding suggests hollow viscus or mesentery injury. The initial presentation of children with this injury is often subtle. Those who do not present with obvious signs of peritoneal irritation may be observed (20). Laparoscopy is a valid alternative to exploration in this situation.
For children who undergo trauma laparotomy for abdominal injury and hemodynamic instability, the option for damage control laparotomy and temporary abdominal closure with abdominal negative pressure dressing such as the Vacuum Assisted Closure® (VAC) device (Kinetic Concepts, Inc., San Antonio, TX) should be considered. If the injuries can be temporarily stabilized by controlling surgical bleeding, packing injured solid organs, and stapling areas where there is perforation of hollow viscus, the child can be resuscitated and warmed in the pediatric ICU to avoid the “bloody vicious cycle” of hypothermia, acidosis, and coagulopathy (21).
Assessment of injured children for need for thoracotomy
Thoracic trauma is less common in children than abdominal trauma, but when it is present there is a 35% increase in mortality. In 2015, 13% of the 144,000 injured children in the Pediatric National Trauma Data Bank sustained thoracic trauma, and this injury carried an eight per cent mortality rate, the highest among all body systems (22). Each hemithorax can hold 40% of the circulating blood volume so hemothorax alone can result in life-threatening shock. ATLS assessment directs the rapid identification of the immediate threats of hemothorax, pneumothorax, and pericardial tamponade. As in adults, if these conditions are identified, rapid treatment with chest tube or pericardial drainage must occur without delay for confirmatory radiography. The FAST exam can reveal pericardial blood as well as pneumothorax via windows under the xiphoid and over the upper chest, respectively. In adults, thoracotomy is indicated when a chest tube returns 1,500 mL blood initially or 200 mL/hr, but these conditions must be adjusted for the smaller circulating blood volume in children. For children weighing less than 50 kg, the formula used to determine need for thoracotomy is >15 mL/kg blood initially from the chest tube or >4 mL/kg/hr ×2 hr. It must be remembered that lung and mediastinal injury can occur in children even without rib fracture due to the flexible nature of the pediatric ribcage.
Hemorrhage and venothromboembolism in injured children
Children will have a smaller circulating blood volume due to their smaller body size. When a child is injured, they will suffer a greater degree of shock for the same volume of blood lost. Paradoxically, children will often exhibit the signs of shock later in the course of hemorrhage, due to the ability of the young circulatory system to increase venous and arterial tone to a greater degree than adults and maintain normal vital signs past a larger percentage of blood lost. Once the threshold of maintaining perfusion pressure is surpassed, children will exhibit sudden and catastrophic cardiovascular collapse and difficulty in resuscitation. The volume of circulating blood can be estimated as milliliters of blood per kilograms of body weight for age ranges using these factors: neonates 100 mL/kg; infants 90 mL/kg; school age children 80 mL/kg; and adolescents 70 mL/kg, which is similar to adults. Fluid and blood bolus for children in shock must be weight based: 20 mL/kg for crystalloid fluid boluses and 10 mL/kg for transfusions of blood. Better outcomes will be obtained when children are transfused in a 1:1:1 ratio of blood, plasma, and platelets than when an unbalanced transfusion of blood is given. Establishment of a Massive Transfusion Protocol for injured children at facilities that see pediatric trauma will enable this balanced transfusion to be given rapidly to children in hemorrhagic shock.
Tranexamic acid (TXA), which has been shown to increase survival in injured adults, has few studies demonstrating effect in children. One retrospective study in which TXA was used in the most severely injured children in a combat setting in Afghanistan demonstrated a survival benefit (23). We currently do not use TXA in the management of injured children treated in our facility. If the use of TXA were to be considered for a severely injured child, a potential indication is detection of fibrinolysis on thromboelastography (TEG) because TXA inhibits fibrinolysis. Future studies may support the routine use of TXA for injured children.
Evaluation of coagulopathy in injured children is more difficult than in adults because severely injured children will often exhibit a transient rise in protime (PT) and partial thromboplastin time (PTT) which resolves spontaneously. At a minimum, all injured children should have a blood count checked on presentation and head injured children should have the PT and PTT checked. Unrecognized coagulopathy in head injury can result in increasing mass effect of intracranial hemorrhage and ongoing hemorrhage can lead to increased secondary brain injury of tissue adjacent to the primary brain injury due to hypoperfusion. Viscoelastic hemostatic assays such as TEG allow for rapid and precise determination of specific defects in clotting during initial assessment of an injured child and are increasingly used in the trauma bay.
Venous thromboembolism (VTE), which included deep venous thrombosis (DVT) and pulmonary embolus (PE), can occur after major trauma. The incidence of VTE in children is relatively low, hard to quantify and difficult to predict. However, VTE can still have devastating effects on this patient population. Victims of trauma in this age group with specific risk factors are more susceptible to both DVT and PE. Who and how to treat is still a subject of debate amongst leaders in this field.
The Eastern Association for the Society of Trauma (EAST) and the Pediatric Trauma Society (PTS) recommend prophylaxis in patients older than 15 years old and with an Injury Severity Score (ISS) greater than 25 (24). The incidence of VTE is variable. One study found it to be 6.8/1,000 hospitalizations in pediatric orthopedic trauma versus 2.4/1,000 overall pediatric admissions (25). The National Trauma Database (NTDB) from 2008–2010 found the overall incidence of VTE in patients younger than 21 years-old to be 0.4% (1,655 of 402,329 patients). The incidence was lowest among children less than 12 years old (0.1%) and highest in children older than 16 years old (0.8%) (26).
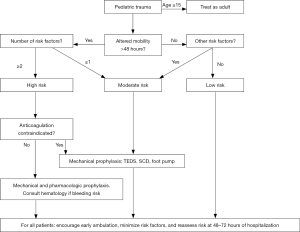
Risk factors for VTE include traumatic brain injury, central venous catheterization, coagulation disorders, NAT, prolonged mechanical ventilation, spinal cord injury, and pelvic and or lower extremity fracture. It is unclear which of these risk factors carries the most significance. There is also little consensus in the pediatric literature regarding for whom and with what form of prophylaxis should be used. Hanson et al. showed a decrease of VTE events using a clinical guideline that included enoxaparin from 2.9% to 0.0% in pediatric trauma patients (27). Our guideline includes both mechanical and chemical prophylaxis and divides patients into different categories dependent on a number of risk factors including central venous catheterization, family history, medications and lower extremity fractures (Figure 3).
Summary
This review addresses the current evaluation and treatment of the injured child in the United States. It is valuable endeavor to focus on trauma in children because injury is the most common reason a child would seek medical care and the most frequent cause of death. Differences between children and adults translate to differences in patterns of injury they suffer as well as the treatment protocols employed in adult and pediatric trauma centers. It is vital that systems of pre hospital care and means of transfer of seriously injured children to pediatric trauma centers be established because there is evidence that pediatric trauma centers will achieve better results in this subgroup. In children, most solid organ injuries will not require operative intervention but centers receiving injured children must have a protocol to guide the evaluation for blunt abdominal injury. Advances in detection and treatment of coagulopathy in injured children may translate to use of new modalities in coming years.
Acknowledgements
The authors would like to acknowledge the tireless efforts of the staff of the Janet Weis Children’s Hospital, as well as the children and families they treat.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Web-based Injury Statistics Query and Reporting System. Injury Prevention and Control. Centers for Disease Control and Prevention, Atlanta, GA, 2015. Available online: www.cdc.gov/injury/wisqars/index.html, Downloaded 30 AUG 2017.
- Borse NN, Gilchrist J, Dellinger AM, et al. CDC Childhood Injury Report, Patterns of Unintentional Injuries among 0-19 Year Olds in the United States, 2000-2006. Centers for Disease Control and Prevention, Atlanta, GA, 2008.
- Danseco ER, Miller TR, Spicer RS. Incidence and costs of 1987-1994 childhood injuries: demographic breakdowns. Pediatrics 2000;105:E27. [Crossref] [PubMed]
- Finklestein EA, Corso PS, Miller TR. Incidence and Economic Burden of Injuries in the United States. Oxford, United Kingdom: Oxford University Press, 2006.
- Sathya C, Alali AS, Wales PW, et al. Mortality among injured children treated at different trauma center types. JAMA Surg 2015;150:874-81. [Crossref] [PubMed]
- Car Seat by Child’s Age and Size. National Highway Traffic Safety Administration, Washington, DC. Available online: https://www.nhtsa.gov/equipment/car-seats-and-booster-seats#age-size-rec, Downloaded 1 OCT 2017.
- Walters BS, Wolf M, Hanson C, et al. Soccer Injuries in Children Requiring Trauma Center Admission. J Emerg Med 2014;46:650-4. [Crossref] [PubMed]
- Su W, Hui T, Shaw K. All-terrain Vehicle Injury Patterns: Are Current Regulations Effective? J Pediatr Surg 2006;41:931-4. [Crossref] [PubMed]
- Task Force for Children's Surgical Care. Optimal resources for children's surgical care in the United States. J Am Coll Surg 2014;218:479-87. [Crossref] [PubMed]
- Ein SH, Shandling B, Simpson JS, et al. Nonoperative management of traumatized spleen in children: how and why. J Pediatr Surg 1978;13:117-9. [Crossref] [PubMed]
- Stylianos S. Evidence-based guidelines for resource utilization in children with isolated spleen and liver injury. J Pediatr Surg 2000;35:164-7. [Crossref] [PubMed]
- de Jong WJ, Stoepker L, Groen H, et al. External Validation of the Blunt Abdominal Trauma in Children (BATiC) score: ruling out significant abdominal trauma in children. J Trauma Acute Care Surg 2014;76:1282-7. [Crossref] [PubMed]
- Scaife ER, Rollins MD, Barnhart DC, et al. The role of focused abdominal sonography for trauma (FAST) in pediatric trauma evaluation. J Pediatr Surg 2013;48:1377-83. [Crossref] [PubMed]
- Holmes JF, Kelley KM, Wooten-Gorges SL, et al. Effect of Abdominal Ultrasound on Clinical Care, Outcomes, and Resource Use Among Children With Blunt Torso Trauma. JAMA 2017;317:2290-6. [Crossref] [PubMed]
- Calder BW, Vogel AM, Zhang J, et al. Focused assessment with sonography for trauma in children after blunt abdominal trauma: A multi-institutional analysis. J Trauma Acute Care Surg 2017;83:218-24. [Crossref] [PubMed]
- Menaker J, Blumburg S, Wisner DH, et al. Use of the focused assessment with sonography for truma (FAST) examination and its impact on abdominal computed tomography use in hemodynamically stable children with blunt torso trauma. J Trauma Acute Care Surg 2014;77:427-32. [Crossref] [PubMed]
- Notrica DM, Eubanks JW III, Tuggle DW, et al. Nonoperative management of blunt liver and spleen injury in children: Evaluation of the ATOMIC guideline using GRADE. J Trauma Acute Care Surg 2015;79:683-93. [Crossref] [PubMed]
- Gross JL, Woll NL, Hanson CA, et al. Embolization for pediatric blunt splenic injury is an alternative to splenectomy when observation fails. J Trauma Acute Care Surg 2013;75:421-5. [Crossref] [PubMed]
- Bansal S, Karrer FM, Hansen K, et al. Contrast blush in pediatric blunt splenic trauma does not warrant the routine use of angiography and embolization. Am J Surg 2015;210:345-50. [Crossref] [PubMed]
- Christiano JG, Tummers M, Kennedy AP. Clinical Significance of isolated intraperitoneal fluid on computed tomography in pediatric blunt abdominal trauma. J Pediatr Surg 2009;44:1242-8. [Crossref] [PubMed]
- Moore EE. Staged laparotomy for the hypothermia, acidosis, and coagulopathy syndrome. Am J Surg 1996;172:405-10. [Crossref] [PubMed]
- Nance ML. editor. National Trauma Data Bank 2015 Pediatric Annual Report. American College of Surgeons, Chicago, IL, 2015
- Eckert MJ, Wertin TM, Tyner SD, et al. Tranexamic Acid Administration to Pediatric Trauma Patients in a Combat Setting: The Pediatric Trauma and Tranexamic Acid Study (PED-TRAX). J Trauma Acute Care Surg 2014;77:852-8. [Crossref] [PubMed]
- Mahajerin A, Perry JK, Hanson SJ, et al. Prophylaxis against venous thromboembolism in pediatric trauma: A practice management guideline from the Eastern Association for the Surgery of Trauma and the Pediatric Trauma Society. J Trauma Acute Care Surg 2017;82:627-36. [Crossref] [PubMed]
- Guzman D, Sabharwal S, Zhao C, et al. Venous thromboembolism among pediatric orthopedic trauma patients: a database analysis. J Pediatr Orthop B 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Van Arendonk KJ, Schneider EB, Haider AH, et al. Venous Thromboembolism After Trauma. When do Children become Adults? JAMA Surg 2013;148:1123-30. [Crossref] [PubMed]
- Hanson SJ, Punzalan RC, Arca MJ, et al. Effectiveness of Clinical Guidelines for deep vein thrombosis in reducing the incidence of venous thromboembolism in critically ill children after trauma. J Trauma Acute Care Surg 2012;72:1292-7. [Crossref] [PubMed]
Cite this article as: Kennedy AP, Scorpio RJ, Coppola CP. Assessment of the pediatric trauma patient: differences in approach. J Emerg Crit Care Med 2018;2:4.


