
Sepsis-induced multi-organ dysfunction syndrome—a mechanistic approach
Introduction
Sepsis is a clinical syndrome invoked by infection with a profound and potentially detrimental impact on general, regional and microvascular hemodynamic, metabolic/endocrine/immune-inflammatory homeostasis, and cell and tissue function. Sepsis is one of the leading causes of death in the intensive care unit (ICU) (1).
Five centuries ago, a renowned Italian philosopher and politician defined sepsis as “a condition of hectic fever that in the beginning is easy to cure but difficult to detect, but in the course of time, not having been detected or treated in the beginning, becomes easy to detect but difficult to cure” (2). This early definition remains remarkably valid today. In fact, it refers to the difficult diagnosis of sepsis which has not been facilitated by the proposed definitions in 1992 (poor specificity, no prognostic value) (3) or 2003 (excessively expanded and complex) (4). At the same time, it points to shock and organ failure as devastating consequences of an evolving septic process.
Recently, an expert task force advocated a new sepsis definition (5). Sepsis was emphasized as a life-threatening organ dysfunction caused by a dysregulated host response to infection. Severity of organ dysfunction can be estimated clinically by calculating the Sequential (formerly “Sepsis-related”) Organ Failure Assessment (SOFA) score (6). An increase of the SOFA score of two points or more indicates significant organ dysfunction and an associated higher mortality risk. Also, a simplified quick SOFA (qSOFA) score was proposed to allow rapid bedside identification of sepsis outside the ICU (5).
The basic approach of sepsis can be summarized as timely recognition, annihilation of infection including source control and early initiation of adequate antimicrobial therapy, volume resuscitation, and vasopressor and/or corticosteroid treatment in case of shock (7). Beyond this, clinicians face the spectre of the multi-organ dysfunction syndrome (MODS). MODS is not a single event but a continuum of processes characterized by serial and incremental physiologic assaults on individual organs. Virtually all organs are involved but damage may vary from hardly detectable or mild to completely irreversible. MODS is a critical condition with high morbidity and mortality and its occurrence substantially increases ICU workload and cost (8).
The pathophysiology of sepsis-induced MODS remains intriguing and has not been completely elucidated. This review aims to concisely line up the different proposed pathways that may explain why a normal host response to infection derails and culminates into devastating organ dysfunction.
Inflammation and endothelial damage
The host response to infection is driven by a complex pro-and anti-inflammatory immune reaction. The initial pro-inflammatory response is prompt and determined by interactions between host factors (e.g., age, co-morbidity, genetic characteristics…) and the pathogen and between pathogen-associated molecular patterns and host cellular pattern recognition receptors located at the cell surface (e.g., Toll-like or C-type lectin receptor) or in the cytoplasm (e.g., retinoic acid inducible gene 1–like or nucleotide-binding oligomerization domain-like receptor) (9). Toll-like receptors (TLR), for instance, recruit adapter proteins to the cell surface. Consequently, cytoplasmic enzymatic processes are initiated that activate various transcription factors which, in turn, produce and release countless inflammatory cytokines and chemokines (10). Exaggerated inflammation induces collateral tissue damage and necrotic cell death which, in turn, propagates the release of damage-associated molecular patterns, aka danger molecules, that perpetuate inflammation (11).
Most prominent instigators of the inflammatory cascade are endotoxin—the lipopolysaccharide component of the cell membrane of Gram-negative bacteria—and comparable substances derived from yeasts, viruses, or Gram-positive microorganisms (12). Intravenous injection of endotoxin in animals and in humans mimics the hemodynamic and metabolic derangements of clinical sepsis whilst producing an intense inflammatory response (13-15). The latter starts with activated monocytes and macrophages which trigger the release of various inflammatory cytokines and chemokines into the extracellular compartment. Cytokines are low-molecular weight and short-lived (glyco)proteins and peptides that act in auto- or paracrine fashion on cells to create a plethora of biological effects (16). Based on immune-elicited outcome, cytokines can be divided in pro- and anti-inflammatory agents. Pro-inflammatory cytokines such as tumor necrosis factor alpha and interleukin (IL)-1 are powerful and synergistic mediators of tissue inflammation, myocardial depression, and endothelial injury (17-21). IL-6 is particularly linked with bacterial sepsis, strongly mediates fever and (sub)acute immune responses, and its plasma concentration is directly associated with severity of sepsis and organ dysfunction (16,22).
Corrupted and dissonant endothelial cell function due to incessant exposure of the endothelium to harmful external and internal inflammatory stimuli is thought to be a strong trigger of MODS. Vascular endothelium is omnipresent and thus universally affected by systemic inflammation. Virtually all endothelial functions, including regulation of vascular tone, barrier function, inflammatory potential, and hemostasis are involved (23). The luminal surfaces of endothelial cells enter a pro-thrombotic state that favours the initiation of disseminated intravascular coagulation and the formation of microvascular thrombosis. The endothelial cell layer becomes increasingly permeable with subsequent fluid extravasation and edema formation. Activated endothelial cells start releasing nitric oxide which is held responsible for the hypotension of septic shock and express surface molecules that attract and adhere neutrophils which enhance inflammation (24). Shedding of the endothelial glycocalyx exposes hidden adhesion molecules that facilitate fixation and ultimately transmigration of leukocytes through the endothelial cell layer into the parenchyma. Destruction of the glycocalyx enhances capillary leakage and accelerates inflammation, platelet aggregation, coagulation, and loss of vascular tone (25). The systemic pro-inflammatory “burst” is then followed by an equally important anti-inflammatory response outlined by high levels of the anti-inflammatory cytokines IL-10 and IL-13 and in part reflected by reduced expression of HLA-DR on myeloid cells (26). Number and responsiveness of circulating lymphocytes and monocytes are reduced which generates an immunosuppressive state (27) resulting in ongoing or late infections. Enhanced apoptosis of follicular dendritic cells, B- and T-lymphocytes and epigenetic regulation of gene expression may be implicated (28,29) in this sepsis-associated “immunoparalysis” (30).
Several cytokine panels have been proposed as biomarkers of sepsis-induced MODS. IL-8 and monocyte chemotactic protein-1 were found to be associated with the SOFA score at sepsis onset. Plasma concentrations of IL-6 and granulocyte-colony stimulating factor are early predictors of worsening MODS (31).
Microcirculatory dysfunction
Anatomically, the microcirculation consists of small resistance (arterioles), exchange (capillaries), and capacitance (venules) vessels. The microcirculation primarily transports oxygen and nutrients to the tissues but also ensures important immunological functions and endothelial cell-derived interactions. Capillary blood flow throughout the microcirculation depends upon arteriolar tone, blood cell rheology, driving pressure, and capillary patency. In addition, microcirculatory perfusion is fine-tuned by regulatory metabolic, myogenic, and neurohumoral stimuli. The endothelial cell lining plays herein a central role by sensing these stimuli, signalling upstream information on downstream flow patterns, and controlling coagulation and immune function (23,32,33).
Septic patients can develop MODS in the presence of normal systemic hemodynamic and blood oxygenation parameters (34). This controversy is linked to a brutal and profound disruption of microcirculatory “homeostasis”. A reduction in capillary density together with outspoken variations in capillary perfusion, ranging from normal or intermittent to almost absent, are observed within the same vascular bed (35,36). Perfusion heterogeneity results in functional shunting and renders the microcirculation hypoxic along with a decrease in oxygen extraction capacity (37). Changes are ubiquitous and affect all organs. Clinically, this vents out as a global tissue oxygen deficit despite (supra)normal systemic oxygen delivery (38).
Mechanisms that may underlie the microcirculatory alterations in septic conditions are failing endothelial cell function and cross-talk, inappropriate activation of coagulation, rolling and adhesion of leukocytes to the endothelial surface, red blood cell deformation, and impaired release of nitric oxide (39). Inflammation-related damage to the microvascular endothelium turns the latter into a sieve through which protein-rich fluid leaks into the surrounding tissues (40). Activated endothelial cells liberate procoagulant factors which activate the coagulation cascade to form microthrombi (23). Additionally, the microcirculation becomes progressively clogged by slowly passing rigid red blood cells and crammed with deformed activated neutrophils (41,42). Smooth muscle cells surrounding the arterioles lose tone and become unresponsive to adrenergic stimuli.
All aforementioned alterations of the microvasculature occur shortly after injection of endotoxin or live bacteria in various animal models of sepsis. Microcirculatory perfusion is also significantly altered in human sepsis and septic shock (43,44). An association between the severity of microcirculatory derangement and the development of MODS has repeatedly been demonstrated (45,46).
Impaired mitochondrial function
Mitochondria are specialized cellular organelles that provide energy, in the form of adenosine triphosphate (ATP), through glycolysis, the tricarboxylic acid cycle, and oxidative phosphorylation (47). As such, they play a crucial role in cell functioning by supporting cells to respond to (patho)physiological stress. Mitochondria also regulate heat production, control intracellular calcium fluxes, are involved in apoptosis, and ascertain redox housekeeping (48-51). They are main producers of reactive oxygen species (ROS) which are required for cell signalling, maintenance of vascular tone, and oxygen sensing (52,53).
Sepsis has a profound impact on mitochondria (54,55). Systemic hypotension, myocardial depression and microcirculatory dysfunction all lead to impaired perfusion and tissue hypoxia, hence compromising oxygen supply at the mitochondrial level (56). Critically low oxygen levels decrease ATP generation and potentially trigger necrotic cell death. Sepsis also creates an “oxidative burst” characterized by excessive release of ROS (57). Phagocytic cells use ROS to eliminate invasive pathogens. However, ROS may become rapidly detrimental by inducing lipid peroxidation and degrading proteins. ROS, nitric oxide, and carbon monoxide all may directly damage the mitochondrial membrane, disrupt key mitochondrial enzyme processes, inhibit mitochondrial respiration, and induce apoptosis (58,59). Additionally, lower metabolic rates in sepsis have been associated with decreased amounts of mitochondrial DNA. Hormonal alterations, in particular the “low T3” syndrome, may adversely influence mitochondrial function (60). Finally, inflammation can downregulate genes transcribing mitochondrial proteins.
Mitochondrial dysfunction correlates with sepsis-related multi-organ failure but it remains to be unveiled whether mitochondria act as victims or amplifiers. Ultrastructural and functional mitochondrial injury related to bioenergetic decompensation, perpetuating oxidant stress, and a defective cell death regulatory function is thought to hamper energy production and inefficient oxygen use (61). In the end, cellular respiration falters and cellular metabolism considerably slows down. This state of “hibernation”, may initially benefit cell survival but may trigger MODS in a later phase. Mitochondria in se may also function as a source of oxidant stress and alterations in the mitochondrial respiratory chain complexes may become the driving force behind reduction in regional oxygen utilization (51). Whether directly implicated or not, impaired mitochondrial function forms the basis of the “cytopathic hypoxia” theory as a potential mechanism of sepsis-induced MODS (56).
Although substantial mitochondrial damage has been evidenced in experimental and human sepsis, it could not be elucidated whether the mitochondrial changes are involved in the development of organ dysfunction or merely a consequence of the inflammatory response to infection. However, the extent of mitochondrial impairment is found to be correlated with mortality and recovery of metabolic activity and organ function during resolving sepsis is expressed by markers of mitochondrial biogenesis (62,63).
Enhanced apoptosis
Apoptosis, or programmed cell death, is a prominent feature of sepsis (64). Although adaptive cell death might be beneficial as an attempt to limit tissue necrosis, a too exaggerated cell destruction is probably counterproductive. For instance, the observed excessive depletion of T and B lymphocytes (65) could substantially weaken the host immune defence capacity against invading pathogens. During ongoing sepsis, a high apoptosis intensity is also observed in intestinal and pulmonary epithelial cells. Overexpression of the anti-apoptotic protein BcL-2 with subsequent decrease in gut epithelial cell death is associated with a significant survival benefit in rodent sepsis (66,67). The Fas death receptor is implicated in the pathogenesis of sepsis-induced acute lung injury and acute respiratory distress syndrome (ARDS) (68). High levels of Fas and its ligand are detected in bronchoalveolar fluid of ARDS patients and correlate with mortality (69,70). Silencing of Fas on lung epithelial cells has been shown to prevent the development of lung injury in septic mice (71).
The gut as a “motor” of MODS
The human gut mucosa comprehends a single layer of columnar epithelial cells covering a surface area of approximately 32 m2 (72). Gut epithelial cells undergo continuous regeneration from multipotent stem cells. The latter express TLR4 which finetunes a delicate balance between cell proliferation and apoptosis (73). The gut epithelial surface primarily ensures absorption of nutrient components but also forms a solid barrier between the intestinal lumen and the para-intestinal structures and vasculature (74). The intestinal barrier allows paracellular movement of water, solutes and immune-modulating substances, while preventing migration of too large molecules and microorganisms. Trans-barrier transport occurs via apical tight junctions and junctional adherent molecules. The gut contains an enormous reservoir of bacterial cells, predominantly consisting of Bacteroides and Firmicutes species (75) which degrade the enteral supply of dietary polysaccharides, lipids, and proteins. Among the most important end products of fermentation are short-chain fatty acids that are essential for maintaining colonic integrity and metabolism (76). Eventually, the gut is also involved in host immune defence as it is the largest lymphoid organ of the human body (77). Critical illness, and sepsis in particular, may considerably perturb the complex crosstalk between the intestinal epithelium, microbiome, and immune system. Plasma levels of intestinal fatty acid-binding protein, a marker of enterocyte damage, and citrulline, a marker of functional enterocyte mass, are significantly altered in critically ill patients and independently associated with shock and 28-day mortality (78). Within this context, the gut may turn into “a motor of MODS” (79).
Several hypotheses about the role of the gut in sepsis-induced MODS have been proposed. One infers “bacterial translocation” as instigator of MODS. Critical illness in general already induces detrimental changes in nature and quality of the mucus layer (reduced thickness, diminished luminal coverage, and poor adherence) (80,81). Potential “disruptors” of mucus protection during sepsis are gut hypoperfusion, ischemia-reperfusion reactions, and inflammation. Consequently, bacteria may cross a weakened intestinal barrier and spread to and beyond the mesenteric lymph nodes (MLNs). This “translocation” with concomitant liberation of endotoxins is thought to propagate systemic inflammation leading to MODS. Bacteria from the normal microbiome may also become virulent and invasive in reaction to this altered host immune response (82).
Detection of viable bacteria in normally sterile MLNs is an indicator of intestinal barrier breakdown and subsequent translocation (83). As such, bacterial translocation has been recognized in patients undergoing major surgery, in severe pancreatitis or cirrhosis, and in organ donors (84,85). However, evidence linking bacterial and/or endotoxin translocation to MODS in critically ill septic patients is mainly indirect. A prospective study of serial portal blood samples in trauma patients found no evidence of portal or systemic bacteraemia within the first 5 days post injury despite a 30% incidence of MODS (86).
Another hypothesis suggests a relationship between MODS following a critical insult, including sepsis, and the release of non-bacterial pro-inflammatory factors from a “stressed” gut. Tissue injury occurs when these molecules reach the systemic circulation through the mesenteric lymphatic system (87). MLNs contain no bacteria and hardly detectable levels of endotoxin or cytokines. Instead, abundant protein and lipid mediators are present which behave as “danger molecules” and, likewise bacteria, stimulate TLR4 (88). In this model, antigen-presenting cells are activated by non-microbial, host-derived products of tissue injury which cause a systemic inflammatory response that may evolve to MODS (89). Several observations sustain this so-called “gut-lymph hypothesis” (90). Firstly, ligation of the major intestinal lymph duct to prohibit spilling of gut-derived factors into the systemic circulation can prevent the development of MODS (91). Secondly, a “shocked” gut releases biologically active factors into the mesenteric lymph which activate neutrophils and injure endothelial cells (90). Lastly, injection of shocked but not sham-shocked lymph into healthy rodents was found to generate a systemic septic state and MODS (90).
In the margin of the two aforementioned hypotheses, it is of note that pancreatic digestive enzymes may actively mediate epithelial cell disruption (92,93). Under normal conditions, these enzymes are sequestered in the intestinal lumen. With increasing mucosal permeability, however, they may penetrate the intestinal wall and precipitate a process of auto digestion that causes further crumbling of gut barrier function. Escape of enzymes or breakdown products from auto digestion into the circulation may enhance MODS (94).
Conclusions
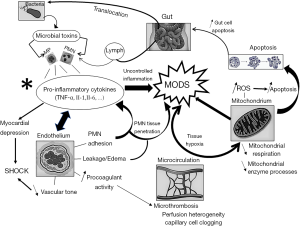
Despite a steadily growing insight in the mechanisms underlying sepsis-induced MODS, it is not known to what proportion all proposed pathways interfere in the process nor how they interact mutually. Excessive or uncontrolled inflammation, either endothelial-bound or gut-driven, is the predominant trigger. The resulting endothelial cell activation and damage is closely related with microcirculatory failure. A dysfunctional microcirculation, in turn, is held responsible for global tissue hypoxia with direct impact on mitochondrial behaviour. Assaulted mitochondria generate an excess of ROS which may contribute to unwarranted apoptosis. Figure 1 summarizes the different mechanisms (and their mutual interactions) involved in the development of MODS. Moreover, complex interactions have been described between organs involved in the process of MODS. For instance, sepsis-related acute kidney injury mediates a systemic inflammatory response that causes remote damage in heart, lung, brain, spleen, liver, and gut (95). Taken together, the pathophysiology of sepsis-induced MODS is fascinating and intricate but, at the same time, underscores the difficulty to develop a single effective therapeutic approach.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med 2013;369:840-51. [Crossref] [PubMed]
- Machiavelli N, Skinner Q, Price R. Machiavelli: The Prince. Cambridge: Cambridge University Press, 1988.
- Bone RC, Sibbald WJ, Sprung CL. The ACCP-SCCM consensus conference on sepsis and organ failure. Chest 1992;101:1481-3. [Crossref] [PubMed]
- Levy MM, Fink MP, Marshall JC, et al. 2001 SCCM/ESICM/ACCP/ ATS/SIS International Sepsis Definitions Conference. Crit Care Med 2003;31:1250-6. [Crossref] [PubMed]
- Shankar-Hari M, Phillips GS, Levy ML, et al. Sepsis Definitions Task Force. Developing a new definition and assessing new clinical criteria for septic shock: for the third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA 2016;315:775-87. [Crossref] [PubMed]
- Vincent JL, de Mendonça A, Cantraine F, et al. Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: results of a multicenter, prospective study. Working group on "sepsis-related problems" of the European Society of Intensive Care Medicine. Crit Care Med 1998;26:1793-800. [Crossref] [PubMed]
- Rhodes A, Evans LE, Alhazzani W, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Intensive Care Med 2017;43:304-77. [Crossref] [PubMed]
- Vincent JL. Organ failure in the intensive care unit. Semin Respir Crit Care Med 2011;32:541-2. [Crossref] [PubMed]
- Dinarello CA. Proinflammatory cytokines. Chest 2000;118:503-8. [Crossref] [PubMed]
- Liu SF, Malik AB. NF-kappa B activation as a pathological mechanism of septic shock and inflammation. Am J Physiol Lung Cell Mol Physiol 2006;290:L622-L645. [Crossref] [PubMed]
- Denk S, Perl M, Huber-Lang M. Damage- and pathogen-associated molecular patterns and alarmins: keys to sepsis? Eur Surg Res 2012;48:171-9. [Crossref] [PubMed]
- Opal SM. Endotoxins and other sepsis triggers. Contrib Nephrol 2010;167:14-24. [Crossref] [PubMed]
- Poli-de-Figueiredo LF, Garrido AG, Nakagawa N, et al. Experimental models of sepsis and their clinical relevance. Shock 2008;30:53-9. [Crossref] [PubMed]
- Martich GD, Boujoukos AJ, Suffredini AF. Response of man to endotoxin. Immunobiology 1993;187:403-16. [Crossref] [PubMed]
- Fullerton JN, Segre E, De Maeyer RP, et al. Intravenous endotoxin challenge in healthy humans: An experimental platform to investigate and modulate systemic inflammation. J Vis Exp 2016. [Crossref] [PubMed]
- Chong DL, Sriskandan S. Pro-inflammatory mechanisms in sepsis. Contrib Microbiol 2011;17:86-107. [Crossref] [PubMed]
- Weinberg JR, Boyle P, Meager A, et al. Lipopolysaccharide, tumor necrosis factor, and interleukin-1 interact to cause hypotension. J Lab Clin Med 1992;120:205-11. [PubMed]
- Marcos-Ramiro B, García-Weber D, Millán J. TNF-induced endothelial barrier disruption: beyond actin and Rho. Thromb Haemost 2014;112:1088-102. [Crossref] [PubMed]
- Zhang H, Park Y, Wu J, et al. Role of TNF-alpha in vascular dysfunction. Clin Sci (Lond) 2009;116:219-30. [Crossref] [PubMed]
- Pober JS. Effects of tumour necrosis factor and related cytokines on vascular endothelial cells. Ciba Found Symp 1987;131:170-84. [PubMed]
- Cain BS, Meldrum DR, Dinarello CA, et al. Tumor necrosis factor-alpha and interleukin-1beta synergistically depress human myocardial function. Crit Care Med 1999;27:1309-18. [Crossref] [PubMed]
- Ishihara K, Hirano T. IL-6 in autoimmune disease and chronic inflammatory proliferative disease. Cytokine Growth Factor Rev 2002;13:357-68. [Crossref] [PubMed]
- Ince C, Mayeux PR, Nguyen T, et al. The endothelium in sepsis. Shock 2016;45:259-70. [Crossref] [PubMed]
- Dimasi D, Sun WY, Bonder CS. Neutrophil interactions with the vascular endothelium. Int Immunopharmacol 2013;17:1167-75. [Crossref] [PubMed]
- Chelazzi C, Villa G, Mancinelli P, et al. Glycocalyx and sepsis-induced alterations in vascular permeability. Crit Care 2015;19:26. [Crossref] [PubMed]
- Boomer JS, To K, Chang KC, et al. Immunosuppression in patients who die of sepsis and multiple organ failure. JAMA 2011;306:2594-605. [Crossref] [PubMed]
- Shubin NJ, Monaghan SF, Ayala A. Anti-inflammatory mechanisms of sepsis. Contrib Microbiol 2011;17:108-24. [Crossref] [PubMed]
- Hotchkiss RS, Tinsley KW, Swanson PE, et al. Depletion of dendritic cells, but not macrophages, in patients with sepsis. J Immunol 2002;168:2493-500. [Crossref] [PubMed]
- Hotchkiss RS, Tinsley KW, Swanson PE, et al. Sepsis-induced apoptosis causes progressive profound depletion of B and CD4+ T lymphocytes in humans. J Immunol 2001;166:6952-63. [Crossref] [PubMed]
- Carson WF, Cavassani KA, Dou Y, et al. Epigenetic regulation of immune cell functions during post-septic immunosuppression. Epigenetics 2011;6:273-83. [Crossref] [PubMed]
- Chaudhry H, Zhou J, Zhong Y, et al. Role of cytokines as a double-edged sword in sepsis. In Vivo 2013;27:669-84. [PubMed]
- Ait-Oufella H, Maury E, Lehoux S, et al. The endothelium: physiological functions and role in microcirculatory failure during severe sepsis. Intensive Care Med 2010;36:1286-98. [Crossref] [PubMed]
- Boerma EC. The microcirculation as a clinical concept: work in progress. Curr Opin Crit Care 2009;15:261-5. [Crossref] [PubMed]
- Hotchkiss RS, Karl IE. The pathophysiology and treatment of sepsis. N Engl J Med 2003;348:138-50. [Crossref] [PubMed]
- Farquhar I, Martin CM, Lam C, et al. Decreased capillary density in vivo in bowel mucosa of rats with normotensive sepsis. J Surg Res 1996;61:190-6. [Crossref] [PubMed]
- Humer MF, Phang PT, Friesen BP, et al. Heterogeneity of gut capillary transit times and impaired gut oxygen extraction in endotoxemic pigs. J Appl Physiol 1985;1996:895-904. [PubMed]
- Goldman D, Bateman RM, Ellis CG. Effect of decreased O2 supply on skeletal muscle oxygenation and O2 consumption during sepsis: role of heterogeneous capillary spacing and blood flow. Am J Physiol Heart Circ Physiol 2006;290:H2277-85. [Crossref] [PubMed]
- Ellis CG, Bateman RM, Sharpe MD, et al. Effect of a maldistribution of microvascular blood flow on capillary O(2) extraction in sepsis. Am J Physiol Heart Circ Physiol 2002;282:H156-64. [PubMed]
- Colbert JF, Schmidt EP. Endothelial and microcirculatory function and dysfunction in sepsis. Clin Chest Med 2016;37:263-75. [Crossref] [PubMed]
- Opal SM, van der Poll T. Endothelial barrier dysfunction in septic shock. J Intern Med 2015;277:277-93. [Crossref] [PubMed]
- Reggiori G, Occhipinti G, De Gasperi A, et al. Early alterations of red blood cell rheology in critically ill patients. Crit Care Med 2009;37:3041-6. [Crossref] [PubMed]
- Pinheiro da Silva F, Soriano FG. Neutrophils recruitment during sepsis: critical points and crossroads. Front Biosci (Landmark Ed) 2009;14:4464-76. [Crossref] [PubMed]
- De Backer D, Creteur J, Preiser JC, et al. Microvascular blood flow is altered in patients with sepsis. Am J Respir Crit Care Med 2002;166:98-104. [Crossref] [PubMed]
- Donati A, Damiani E, Domizi R, et al. Alteration of the sublingual microvascular glycocalyx in critically ill patients. Microvasc Res 2013;90:86-9. [Crossref] [PubMed]
- Shapiro NI, Arnold R, Sherwin R, et al. The association of near infrared spectroscopy-derived tissue oxygenation measurements with sepsis syndromes, organ dysfunction and mortality in emergency department patients with sepsis. Crit Care 2011;15:R223. [Crossref] [PubMed]
- Top AP, Ince C, de Meij N, et al. Persistent low microcirculatory vessel density in nonsurvivors of sepsis in pediatric intensive care. Crit Care Med 2011;39:8-13. [Crossref] [PubMed]
- Chan DC. Mitochondria: dynamic organelles in disease, aging, and development. Cell 2006;125:1241-52. [Crossref] [PubMed]
- McBride HM, Neuspiel M, Wasiak S. Mitochondria: more than just a powerhouse. Curr Biol 2006;16:R551-60. [Crossref] [PubMed]
- Yin F, Sancheti H, Cadenas E. Mitochondrial thiols in the regulation of cell death pathways. Antioxid Redox Signal 2012;17:1714-27. [Crossref] [PubMed]
- Osellame LD, Blacker TS, Duchen MR. Cellular and molecular mechanisms of mitochondrial function. Best Pract Res Clin Endocrinol Metab 2012;26:711-23. [Crossref] [PubMed]
- Figueira TR, Barros MH, Camargo AA, et al. Mitochondria as a source of reactive oxygen and nitrogen species: from molecular mechanisms to human health. Antioxid Redox Signal 2013;18:2029-74. [Crossref] [PubMed]
- Turrens JF. Mitochondrial formation of reactive oxygen species. J Physiol 2003;552:335-44. [Crossref] [PubMed]
- Larsen FJ, Schiffer TA, Weitzberg E, et al. Regulation of mitochondrial function and energetics by reactive nitrogen oxides. Free Radic Biol Med 2012;53:1919-28. [Crossref] [PubMed]
- Duran-Bedolla J, Montes de Oca-Sandoval MA, Saldaña-Navor V, et al. Sepsis, mitochondrial failure and multiple organ dysfunction. Clin Invest Med 2014;37:E58-69. [Crossref] [PubMed]
- Crouser ED, Julian MW, Huff JE, et al. Abnormal permeability of inner and outer mitochondrial membranes contributes independently to mitochondrial dysfunction in the liver during acute endotoxemia. Crit Care Med 2004;32:478-88. [Crossref] [PubMed]
- Fink MP. Cytopathic hypoxia. Mitochondrial dysfunction as mechanism contributing to organ dysfunction in sepsis. Crit Care Clin 2001;17:219-37. [Crossref] [PubMed]
- Galley HF. Oxidative stress and mitochondrial dysfunction in sepsis. Br J Anaesth 2011;107:57-64. [Crossref] [PubMed]
- Szabó C, Módis K. Pathophysiological roles of peroxynitrite in circulatory shock. Shock 2010;34:4-14. [Crossref] [PubMed]
- Green DR, Kroemer G. The pathophysiology of mitochondrial cell death. Science 2004;305:626-9. [Crossref] [PubMed]
- Harper ME, Seifert EL. Thyroid hormone effects on mitochondrial energetics. Thyroid 2008;18:145-56. [Crossref] [PubMed]
- Levy RJ. Mitochondrial dysfunction, bioenergetic impairment, and metabolic down-regulation in sepsis. Shock 2007;28:24-8. [Crossref] [PubMed]
- Carré JE, Orban JC, Re L, et al. Survival in critical illness is associated with early activation of mitochondrial biogenesis. Am J Respir Crit Care Med 2010;182:745-51. [Crossref] [PubMed]
- Haden DW, Suliman HB, Carraway MS, et al. Mitochondrial biogenesis restores oxidative metabolism during Staphylococcus aureus sepsis. Am J Respir Crit Care Med 2007;176:768-77. [Crossref] [PubMed]
- Hattori Y, Takano K, Teramae H, et al. Insights into sepsis therapeutic design based on the apoptotic death pathway. J Pharmacol Sci 2010;114:354-65. [Crossref] [PubMed]
- Hotchkiss RS, Swanson PE, Knudson CM, et al. Overexpression of Bcl-2 in transgenic mice decreases apoptosis and improves survival in sepsis. J Immunol 1999;162:4148-56. [PubMed]
- Coopersmith CM, Chang KC, Swanson PE, et al. Overexpression of Bcl-2 in the intestinal epithelium improves survival in septic mice. Crit Care Med 2002;30:195-201. [Crossref] [PubMed]
- Coopersmith CM, Stromberg PE, Dunne WM, et al. Inhibition of intestinal epithelial apoptosis and survival in a murine model of pneumonia-induced sepsis. JAMA 2002;287:1716-21. [Crossref] [PubMed]
- Chopra M, Reuben JS, Sharma AC. Acute lung injury: apoptosis and signalling mechanisms. Exp Biol Med (Maywood) 2009;234:361-71. [Crossref] [PubMed]
- Albertine KH, Soulier MF, Wang Z, et al. Fas and fas ligand are up-regulated in pulmonary edema fluid and lung tissue of patients with acute lung injury and the acute respiratory distress syndrome. Am J Pathol 2002;161:1783-96. [Crossref] [PubMed]
- Matute-Bello G, Liles WC, Steinberg KP, et al. Soluble Fas ligand induces epithelial cell apoptosis in humans with acute lung injury (ARDS). J Immunol 1999;163:2217-25. [PubMed]
- Perl M, Chung CS, Lomas-Neira J, et al. Silencing of Fas, but not caspase-8, in lung epithelial cells ameliorates pulmonary apoptosis, inflammation, and neutrophil influx after hemorrhagic shock and sepsis. Am J Pathol 2005;167:1545-59. [Crossref] [PubMed]
- Helander HF, Fandriks L. Surface area of the digestive tract-revisited. Scand J Gastroenterol 2014;49:681-9. [Crossref] [PubMed]
- Neal MD, Sodhi CP, Jia H, et al. Toll-like receptor 4 is expressed on intestinal stem cells and regulates their proliferation and apoptosis via the p53 up-regulated modulator of apoptosis. J Biol Chem 2012;287:37296-308. [Crossref] [PubMed]
- Bischoff SC, Barbara G, Buurman W, et al. Intestinal permeability-a new target for disease prevention and therapy. BMC Gastroenterol 2014;14:189. [Crossref] [PubMed]
- Defazio J, Fleming ID, Shakhsheer B, et al. The opposing forces of the intestinal microbiome and the emerging pathobiome. Surg Clin North Am 2014;94:1151-61. [Crossref] [PubMed]
- Patterson E, Cryan JF, Fitzgerald GF, et al. Gut microbiota, the pharmabiotics they produce and host health. Proc Nutr Soc 2014;73:477-89. [Crossref] [PubMed]
- Galperin C, Gershwin ME. Immunopathogenesis of gastrointestinal and hepatobiliary diseases. JAMA 1997;278:1946-55. [Crossref] [PubMed]
- Piton G, Belon F, Cypriani B, et al. Enterocyte damage in critically ill patients is associated with shock condition and 28-day mortality. Crit Care Med 2013;41:2169-76. [Crossref] [PubMed]
- Clark JA, Coopersmith CM. Intestinal crosstalk: a new paradigm for understanding the gut as the "motor" of critical illness. Shock 2007;28:384-93. [Crossref] [PubMed]
- MacFie J, O'Boyle CJ, Mitchell CJ, et al. Gut origin of sepsis: a prospective study investigating the association between bacterial translocation, gastric microflora, and septic morbidity. Gut 1999;45:223-8. [Crossref] [PubMed]
- Qin X, Sheth SU, Sharpe SM, et al. The mucus layer is critical in protecting against ischemia-reperfusion-mediated gut injury and in the restitution of gut barrier function. Shock 2011;35:275-81. [Crossref] [PubMed]
- Wu L, Estrada O, Zaborina O, et al. Recognition of host immune activation by Pseudomonas aeruginosa. Science 2005;309:774-7. [Crossref] [PubMed]
- Gatt M, Reddy BS, MacFie J. Bacterial translocation in the critically ill-evidence and methods of prevention. Aliment Pharmacol Ther 2007;25:741-57. [Crossref] [PubMed]
- MacFie J, Reddy BS, Gatt M, et al. Bacterial translocation studied in 927 patients over 13 years. Br J Surg 2006;93:87-93. [Crossref] [PubMed]
- Reddy BS, MacFie J, Gatt M, et al. Commensal bacteria do translocate across the intestinal barrier in surgical patients. Clin Nutr 2007;26:208-15. [Crossref] [PubMed]
- Moore FA, Moore EE, Poggetti R, et al. Gut bacterial translocation via the portal vein: a clinical perspective with major torso trauma. J Trauma 1991;31:629-36. [Crossref] [PubMed]
- Deitch EA. Gut-origin sepsis: evolution of a concept. Surgeon 2012;10:350-6. [Crossref] [PubMed]
- Reino DC, Pisarenko V, Palange D, et al. Trauma hemorrhagic shock-induced lung injury involves a gut-lymph-induced TLR4 pathway in mice. PLoS One 2011;6:e14829. [Crossref] [PubMed]
- Matzinger P. The danger model: a renewed sense of self. Science 2002;296:301-5. [Crossref] [PubMed]
- Deitch EA, Xu DZ, Lu Q. Gut lymph hypothesis of early shock and trauma-induced multiple organ dysfunction syndrome: A new look at gut origin sepsis. J Organ Dysf 2009;2:70-9. [Crossref]
- Badami CD, Senthil M, Caputo FJ, et al. Mesenteric lymph duct ligation improves survival in a lethal shock model. Shock 2008;30:680-5. [Crossref] [PubMed]
- Chang M, Alsaigh T, Kistler EB, et al. Breakdown of mucin as barrier to digestive enzymes in the ischemic rat small intestine. PLoS One 2012;7:e40087. [Crossref] [PubMed]
- Sharpe SM, Qin X, Lu Q, et al. Loss of the intestinal mucus layer in the normal rat causes gut injury but not toxic mesenteric lymph nor lung injury. Shock 2010;34:475-81. [Crossref] [PubMed]
- Schmid-Schonbein GW, DeLano FA, Penn AH, et al. An elementary analysis of physiologic shock and multi-organ failure: the autodigestion hypothesis. Conf Proc IEEE Eng Med Biol Soc 2012;2012:3114-5.
- Ologunde R, Zhao H, Lu K, et al. Organ cross talk and remote organ damage following acute kidney injury. Int Urol Nephrol 2014;46:2337-45. [Crossref] [PubMed]
Cite this article as: Spapen HD, Jacobs R, Honoré PM. Sepsis-induced multi-organ dysfunction syndrome—a mechanistic approach. J Emerg Crit Care Med 2017;1:27.


