Hyperoxemia reduces cerebrovascular accidents post coronary artery bypass graft surgery: a retrospective cohort study
Introduction
Cerebrovascular accidents (CVAs) including strokes and transient ischaemic attacks (TIAs) form a major portion of perioperative morbidity, occurring in approximately three to ten percent of surgical patients requiring cardiopulmonary bypass (CPB) (1,2). These can be severely disabling complications, associated with a 3- to 6-fold increase in death after cardiac surgery (3). Despite multiple anaesthetic and surgical advancements over the past decade, these patients remain at high risk of adverse post-operative neurologic outcomes (4).
There remains a poor understanding of the pathophysiology of neurologic dysfunction in these post-operative patients (5). Proposed causes include tissue hypoxia, formation of reactive oxygen species (ROS), along with the formation of micro/macro-emboli (6). These theories are closely related to the (well established) risk factors for the development of perioperative CVA such as diabetes, hypertension, previous stroke and intra-operative hypothermia (7).
Whilst the exact pathophysiology of these complications remains unknown, their impacts on patient morbidity is unquestionable. Severe pre-operative, intra-operative and post-operative cerebral and circulatory oxygen levels in patients undergoing cardiac surgery have been associated with postoperative cognitive & neuropsychological dysfunction, prolonged mechanical ventilation time, intensive care unit admission, tracheostomy requirement, increased resource utilisation and most recently, CVAs (8,9).
Treatments to counteract this effect are still being studied, however current suggestions include ongoing intraoperative cerebral oxygen monitoring and supplemental oxygen (10). Given the high rate and severity of complications, any intervention reducing the rate of complications would be expected to significantly benefit patient outcomes (11).
Further to the above, a recent study has established intraoperative hypoxia as an independent risk factor for perioperative CVA (12). Another study extrapolated their data to suggest cerebral oxygen saturation be used as an independent predictor for preoperative cardiac surgery risk stratification (13). Interestingly, whilst intraoperative hyperoxia has been linked to numerous adverse end-organ effects (14), these outcomes have not been demonstrated in the cardiac surgery population.
Objectives
The primary aim of this study was to examine whether there was a relationship between increasing the intraoperative partial pressure of arterial oxygen (PaO2) and decreasing postoperative CVA risk.
Secondary aims were to examine the relationship between intraoperative PaO2 and other post-operative adverse outcomes including pneumonia, prolonged postoperative ventilation, perioperative myocardial infarction or cardiac arrest, sternal wound infection, sepsis and renal failure.
Methods
Patients and data sources
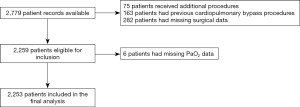
This was a retrospective single-centre cohort study that included 2,779 consecutive patients who underwent on-pump coronary artery bypass grafting (CABG) between June 2000 and May 2013 at St. Vincent’s Hospital, a major Australian cardiothoracic surgical and teaching hospital in Darlinghurst, Sydney (Figure 1).
The St. Vincent’s Department of Cardiothoracic Surgery databases were analysed to extract information on all patients who met the inclusion criteria. Variables assessed included patient co-morbidities, surgical outcomes, and perioperative observations collected at 5-minute intervals. To minimize error, all data was prospectively collected and validated by reviewing original patient medical records and checked against electronic recordings from the central surgical databases and the CPB machine.
Inclusion and exclusion criteria
For this analysis, we included adult patients, who were scheduled for CABG with CPB. All rescue, emergency and elective procedures were included in this cohort.
Exclusion criteria included receiving multiple cardiothoracic procedures at the time of surgery (including valve or ascending aorta replacements), previous cardiothoracic surgery requiring CPB, death within 48 hours after the operation, and incomplete patient data.
Human Research Ethics Committee (HREC) approval was obtained for the study via the HREC at St. Vincent’s Hospital, Sydney (Reference number 13/011).
Surgical procedure
All procedures were performed through a median sternotomy. CPB was established via a two-stage right atrial venous cannula with arterial return to the ascending aorta. Standard cold blood cardioplegia was used for myocardial protection, solely antegrade in 72.5% of cases, both ante- and retrograde in 22.8% of cases and either warm blood or cold crystalloid in the remainder.
Variables
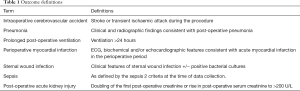
The primary outcome variable for this study was a perioperative CVA, defined as either a stroke or TIA during the procedure or within 30 days after the surgery (15). A stroke was further defined as a persistent neurological deficit with changes on neurological imaging consistent with clinical signs. A TIA was defined as either an initial neurological deficit consistent with vascular pathology and resolving within 24 hours (as per the old definition), or by the absence of radiographic changes on MRI of the brain (as per the new definition) (16).
The main explanatory variable was intraoperative PaO2. Arterial blood samples were taken at 5-minute intervals and a range of parameters were measured. For the purposes of this study the average and nadir PaO2 were used for statistical analysis. The odds ratio (OR) for PaO2 was calculated as follows:
- Intraoperative PaO2 in CVA patients;
- Intraoperative PaO2 in non-CVA patients.
Medically focused covariates used in the primary analysis included previous CVA, myocardial infarction, smoking (prior or current), hypertension, hypercholesterolaemia, family history of cardiovascular disease, pre-existing peripheral vascular disease and pre-existing heart failure. Surgically related co-variates reviewed in this study included the use of a perioperative intra-aortic balloon pump, total bypass time and total aortic cross-clamp time. Definitions for the above are listed in Table 1.
Full table
Secondary outcomes to assess for adverse postoperative outcomes included pneumonia (diagnosed on the basis of clinical and radiographic findings during admission), prolonged post-operative ventilation (>24 hours), perioperative myocardial infarction or cardiac arrest, sternal wound infection, sepsis and post-operative acute kidney injury. Acute kidney injury was further defined as a doubling of the first post-operative creatinine or rise in post-operative serum creatinine to >200 U/L. Co-variates used in these analyses included additional clinically relevant risk factors, immunosuppression, pre-existing renal or respiratory disease, and pre-operative cardiogenic shock.
Statistical analysis
Statistical analysis was performed using Stata statistics software package, version 14.2 [2012]. Univariate and then multivariate regression analyses were used to analyse the association between the incidence of perioperative CVA and average/nadir intra-operative PaO2, controlling for cardiovascular and intraoperative risk factors. Results of the univariate analysis were considered for inclusion in the multivariate model for significance level P≤0.025. Results from multivariate analysis were considered significant for P<0.05.
Results
A total of 2,779 patients were available for analysis, of these 526 were excluded. Exclusions for this study included: 75 patients who received additional cardiothoracic procedures at the time of surgery, including valve or ascending aorta replacement; 163 who had previous cardiothoracic surgery requiring CPB; and 282 for whom surgical outcome data were unavailable. Six patients had incomplete data pertaining to their intraoperative PaO2 levels and were excluded from analysis. The dataset was complete for all other variables. A total of 2,253 patients were therefore included in the final multivariate analysis.
Patient demographic data and comorbidities are contained in Table 2 below. There were more male than female patients in this cohort (78% compared to 22%). Average age was 65 years. The majority (70%) of CABG procedures were elective. The mean PaO2 was 519.6 mmHg (SD 40.0 mmHg; range, 313–654 mmHg). The PaO2 nadir ranged from 38 to 654 mmHg. The number of patients who had a perioperative CVA was 28 (1.2%).

Full table
Primary analysis
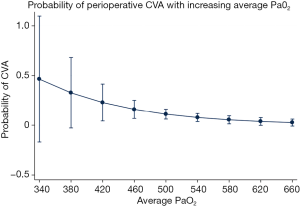
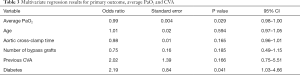
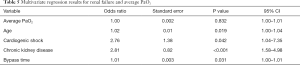
Results from the multivariate logistic regression model are included in Tables 3,4. There was a significant correlation between perioperative CVA and average PaO2 (OR 0.99; P=0.029; 95% CI, 0.98–1.00), indicating a reduction in CVA risk with increasing PaO2. This is demonstrated visually with Figure 2. There was also a significant correlation between perioperative CVA and diabetes (OR 2.19; P=0.041; 95% CI: 1.03–4.66).
Full table
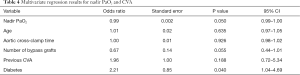
Full table
These results were reflected when using the nadir PaO2 (Table 4) with a significant correlation between perioperative CVA and nadir PaO2 (OR 0.99; P=0.050; 95% CI: 0.99–1.00). Unsurprisingly, the significant correlation between perioperative CVA and diabetes remained (OR 2.21; P=0.040; 95% CI: 1.04–4.69).
Secondary outcomes
Post-operative pneumonia occurred in 30 cases (1.33%). There was no correlation between post-operative pneumonia and perioperative average PaO2 (OR 1.00; P=0.51; 95% CI: 0.99–1.01).
Prolonged ventilation occurred in 102 cases (4.5%). Multivariate analysis revealed a significant association between prolonged ventilation and average PaO2 (OR 0.99; P=0.03; 95% CI: 0.99–1.00). This suggested that increasing the average PaO2 reduced the likelihood of patients requiring prolonged ventilation of more than 24. Pre-existing respiratory disease was a better predictor of prolonged ventilation (OR 1.73; P=0.03; 95% CI: 1.04–2.87).
Perioperative myocardial infarction or cardiac arrest occurred in 42 (1.86%) cases. These cardiac complications had no association with intraoperative average PaO2 (OR 1.00; P=0.25; 95% CI: 1.00–1.01). Deep sternal wound infection occurred in 20 cases (0.9%) and sepsis occurred in 10 cases (0.4%). In multivariate analyses there was no association between sternal wound infection and average PaO2 (OR 0.99; P=0.21; 95% CI: 0.98–1.00). There was no significant correlation between sepsis and average PaO2, however immunosuppression was associated with increased risk of sepsis (OR 9.33; P=0.04; 95% CI: 1.09–79.54).
Finally, renal failure occurred in 118 cases (5.2%). In multivariate analysis average PaO2 was not associated with post-operative renal failure, however age, cardiogenic shock, chronic kidney disease and bypass time were significant predictors of renal failure in this analysis (Table 5).
Full table
Discussion
This study found that the increased average PaO2 and nadir PaO2 values were associated with a decreased risk of perioperative CVA. Even though there may be a limited magnitude of clinical significance, this data suggests that avoiding hypoxia might be a potential target for the treatment to reduce the debilitating and life-threatening burden of perioperative CVAs.
This thesis follows the paradigm of a recent study by Dunham et al. which demonstrated an increased stroke risk with intraoperative hypoxaemia as measured by arterial oxygen nadir and oxygen pressures (12). Our study builds on this by investigating a lager cohort with a broader arterial oxygen nadir (38–654 mmHg) and higher mean intra-operative arterial oxygen pressures (340–660 mmHg). Overall, given the findings, it would be reasonable to hypothesise a direct relationship between CPB CVA and reduced perioperative PaO2.
As with most disease processes, the cause is likely to be multifactorial. Pre-existing cerebrovascular disease, post-operative arrhythmias and the older age group all increase the risk of CVA (17). Further, the use of CPB increases the risk of introducing synthetic particles or air emboli into the blood stream, though this has been minimized through using an arterial screen filter (18). As such, hyperoxemia may reduce the incidence of post CPB stroke through reducing gaseous microemboli (19-21). The formation of microemboli during CPB by the extracorporeal circuit and entrainment into open heart chambers has been well described (6). These microemboli may progress to obstruct end-organ capillary beds. Given that these microemboli consist of air, hyperoxia will cause denitrogenation and reduce the size of these microemboli. Prior ultrasound studies have demonstrated the utility of post-operative hyperoxia to significantly reduce the microemboli burden following CPB (22).
Interestingly, Stroobant et al. failed to find any relationship between cognitive decline and US imaging findings in perioperative CABG patients (23). The role of intra-operative emboli needs further investigation, however, such analysis is outside the scope of this study.
Our findings are more suggestive of postoperative CPB CVA being related to tissue level hypoxia, as opposed to embolization. However, it should be noted that hypo-perfusion and embolism may co-exist.
A potential mechanism for the protective effect of increased arterial oxygen pressure may relate to a reduction in the initial tissue level hypoxic insult and thus preventing the cascade resulting in the formation of ROS. The protective effect of hyperoxemia is contrary to the growing body of non-cardiac surgery literature, which demonstrates the harmful effect of hyperoxia through the formation of ROS (24,25). If this is the case, the hyperoxia would counter-intuitively reduce ROS formation and thus prevent end organ ischaemia.
One final potential mechanism includes hyperoxia induced preconditioning. Both animal and human studies have previously demonstrated a neuroprotective effect with hyperbaric hyperoxic conditions. The mechanism by which this hyperoxia induces tolerance to ischaemic events is unclear.
Importantly, the reduction in post CABG CVA risk did not carry any associated increase in adverse effects. There was no correlation between increasing levels of hyperoxia and infective outcomes such as pneumonia, deep sternal wound infection and sepsis. This is in keeping with many prior studies which have demonstrated increased rates of infection associated with hypoxia and a no effect with increasing oxygen levels ranging from normoxia through to hyperoxia (22,26).
Increasing levels of hyperoxia also had no impact on the incidence of renal failure, myocardial infarction or cardiac arrest. The incidence of renal failure is a poorly studied outcome with varying definitions used for renal failure and acute kidney injury (6). This reinforces the results of a randomised controlled trials by Toraman et al. and McGuinness et al. which showed similar results, with no significant difference between near physiologic and hyperoxic groups (27,28). Similar to acute kidney injury, a wide variety of definitions have been used to investigate myocardial injury and infarction (6). The vast majority of these studies have found no link between hyperoxia and myocardial infarction. The present study is in keeping with current literature.
Limitations
There were several limitations to this retrospective study. Firstly, there were no hypoxic patients to compare stroke risk to. However, the incidence of reduced stroke risk has been demonstrated in several prior studies and the aim of the present study was to determine if increasing levels of hyperoxia further reduced stroke risk without an increase in other complications. Secondly, there was a low-normal incidence of post-CABG CVA in the cohort of patients included in this study. The incidence was 1.2%, this was potentially related to the much greater levels of intraoperative hyperoxemia than has been reported in previous studies. There were also the intrinsic limitations of any retrospective study including the potential for unmeasured confounding variables that may have been missed in the multi-variate analysis.
Conclusions
We have demonstrated a significant trend between average intraoperative PaO2 and perioperative CVA, with CVA risk declining with increasing PaO2. Higher PaO2 was not demonstrated to be associated with increased risk of post-operative adverse outcomes including pneumonia, prolonged ventilation, perioperative myocardial infarction or cardiac arrest, sternal wound infection, sepsis or renal failure.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- McKhann GM, Goldsborough M, Borowicz L, et al. Predictors of stroke risk in coronary artery bypass patients. Ann Thorac Surg 1997;63:516-21. [Crossref] [PubMed]
- Selim M. Perioperative stroke. N Engl J Med 2007;356:706-13. [Crossref] [PubMed]
- Dacey LJ, Likosky D, Leavitt B, et al. Perioperative stroke and long-term survival after coronary bypass graft surgery. Ann Thorac Surg 2005;79:532-6. [Crossref] [PubMed]
- Selnes OA, Gottesman RF, Grega MA, et al. Cognitive and Neurologic Outcomes after Coronary-Artery Bypass Surgery. N Engl J Med 2012;366:250-7. [Crossref] [PubMed]
- Tan AMY, Amoako D. Postoperative cognitive dysfunction after cardiac surgery. Contin Educ Anaesth Crit Care Pain 2013;13:218-23. [Crossref]
- Heinrichs J, Grocott H. Pro: Hyperoxia Should Be Used During Cardiac Surgery. J Cardiothorac Vasc Anesth 2019;33:2070-4. [Crossref] [PubMed]
- Ko SB. Perioperative stroke: pathophysiology and management. Korean J Anesthesiol 2018;71:3-11. [Crossref] [PubMed]
- Yao FS, Tseng C, Ho C, et al. Cerebral oxygen desaturation is assoc w early postoperative neuropsychological dysfunction I patients undergoing cardiac surgery. J Cardiothorac Vasc Anesth 2004;18:552-8. [Crossref] [PubMed]
- Murkin JM, Adams S, Novick R, et al. Monitoring brain oxygen saturation during coronary bypass surgery: a randomized, prospective study. Anesth Analg 2007;104:51-8. [Crossref] [PubMed]
- Ranucci M, Ballotta A, La Rovere M, Castelvecchio S. Postoperative hypoxia and length of ICU stay after cardiac surgery: the underweight paradox. PLoS One 2014;9:e93992. [Crossref] [PubMed]
- Cowie DA, Nazareth J, Story D. Cerebral oximetry to reduce perioperative morbidity. Anaesth Intensive Care 2014;42:310-4. [Crossref] [PubMed]
- Dunham AM, Grega M, Brown C, et al. Perioperative Low Arterial Oxygenation Is Associated With Increased Stroke Risk in Cardiac Surgery. Anesth Analg 2017;125:38-43. [Crossref] [PubMed]
- Heringlake M, Garbers C, Käbler J, et al. Preoperative cerebral oxygen saturation and clinical outcomes in cardiac surgery. Anesthesiology 2011;114:58-69. [Crossref] [PubMed]
- Beck-Nielsen H, Hother-Nielsen O, Staehr P. Is hepatic glucose production increased in type 2 diabetes mellitus? Curr Diab Rep 2002;2:231-6. [Crossref] [PubMed]
- Mashour GA, Moore L, Lele A, et al. Perioperative care of patients at high risk for stroke during or after non-cardiac, non-neurologic surgery: consensus statement from the Society for Neuroscience in Anesthesiology and Critical Care. J Neurosurg Anesthesiol 2014;26:273-85. [Crossref] [PubMed]
- Saver JL. Proposal for a universal definition of cerebral infarction. Stroke 2008;39:3110-5. [Crossref] [PubMed]
- Bowles BJ, Lee J, Dang C, et al. Coronary artery bypass performed without the use of cardiopulmonary bypass is associated with reduced cerebral microemboli and improved clinical results. Chest 2001;119:25-30. [Crossref] [PubMed]
- Machin D, Allsager C. Principles of cardiopulmonary bypass. Contin Educ Anaesth Crit Care Pain 2006;6:176-81. [Crossref]
- Pugsley W, Klinger L, Paschalis C, et al. The impact of microemboli during cardiopulmonary bypass on neuropsychological functioning. Stroke 1994;25:1393-9. [Crossref] [PubMed]
- Sreeram GM, Grocott H, White W, et al. Transcranial Doppler emboli count predicts rise in creatinine after coronary artery bypass graft surgery. J Cardiothorac Vasc Anesth 2004;18:548-51. [Crossref] [PubMed]
- Borger MA, Feindel C. Cerebral emboli during cardiopulmonary bypass: effect of perfusionist interventions and aortic cannulas. J Extra Corpor Technol 2002;34:29-33. [PubMed]
- Greif R, Akça O, Horn E, et al. Supplemental perioperative oxygen to reduce the incidence of surgical-wound infection. N Engl J Med 2000;342:161-7. [Crossref] [PubMed]
- Stroobant N, Van Nooten G, Van Belleghem Y, et al. The effect of CABG on neurocognitive functioning. Acta Cardiol 2010;65:557-64. [Crossref] [PubMed]
- Frank A, Bonney M, Bonney S, et al. Myocardial ischemia reperfusion injury: from basic science to clinical bedside. Semin Cardiothorac Vasc Anesth 2012;16:123-32. [Crossref] [PubMed]
- Ansley DM, Raedschelders K, Choi P, et al. Propofol cardioprotection for on-pump aortocoronary bypass surgery in patients with type 2 diabetes mellitus (PRO-TECT II): a phase 2 randomized-controlled trial. Can J Anaesth 2016;63:442-53. [Crossref] [PubMed]
- Young RW. Hyperoxia: A Review of the Risks and Benefits in Adult Cardiac Surgery. J Extra Corpor Technol 2012;44:241-9. [PubMed]
- McGuinness SP, Parke R, Drummond K, et al. No A Multicenter, Randomized, Controlled Phase IIb Trial of Avoidance of Hyperoxemia during Cardiopulmonary Bypass. Anesthesiology 2016;125:465-73. [Crossref] [PubMed]
- Toraman F, Evrenkaya S, Senay S, et al. Adjusting oxygen fraction to avoid hyperoxemia during cardiopulmonary bypass. Asian Cardiovasc Thorac Ann 2007;15:303-6. [Crossref] [PubMed]
Cite this article as: Kempton H, Chong C, Vlok R, Melhuish T, Holyoak R, White L. Hyperoxemia reduces cerebrovascular accidents post coronary artery bypass graft surgery: a retrospective cohort study. J Emerg Crit Care Med 2020;4:16.