
Burden of respiratory support differs between critically ill children with severe bacterial and viral pneumonia
Introduction
Pneumonia is one of the major causes of morbidity and mortality in children (1). Approximately 120 million new cases of community-acquired pneumonia (CAP) are reported each year with 1 million deaths among children aged <5 years (1). CAP is a global problem but it is especially prevalent in South-East Asia and sub-Saharan Africa (2). Severe pneumonia accounts for a significant number of pediatric intensive care unit (PICU) admissions and remains as one of the leading reasons for mechanical ventilation (MV) (3).
Accurate attribution of the causes of pneumonia is vital in estimating burden of disease and implementation of targeted preventive or treatment strategies for better clinical outcomes in children who require MV. Several adult studies described increased mortality in patients with viral pneumonia, patients with multiple viruses and patients with secondary bacterial pneumonia (4,5). A pediatric study done in our center described a higher rate of acute respiratory distress syndrome (ARDS) in patients with viral pneumonia, attributing the difference to higher risk of direct lung injury hence oxygenation defects caused by viral pathogens (6). The same study also showed a higher rate of MV, and decreased ventilator and PICU free days in patients with co-detection of pathogens compared to those with single organism. Understanding MV requirements of patients with different primary pathogen can aid decision making in ventilation strategies for patients with severe pneumonia in PICU.
We embarked on this study to look at the difference in respiratory support requirement between children with sole viral and bacterial severe pneumonia in the subpopulation of our prior study. We hypothesized that children with viral pneumonia have higher severity of illness and higher MV requirement than children with bacterial pneumonia. The primary aim of this study is to compare respiratory support requirements of severe viral and bacterial pneumonia cases admitted to our PICU.
Methods
This is a retrospective study of all patients (1 month–18 years old) with sole bacterial or viral pneumonia admitted to PICU in KK Women’s and Children’s Hospital, Singapore, from January 2010 to December 2014. Our hospital is one of two tertiary and teaching pediatric hospitals in Singapore. The PICU is a 16-bedded multidisciplinary facility that admits general medical, oncology, general surgical, neurosurgical and cardiothoracic patients. For this study, approval was obtained from the SingHealth Centralized Institution Review Board (CIRB reference number: 201506-00083).
Patients
A patient list was generated using International Classification of Disease [ICD9CM or ICD10AM (from 2012 onwards)] or the SNOMED Clinical Terminology (SCT) code for a primary or secondary discharge diagnosis of “pneumonia” in patients admitted to the PICU. Only patients with sole viral or bacterial pathogens were included. Electronic and paper clinical records were reviewed to include patients who fulfilled criteria for severe pneumonia. We excluded patients ≤1-month old as lung disease in this group may be due to congenital factors. Patients with mixed infection were also excluded as studies had shown that this group of patients had longer hospital and PICU length of stay and longer duration of MV (4,5,7).
Data collection
Data pertaining to demographic profile, clinical outcomes, complications, microbiological investigations and types of respiratory support were collected from case notes and electronic records. We recorded MV modes and settings on day of diagnosis and every alternate day up to day 14 of PICU admission. MV parameters such as fraction of inspired oxygen (FiO2), peak inspiratory pressure (PIP), peak end expiratory pressure (PEEP), mean airway pressure (MAP) and tidal volume (TV) were collected at 06:00 hrs every day. The pediatric index of mortality 2 (PIM2) score was calculated on admission to PICU (8).
Definitions
Pneumonias were diagnosed by the presence of both clinical (i.e., febrile illness with lower respiratory tract signs) and radiological evidence. Examples of positive chest X-ray images included alveolar consolidation or pleural effusion (9,10). All radiographs were reported by in-house radiologists. Children with any general danger signs (e.g., desaturation, cyanosis, altered consciousness, convulsions) are classified as having severe pneumonia (11,12). Viral pneumonia was diagnosed by detection of a virus in respiratory fluids (e.g., nasopharyngeal aspirate, endotracheal tube aspirate or bronchoalveolar lavage) by multiplex polymerase chain reaction (PCR). Organisms tested in the multiplex PCR panel were influenza A and B, parainfluenza, respiratory syncytial virus (RSV), metapneumovirus, human coronavirus, adenovirus, Bordetella pertussis, Mycoplasma pneumoniae, Chlamydophila pneumoniae. In our center, bacterial pneumonia was diagnosed by culture of bacteria in blood, pleural fluid, endotracheal aspirates or bronchoalveolar lavage fluid (13). Streptococcal pneumonia was also diagnosed if results were positive on latex agglutination of common bacterial antigens in pleural fluid, urine streptococcal antigen or anti-streptolysin O titer (≥1/200) (14). Mycoplasma pneumoniae was diagnosed by PCR of respiratory fluids or positive blood mycoplasma serology (≥1/320) (15). Septic shock and multi-organ dysfunction were defined according to the International Pediatric Sepsis Consensus Conference (16). The definition of pediatric ARDS was based on the Pediatric Acute Lung Injury Consensus Conference (17).
MV in our unit was not protocolized at the time of data collection. In general, the most common mode of conventional MV in our center is pressure-synchronized intermittent mandatory ventilation (P-SIMV). Patients who had increased respiratory demands (i.e., increased work of breathing, hypoxia or hypercarbia) despite conventional MV would require escalation to alternative modes of MV such as airway pressure release ventilation (APRV) and high-frequency oscillatory ventilation (HFOV). Patients ventilated on at least 1 day of APRV or HFOV were considered to require alternative modes of MV. Blood gases were performed on capillary or arterial samples. The primary outcome was requirement for alternative modes of MV. Secondary outcomes were PICU mortality, length of PICU stay, length of hospital stay, changes in FiO2, PIP, PEEP and MAP.
Statistical analysis
Patients were analyzed in two groups: viral and bacterial pneumonia groups. Categorical data were expressed as counts and percentages whereas continuous data were expressed as median and interquartile ranges (IQRs). Differences between categorical data were analyzed by chi-square tests or Fisher’s exact test (when cell sizes were less than 5). Differences between continuous data were analyzed by Mann-Whitney test. We used multivariate logistic regression models to quantify the association between potential confounding factors and requirement for alternative modes of MV. All statistical tests were 2-tailed and P values of <0.050 were considered to be statistically significant. Statistical analysis was carried out using SPSS version 19 (IBM, Armonk, NY, USA).
Results
Two hundred and forty-one patients were admitted to our PICU with the diagnosis of severe pneumonia, of which 117 patients had sole etiological agent identified. Forty-nine (41.9%) and 68 (58.1%) had sole viral and bacterial infections, respectively. In our cohort, the most common viral pathogens were RSV (n=13, 11.1%), influenza A virus (n=9, 7.7%) and adenovirus (n=7, 6.0%). The most common bacterial pathogens were Streptococcus pneumoniae (n=36, 30.8%), Pseudomonas aeruginosa (n=6, 5.1%) and Klebsiella pneumoniae (n=4, 3.4%).
Characteristics and outcomes of patients with viral or bacterial pneumonia were described in Table 1. Patients with viral pneumonia were more likely to be <2 years old (51.0% vs. 27.9%, P=0.011), to have underlying comorbidities (59.2% vs. 35.3%, P=0.010) and had higher PIM2 score [3.0 (1.1, 8.0) vs. 1.6 (0.8, 3.0), P<0.001]. Pediatric ARDS was more common in children with viral pneumonia compared to those with bacterial pneumonia (22.4% vs. 0%, P<0.001). However, children with bacterial pneumonia were more likely to get complications such as septic shock (27.9% vs. 12.2%, P=0.041), necrotising pneumonia (27.9% vs. 0%, P<0.001) and pleural effusion (66.2% vs. 26.5%, P<0.001) compared to children with viral pneumonia. Children with viral pneumonia required longer length of stay in PICU compared to those with bacterial pneumonia [7.0 (3.0, 11.5) vs. 3.0 (1.0, 8.0) days, P=0.018]. Mortality rates of children with severe viral and bacterial pneumonia were not significantly different.
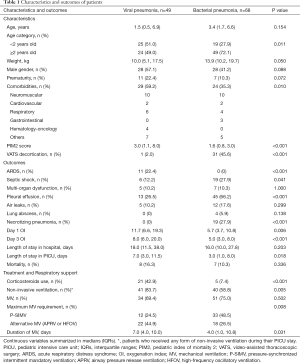
Full table
Of all 117 patients, 85 (72.6%) patients required MV during their PICU stay. Forty-five/eighty-five (52.9%) were supported on P-SIMV, 31/85 (36.5%) on APRV and 9/85 (10.6%) on HFOV. Patients with viral pneumonia required longer duration of MV [7.0 (4.0, 10.0) vs. 4.0 (1.0, 10.8) days, P=0.031] and were more likely to require alternative modes of MV compared to those with bacterial pneumonia (48.5% vs. 24.5%, P=0.008). Table 2 described the factors affecting the need for alternative MV using a multivariate regression model. The association between the need for alternative modes of MV and viral pneumonia remained significant after adjustment for age [adjusted odds ratio 3.32 (95% CI: 1.33–8.28)].
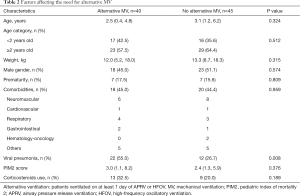
Full table
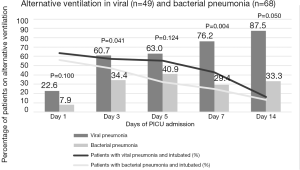
Figure 1 compared the need for alternative ventilation in the first 14 days of PICU stay. Seventeen (14.5%) patients remained intubated beyond day 14 of PICU stay. Higher percentage of intubated patients with viral pneumonia needed alternative ventilation on day 3 (60.7% vs. 34.4%, P=0.041) and day 7 of PICU stay (76.2% vs. 29.4%, P=0.004) compared to those with bacterial pneumonia. Among patients on conventional MV, there were no significant differences in ventilator settings (FiO2, MAP, PIP, PEEP and TV) between viral and bacterial pneumonia in the first 14 days of PICU stay. Oxygenation index (OI) of children with viral pneumonia was significantly higher than that of bacterial pneumonia on day 1 [OI: 11.7 (6.6, 19.3) vs. 5.7 (3.7, 10.8), P=0.006] and 3 [OI: 8.0 (6.0, 20.0) vs. 5.0 (3.0, 8.0), P<0.001] of PICU admission PICU stay. OI between the 2 groups was however not significant from day 5 to 14 of PICU stay.
All our patients were started on empirical antibiotics. The most common types of empirical antibiotics prescribed in our cohort were intravenous penicillin {e.g., ampicillin or Augmentin [68/117 (58.1%)]}, followed by intravenous cephalosporin {e.g., ceftriaxone [34/117 (29.1%)]}. Most of the patients with viral pneumonia received supportive treatment only; 2/13 (15.4%) patients with RSV pneumonia and 6/9 (66.7%) patients with influenza A pneumonia received 10-day course of intravenous ribavirin and 5-day course of oral oseltamivir, respectively. Patients with viral pneumonia were more likely to receive steroids compared to those with bacterial pneumonia (42.9% vs. 7.4%, P<0.001).
Discussion
Our study demonstrated that patients with viral pneumonia were more likely to require alternative modes and longer duration of MV. Pediatric ARDS was also more common in children with viral pneumonia. Patients with viral pneumonia required longer length of stay in PICU but there was no significant difference in overall length of stay in hospital. Despite these differences, we did not find any difference in mortality rate between patients with severe viral and bacterial pneumonia in our cohort.
Patients with viral pneumonia were more likely to require alternative modes of MV such as APRV and HFOV around day 3–7 of PICU stay compared to those with bacterial pneumonia. This requirement seemed to mirror the increased OI in patients with viral pneumonia, especially around day 3. One possible explanation of increased OI in viral infection is the increased epithelial sloughing associated with epithelial and interstitial inflammation leading to ventilation-perfusion mismatch, typically seen in children with RSV infection (18). Disruption of the alveolar-capillary barrier also impairs surfactant production via the leakage of plasma proteins into the alveoli, resulting in lung collapse (19). During our study period, escalation to APRV was deemed reasonable for patients with ARDS who did not improve with conventional MV as APRV allows spontaneous breathing with higher MAP and prevents collapse of unstable alveoli units. However, a recent RCT was terminated early as the study revealed higher mortality in children with ARDS ventilated on APRV compared to the conventional low TV ventilation (20). HFOV, as a strategy to deliver low TV at high flow rate, is thought to facilitate lung recruitment and prevent lung collapse. Studies have shown that HFOV could safely improve hypercarbia and hypoxia in children with severe ARDS (21,22). The Pediatric Acute Lung Injury Consensus Conference has recommended the use of HFOV in pediatric patients with ARDS when conventional MV fails (23). This knowledge can aid the clinical team to anticipate more severe oxygenation problems in patients with viral pneumonia and consider early escalation to alternative modes of MV.
In our study, patients with viral pneumonia had a more severe clinical course with higher rate of ARDS and greater hypoxemia [day 1 median OI: 11.7 (6.6, 19.3) vs. 5.7 (3.7, 10.8), P=0.006]. Similarly, a recent pediatric observational study reported that one-third of children with human metapneumovirus or RSV and respiratory failure developed ARDS (24). A prospective, multicenter, observational study in Spain (n=146) reported that one-fifth of patients with ARDS and requirement of MV in PICU had RSV infection, with reported mortality of approximately 15% (25). Pathologic mechanisms implicated in the development of ARDS caused by respiratory viruses are not completely understood. Reports on molecular pathology of influenza virus and coronavirus infections describe a complex interaction between viral pathogenicity and host immune response leading to endothelial injury, cytokines release and finally a common end pathway of diffuse alveolar damage (26,27). In RSV and influenza A infection, studies have reported that viral load is independently associated with increased risk of respiratory failure and need for MV (28,29). Despite higher incidence of ARDS in the viral pneumonia group, we did not find any difference in mortality rate between patients with viral and bacterial pneumonia.
A more severe clinical course induced by viral pneumonia with higher ventilatory demands, as shown in our study, have several implications. Firstly, identification of viral pathogens could potentially allow clinicians to choose specific ventilator strategies for patients. Secondly, anti-viral treatment should be considered in the management of severe viral pneumonia. In our cohort, only 2/13 (15.4%) patients with RSV pneumonia received a 10-day course of intravenous ribavirin. A meta-analysis of 12 pediatric trials reported shorter days of MV and hospitalization when aerosolized ribavirin was administered to critically ill children who had RSV infection and required MV (30). In contrary to aerosolized ribavirin, there is a paucity of studies on the safety and use of intravenous ribavirin in the pediatric population. A case series of 6 pediatric hematopoietic stem cell transplant recipients who received intravenous ribavirin for RSV infection showed resolution of infection without associated side effects (31). In our center, ribavirin was only available in the intravenous form and its use was limited for immunocompromised patients. Six/nine (66.7%) patients with influenza A pneumonia received a 5 days course of oral oseltamivir. Studies have shown improved survival, shorter days of MV and length of stay in hospital in patients treated with oseltamivir, especially when initiated in the first 4 days of illness (32,33). Future prospective controlled studies are needed to evaluate the long-term efficacy of anti-viral treatment in the pediatric population.
Besides antiviral medications, clinicians should also be mindful of potential impact of other pharmacological treatments in modulating illness severity and need for MV in children with severe pneumonia. In our institution, all our patients were started on empirical antibiotics. Several studies have shown that empirical antibiotic therapy was associated with lower intensive care unit (ICU) mortality, especially in patients with septic shock and who required MV (34,35). Our study also showed that patients with viral pneumonia were likely to have received corticosteroids compared to those with bacterial pneumonia (42.9% vs. 7.4%, P<0.001). A recent meta-analysis (n=528) showed that adjunctive corticosteroids significantly reduced mortality and need for MV in adult patients with severe pneumonia (36).
The main strength of the study is the complete identification of all patients with severe pneumonia who required PICU admission. The list of patients was generated from diagnostic codes as well as via a manual search. Accuracy was enhanced by reviewing clinical and radiological findings from charts. The main limitation of our study is the correct classification of the infective etiologies. Both viral and bacterial microbiological diagnostic tests have their limitations (13-15). As such, we expect certain degree of misclassification in our study. Other limitations include the small retrospective and single-centered nature of the study. In addition, we did not collect data pertaining to socioeconomic status and vaccination status which might affect severity of pneumonia.
In conclusion, our study showed patients with viral and bacterial pneumonia have varying needs for respiratory support. Critically ill children with viral pneumonia were more likely to require alternative modes of ventilation and longer duration of MV. Although the clinical course of severe viral and bacterial pneumonia was different, there was no difference in mortality between the two groups.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethics Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. For this study, approval was obtained from the SingHealth Centralized Institution Review Board (CIRB reference number: 201506-00083).
References
- Liu L, Johnson HL, Cousens S, et al. Global, regional, and national causes of child mortality: an updated systematic analysis for 2010 with time trends since 2000. Lancet 2012;379:2151-61. [Crossref] [PubMed]
- Walker CLF, Rudan I, Liu L, et al. Global burden of childhood pneumonia and diarrhoea. Lancet 2013;381:1405-16. [Crossref] [PubMed]
- Rudan I, O’Brien KL, Nair H, et al. Epidemiology and etiology of childhood pneumonia in 2010: estimates of incidence, severe morbidity, mortality, underlying risk factors and causative pathogens for 192 countries. J Glob Health 2013;3:010401. [PubMed]
- Crotty MP, Meyers S, Hampton N, et al. Epidemiology, co-infections, and outcomes of viral pneumonia in adults: an observational cohort study. Medicine (Baltimore) 2015;94:e2332. [Crossref] [PubMed]
- van der Sluijs KF, van der Poll T, Lutter R, et al. Bench-to-bedside review: bacterial pneumonia with influenza - pathogenesis and clinical implications. Crit Care 2010;14:219. [Crossref] [PubMed]
- Koh JWJC, Wong JJ, Sultana R, et al. Risk factors for mortality in children with pneumonia admitted to the pediatric intensive care unit. Pediatr Pulmonol 2017;52:1076-84. [Crossref] [PubMed]
- Voiriot G, Visseaux B, Cohen J, et al. Viral-bacterial coinfection affects the presentation and alters the prognosis of severe community-acquired pneumonia. Crit Care 2016;20:375. [Crossref] [PubMed]
- Slater A, Shann F, Pearson G, et al. PIM2: a revised version of the paediatric index of mortality. Intensive Care Med 2003;29:278-85. [Crossref] [PubMed]
- Cherian T, Mulholland EK, Carlin JB, et al. Standardized interpretation of paediatric chest radiographs for the diagnosis of pneumonia in epidemiological studies. Bull World Health Organ 2005;83:353-9. [PubMed]
- Fancourt N, Deloria Knoll M, Barger-Kamate B, et al. Standardized interpretation of chest radiographs in cases of pediatric pneumonia from the PERCH study. Clin Infect Dis 2017;64:S253-61. [Crossref] [PubMed]
- World Health Organization. Chart booklet: integrated management of childhood illness. Geneva: World Health Organization, 2014.
- World Health Organization. WHO recommendations on the management of diarrhoea and pneumonia in HIV-infected infants and children: integrated management of childhood illness (IMCI). Geneva: World Health Organization, 2010.
- Stein RT, Marostica PJ. Community-acquired pneumonia: a review and recent advances. Pediatr Pulmonol 2007;42:1095-103. [Crossref] [PubMed]
- Ferretti JJ, Stevens DL, Fischetti VA. Streptococcus pyogenes: Basic Biology to Clinical Manifestations. Oklahoma City: University of Oklahoma Health Sciences Center, 2016.
- Saraya T. Mycoplasma pneumoniae infection: basics. J Gen Fam Med 2017;18:118-25. [Crossref] [PubMed]
- Goldstein B, Giroir B, Randolph A, et al. International pediatric sepsis consensus conference: definitions for sepsis and organ dysfunction in pediatrics. Pediatr Crit Care Med 2005;6:2-8. [Crossref] [PubMed]
- Medina A, Modesto I, Alapont V. Del Villar-Guerra P. PALICC definition of ARDS. Don't remove that brick from the wall and keep it smart and simple. Med Intensiva 2016;40:311-4. [Crossref] [PubMed]
- Johnson JE, Gonzales RA, Olson SJ, et al. The histopathology of fatal untreated human respiratory syncytial virus infection. Mod Pathol 2007;20:108-19. [Crossref] [PubMed]
- Nelson JK. Surfactant abnormalities in infants with severe viral bronchiolitis. Arch Dis Child Fetal Neonatal Ed 1997;77:F78. [Crossref] [PubMed]
- Lalgudi Ganesan S, Jayashree M, Chandra Singhi S, et al. Airway pressure release ventilation in pediatric acute respiratory distress syndrome. A randomized controlled trial. Am J Respir Crit Care Med 2018;198:1199-207. [Crossref] [PubMed]
- Moniz M, Silvestre C, Nunes P, et al. High-frequency oscillatory ventilation in children: a 10-year experience. J Pediatr (Rio J) 2013;89:48-55. [Crossref] [PubMed]
- Babbitt CJ, Cooper MC, Nussbaum E, et al. High-frequency oscillatory ventilation in pediatric acute hypoxemic respiratory failure: disease-specific morbidity survival analysis. Lung 2012;190:685-90. [Crossref] [PubMed]
- Pediatric Acute Lung Injury Consensus Conference Group. Pediatric acute respiratory distress syndrome: consensus recommendations from the Pediatric Acute Lung Injury Consensus Conference. Pediatr Crit Care Med 2015;16:428-39. [Crossref] [PubMed]
- Ravindranath TM, Gomez A, Harwayne-Gidansky I, et al. Pediatric acute respiratory distress syndrome associated with human metapneumovirus and respiratory syncytial virus. Pediatr Pulmonol 2018;53:929-35. [Crossref] [PubMed]
- López-Fernández Y, Azagra AM, de la Oliva P, et al. Pediatric acute lung injury epidemiology and natural history study: Incidence and outcome of the acute respiratory distress syndrome in children. Crit Care Med 2012;40:3238-45. [Crossref] [PubMed]
- Howard WA, Peiris M, Hayden FG. Report of the 'mechanisms of lung injury and immunomodulator interventions in influenza' workshop, 21 March 2010, Ventura, California, USA. Influenza Other Respir Viruses 2011;5:453-4, e458-75.
- Gralinski LE, Baric RS. Molecular pathology of emerging coronavirus infections. J Pathol 2015;235:185-95. [Crossref] [PubMed]
- Houben ML, Coenjaerts FE, Rossen JW, et al. Disease severity and viral load are correlated in infants with primary respiratory syncytial virus infection in the community. J Med Virol 2010;82:1266-71. [Crossref] [PubMed]
- Launes C, Garcia-Garcia JJ, Jordan I, et al. Viral load at diagnosis and influenza A H1N1 (2009) disease severity in children. Influenza Other Respir Viruses 2012;6:e89-92. [Crossref] [PubMed]
- Ventre K, Randolph AG. Ribavirin for respiratory syncytial virus infection of the lower respiratory tract in infants and young children. Cochrane Database Syst Rev 2007.CD000181. [PubMed]
- Molinos-Quintana A, Pérez-de Soto C, Gómez-Rosa M, et al. Intravenous ribavirin for respiratory syncytial viral infections in pediatric hematopoietic SCT recipients. Bone Marrow Transplant 2013;48:265-8. [Crossref] [PubMed]
- McGeer A, Green KA, Plevneshi A, et al. Antiviral therapy and outcomes of influenza requiring hospitalization in Ontario, Canada. Clin Infect Dis 2007;45:1568-75. [Crossref] [PubMed]
- Lee N, Cockram CS, Chan PK, et al. Antiviral treatment for patients hospitalized with severe influenza infection may affect clinical outcomes. Clin Infect Dis 2008;46:1323-4. [Crossref] [PubMed]
- Martin-Loeches I, Lisboa T, Rodriguez A, et al. Combination antibiotic therapy with macrolides improves survival in intubated patients with community-acquired pneumonia. Intensive Care Med 2010;36:612-20. [Crossref] [PubMed]
- Rodríguez A, Mendia A, Sirvent JM, et al. Combination antibiotic therapy improves survival in patients with community-acquired pneumonia and shock. Crit Care Med 2007;35:1493-8. [Crossref] [PubMed]
- Bi J, Yang J, Wang Y, et al. Efficacy and safety of adjunctive corticosteroids therapy for severe community-acquired pneumonia in adults: an updated systematic review and meta-analysis. PLoS One 2016;11:e0165942. [Crossref] [PubMed]
Cite this article as: Loh SW, Wong JJ, Teoh OH, Koh JW, Mok YH, Lee JH. Burden of respiratory support differs between critically ill children with severe bacterial and viral pneumonia. J Emerg Crit Care Med 2020;4:11.


