
Perioperative hypotension: clinical impact, diagnosis, and therapeutic approaches
Introduction
Postoperative mortality is a major healthcare problem. Death within a month after surgery would be the third leading cause of death worldwide if it would be considered a disease of its own (1). The EUSOS study (2) showed that overall in-hospital mortality after non-cardiac surgery in Europe is about 4%. In the United States, the 30-day mortality after surgery is roughly 2% (3). Death after surgery occurs as a consequence of postoperative complications (3,4). In this context, it has been shown that myocardial injury, acute kidney injury, stroke, congestive heart failure, sepsis, major bleeding, and new-onset atrial fibrillation are independently associated with death within the first 30 days after non-cardiac surgery (3). Therefore, in order to decrease postoperative mortality, avoiding postoperative complications is key. In turn, to avoid postoperative complications, modifiable risk factors independently associated with these complications need to be identified and avoided. Only then, strategies can be implemented in the clinical routine to decrease postoperative mortality and adverse outcomes after surgery. One of the modifiable risk factors for postoperative complications may be perioperative hypotension—i.e., low blood pressure in the perioperative period. Perioperative hypotension can be divided into different phases depending on when in the perioperative period hypotension occurs. While intraoperative hypotension (IOH) describes a low blood pressure during surgery, postoperative hypotension (POH) refers to hypotension occurring on the remaining day of surgery (after the emergence from anesthesia) and during the first days after surgery. During the last years, many studies focused on the association of IOH and postoperative outcomes, but POH only recently received more attention. POH is a challenge for clinicians because—in sharp contrast to the intraoperative period—patient surveillance and hemodynamic monitoring in patients treated on the general care ward after surgery is very basic and makes detection of hypotension difficult.
In this review, we discuss perioperative hypotension, specifically focusing on POH and its impact on postoperative patient outcomes, challenges related to its diagnosis, and potential therapeutic approaches.
IOH
During the last years, the majority of studies on perioperative blood pressure physiology and management focused on IOH. It has been shown that IOH is common in patients having non-cardiac surgery under general anesthesia (5). Retrospective cohort analysis revealed that IOH is associated with acute kidney injury (6,7), myocardial injury [myocardial injury after non-cardiac surgery (MINS)] (6-8), and death (9-12). The relationship between IOH and serious postoperative complications is supported by many observational analyses, but only a single randomized trial. This randomized controlled trial (INPRESS study) (13) compared individualized with standard blood pressure management in 292 adults having major surgery. The individual intraoperative blood pressure target was defined as a relative systolic blood pressure threshold considering a single preoperative baseline measurement of the patients’ resting blood pressure. Targeting an individualized systolic blood pressure reduced the risk of postoperative organ dysfunction compared with standard management. However, the INPRESS trial has several major limitations that limit the internal validity of the trial. First, the trial not only compared two different strategies for blood pressure management but also used two different vasopressors (norepinephrine vs. ephedrine) in the study and the control group (14,15). These co-interventions may be responsible for the differences in the incidence of the primary endpoint in the groups (14,15). Second, the definition of the individual baseline resting systolic blood pressure was based on a single non-standardized blood pressure measurement. The authors used the resting blood pressure from the preoperative anesthesiology consultation or the blood pressure measurement recorded by a nurse of the surgical ward the day before surgery as the reference value. However, the individual blood pressure profile of a patient is not adequately reflected by a single blood pressure measurement performed in the preoperative evaluation clinic (16) or directly before induction of general anesthesia (17,18). Therefore, an international consensus group recently defined automated ambulatory blood pressure monitoring as “the optimal method to establish baseline values” before surgery (19). Despite these limitations, the INPRESS trial is highly important because it provides evidence that there is not only an association but also a causal relationship between intraoperative blood pressure and patient-centered postoperative outcomes.
Nevertheless, to date, it remains unclear which blood pressure value should be targeted in the individual patient during surgery to avoid physiologically relevant IOH (20). Based on the results of retrospective database studies, an absolute mean arterial pressure threshold of 65 mmHg is often recommended as a lower “one-size-fits-all” population-derived intervention threshold (7,20). However, by no means it should be assumed that the population harm threshold of 65 mmHg constitutes the individual patient’s optimal intraoperative blood pressure. Using an absolute mean arterial pressure threshold of 65 mmHg for every patient would ignore that blood pressure regulation depends on complex autoregulatory mechanisms, circadian rhythms, and neural/hormonal changes (19) and that, therefore, normal blood pressure varies considerably among individuals (17). However, more randomized controlled trials are needed before recommendations can be given on how to individualize intraoperative blood pressure targets in clinical routine.
POH
Perioperative hypotension associated with organ injury is not restricted to the intraoperative period but also occurs after surgery as POH (12). However, data on the incidence of POH and its impact on patient outcomes are scarce.
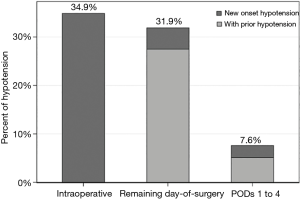
A secondary analysis of the large prospective POISE-2 trial, including 9,765 patients, investigated the effects of hypotension during different perioperative periods including the intraoperative period, the remaining day of surgery, and the first four postoperative days after surgery (12). The primary outcome was a composite of myocardial infarction or death within 30 days after surgery. Hypotension was defined as a systolic blood pressure of less than 90 mmHg requiring treatment. IOH according to this definition occurred in about one-third of patients. The incidence of POH on the remaining day of surgery was also more than 30% (Figure 1). POH on postoperative days 1 to 4 was 7.6%. Interestingly, there were patients with POH who were not hypotensive during surgery. Not only IOH but also POH occurring on the remaining day of surgery and within the first four days after surgery was significantly associated with an increased risk of the primary outcome.
A secondary analysis of a prospective cohort including 2,211 patients after moderate or major non-cardiac surgery investigated the association between POH and MINS (21). Patients were stratified according to quartiles based on the lowest mean arterial pressure preceding the peak of the high-sensitivity troponin T elevation. Multivariate regression analysis revealed that lower mean arterial pressures were associated with an increasing risk of high-sensitivity troponin T elevation.
A retrospective cohort study including 2,833 postoperative patients who were admitted to the intensive care unit after surgery investigated the association between blood pressure and acute kidney injury as well as a composite of myocardial injury and mortality (22). POH was quantified as the lowest mean arterial pressure on each intensive care unit day. The risk of acute kidney injury increased with decreasing minimum daily mean arterial pressures (over a range from 50 to 110 mmHg) (22). In addition, POH was associated with the composite outcome of myocardial injury or death, with estimated risk increasing at lower mean arterial pressures (22). The risk of this composite outcome was more than 20% higher at the 25th percentile of lowest mean arterial pressure (78 mmHg) compared with at the median of lowest mean arterial pressure (87 mmHg). Thus, increasing amounts of POH seem to be associated with acute kidney injury and a composite of myocardial injury and death in postoperative intensive care unit patients.
In summary, considering the results of the few available studies on POH, there seems to be an association between POH and adverse postoperative outcomes in terms of MINS, acute kidney injury, and postoperative mortality. However, there are several crucial open questions regarding the pathophysiology, impact, and management of POH.
It is noteworthy that the thresholds of harm for hypotension during the postoperative period may be higher than during the intraoperative period. Patients after surgery and emergence from general anesthesia may need different—presumably higher—organ perfusion pressures than patients during surgery with general anesthesia because general anesthesia represents a state of reduced metabolic activity. Further research is needed to accurately define thresholds for physiologically relevant POH.
The early recognition and timely treatment of POH are especially challenging. Patient surveillance is highly standardized in the operating room during surgery and includes continuous basic and advanced hemodynamic monitoring. In addition, during surgery, highly trained medical personal is closely monitoring the patient all of the time. While continuous hemodynamic monitoring is also standard of care in post-anesthesia care units, monitoring of postoperative patients on general care wards is intermittent and usually only consists of spot checks of vital signs by a nurse every 4–8 hours.
A prospective observational study including 312 patients recovering from abdominal surgery on the general care ward investigated the incidence, severity, and detection of postoperative blood pressure perturbations (23). The study demonstrates that POH defined as a mean arterial pressure of less than 65 mmHg for at least 15 minutes occurred in about 20% of the patients. POH remained unnoticed by intermittent vital sign checks in about half of the cases.
Therefore, there is increasing interest in continuous ward monitoring that may allow detecting POH earlier (24-28). Continuous ward monitoring may enable alterations in vital signs to be recognized in real-time and thus allow timely identification of POH. Typically, patients admitted to the general care ward do not deteriorate all of a sudden; subtle changes and abnormalities in vital signs usually precede adverse events (29,30). These subtle changes include changes in blood pressure that may be detected earlier when using continuous ward monitoring instead of intermittent spot checks. There is increasing evidence that the implementation of ward monitoring on general care wards can improve patient-centered outcomes and reduce rates of rescue events and intensive care unit transfers (31-33). Although, numerous wireless and wearable sensors for ward monitoring are already available (34) there is still the need for technological refinements regarding the signal quality and measurement performance (24-28). Especially the continuous measurement of blood pressure on general care wards is challenging, as reliable portable monitoring systems are missing, and refinements in sensor technology are needed (35). Finally, strategies to prevent or treat POH based on continuous ward monitoring need to be tested for their effectiveness to improve quality of care or patient-centered outcomes in large-scale interventional trials.
Conclusions
Perioperative hypotension includes IOH and POH. IOH is common in patients having non-cardiac surgery under general anesthesia (5) and is associated with acute kidney injury (6,7), myocardial injury (6-8), and death (9-12). The relationship between IOH and serious postoperative complications is supported by many observational analyses and one randomized trial (INPRESS study) (13) that suggests that individualized blood pressure management reduces the risk of postoperative organ dysfunction compared with usual care. More randomized controlled trials are needed before recommendations can be given on how to individualize intraoperative blood pressure targets in clinical routine.
POH refers to hypotension occurring on the remaining day of surgery and during the first days after surgery. Available data suggest that POH after non-cardiac surgery is common, profound, and largely undetected by current vital sign monitoring on the general care ward. There is evidence that POH is associated with adverse postoperative outcomes, especially MINS, acute kidney injury, and death. However, there is a need for more data on the pathophysiology, impact, and management of POH. Continuous ward monitoring might enable POH to be detected and treated in a timely manner (24,28). However, strategies to prevent or treat POH based on continuous ward monitoring need to be tested for their effectiveness to improve quality of care or patient-centered outcomes in large-scale interventional trials.
Acknowledgments
Support was provided only from institutional and/or departmental funds.
Footnote
Conflicts of Interest: Phillip Hoppe and Karim Kouz have no conflicts of interest to declare. Bernd Saugel has received honoraria for consulting, honoraria for giving lectures, and refunds of travel expenses from Edwards Lifesciences Inc. (Irvine, CA, USA). Bernd Saugel has received honoraria for consulting, institutional restricted research grants, honoraria for giving lectures, and refunds of travel expenses from Pulsion Medical Systems SE (Feldkirchen, Germany). Bernd Saugel has received institutional restricted research grants, honoraria for giving lectures, and refunds of travel expenses from CNSystems Medizintechnik GmbH (Graz, Austria). Bernd Saugel has received institutional restricted research grants from Retia Medical LLC. (Valhalla, NY, USA). Bernd Saugel has received honoraria for giving lectures from Philips Medizin Systeme Böblingen GmbH (Böblingen, Germany). Bernd Saugel has received honoraria for consulting, institutional restricted research grants, and refunds of travel expenses from Tensys Medical Inc. (San Diego, CA, USA).
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Nepogodiev D, Martin J, Biccard B, et al. Global burden of postoperative death. Lancet 2019;393:401. [Crossref] [PubMed]
- Pearse RM, Moreno RP, Bauer P, et al. Mortality after surgery in Europe: a 7 day cohort study. Lancet 2012;380:1059-65. [Crossref] [PubMed]
- Vascular Events in Noncardiac Surgery Patients Cohort Evaluation (VISION) Study Investigators. Association between complications and death within 30 days after noncardiac surgery. CMAJ 2019;191:E830-7. [Crossref] [PubMed]
- Fecho K, Lunney AT, Boysen PG, et al. Postoperative mortality after inpatient surgery: Incidence and risk factors. Ther Clin Risk Manag 2008;4:681-8. [Crossref] [PubMed]
- Bijker JB, van Klei WA, Kappen TH, et al. Incidence of intraoperative hypotension as a function of the chosen definition: literature definitions applied to a retrospective cohort using automated data collection. Anesthesiology 2007;107:213-20. [Crossref] [PubMed]
- Walsh M, Devereaux PJ, Garg AX, et al. Relationship between intraoperative mean arterial pressure and clinical outcomes after noncardiac surgery: toward an empirical definition of hypotension. Anesthesiology 2013;119:507-15. [Crossref] [PubMed]
- Salmasi V, Maheshwari K, Yang D, et al. Relationship between intraoperative hypotension, defined by either reduction from baseline or absolute thresholds, and acute kidney and myocardial injury after noncardiac surgery: a retrospective cohort analysis. Anesthesiology 2017;126:47-65. [Crossref] [PubMed]
- Sessler DI, Khanna AK. Perioperative myocardial injury and the contribution of hypotension. Intensive Care Med 2018;44:811-22. [Crossref] [PubMed]
- Monk TG, Bronsert MR, Henderson WG, et al. Association between intraoperative hypotension and hypertension and 30-day postoperative mortality in noncardiac surgery. Anesthesiology 2015;123:307-19. [Crossref] [PubMed]
- Mascha EJ, Yang D, Weiss S, et al. Intraoperative mean arterial pressure variability and 30-day mortality in patients having noncardiac surgery. Anesthesiology 2015;123:79-91. [Crossref] [PubMed]
- Stapelfeldt WH, Yuan H, Dryden JK, et al. The SLUScore: a novel method for detecting hazardous hypotension in adult patients undergoing noncardiac surgical procedures. Anesth Analg 2017;124:1135-52. [Crossref] [PubMed]
- Sessler DI, Meyhoff CS, Zimmerman NM, et al. Period-dependent associations between hypotension during and for four days after noncardiac surgery and a composite of myocardial infarction and death: a substudy of the POISE-2 trial. Anesthesiology 2018;128:317-27. [Crossref] [PubMed]
- Futier E, Lefrant JY, Guinot PG, et al. Effect of individualized vs standard blood pressure management strategies on postoperative organ dysfunction among high-risk patients undergoing major surgery: a randomized clinical trial. JAMA 2017;318:1346-57. [Crossref] [PubMed]
- Daoud M. Organ dysfunction after surgery in patients treated with individualized or standard blood pressure management. JAMA 2018;319:720-1. [Crossref] [PubMed]
- Mitchell K, Adams D, McHugh SM. Organ dysfunction after surgery in patients treated with individualized or standard blood pressure management. JAMA 2018;319:719-20. [Crossref] [PubMed]
- Kallioinen N, Hill A, Horswill MS, et al. Sources of inaccuracy in the measurement of adult patients' resting blood pressure in clinical settings: a systematic review. J Hypertens 2017;35:421-41. [Crossref] [PubMed]
- Saugel B, Reese PC, Sessler DI, et al. Automated ambulatory blood pressure measurements and intraoperative hypotension in patients having noncardiac surgery with general anesthesia: a prospective observational study. Anesthesiology 2019;131:74-83. [Crossref] [PubMed]
- van Klei WA, van Waes JA, Pasma W, et al. Relationship between preoperative evaluation blood pressure and preinduction blood pressure: a cohort study in patients undergoing general anesthesia. Anesth Analg 2017;124:431-7. [Crossref] [PubMed]
- Ackland GL, Brudney CS, Cecconi M, et al. Perioperative Quality Initiative consensus statement on the physiology of arterial blood pressure control in perioperative medicine. Br J Anaesth 2019;122:542-51. [Crossref] [PubMed]
- Sessler DI, Bloomstone JA, Aronson S, et al. Perioperative Quality Initiative consensus statement on intraoperative blood pressure, risk and outcomes for elective surgery. Br J Anaesth 2019;122:563-74. [Crossref] [PubMed]
- van Lier F, Wesdorp FHIM, Liem VGB, et al. Association between postoperative mean arterial blood pressure and myocardial injury after noncardiac surgery. Br J Anaesth 2018;120:77-83. [Crossref] [PubMed]
- Khanna AK, Maheshwari K, Mao G, et al. Association between mean arterial pressure and acute kidney injury and a composite of myocardial injury and mortality in postoperative critically ill patients: a retrospective cohort analysis. Crit Care Med 2019;47:910-7. [Crossref] [PubMed]
- Turan A, Chang C, Cohen B, et al. Incidence, severity, and detection of blood pressure perturbations after abdominal surgery: a prospective blinded observational study. Anesthesiology 2019;130:550-9. [Crossref] [PubMed]
- McGillion MH, Duceppe E, Allan K, et al. Postoperative remote automated monitoring: need for and state of the science. Can J Cardiol 2018;34:850-62. [Crossref] [PubMed]
- Michard F, Sessler DI. Ward monitoring 3.0. Br J Anaesth 2018;121:999-1001. [Crossref] [PubMed]
- Michard F, Bellomo R, Taenzer A. The rise of ward monitoring: opportunities and challenges for critical care specialists. Intensive Care Med 2019;45:671-3. [Crossref] [PubMed]
- Sessler DI, Saugel B. Beyond 'failure to rescue': the time has come for continuous ward monitoring. Br J Anaesth 2019;122:304-6. [Crossref] [PubMed]
- Khanna AK, Hoppe P, Saugel B. Automated continuous noninvasive ward monitoring: future directions and challenges. Crit Care 2019;23:194. [Crossref] [PubMed]
- Chen L, Ogundele O, Clermont G, et al. Dynamic and personalized risk forecast in step-down units. implications for monitoring paradigms. Ann Am Thorac Soc 2017;14:384-91. [Crossref] [PubMed]
- Jones D, Mitchell I, Hillman K, et al. Defining clinical deterioration. Resuscitation 2013;84:1029-34. [Crossref] [PubMed]
- Bellomo R, Ackerman M, Bailey M, et al. A controlled trial of electronic automated advisory vital signs monitoring in general hospital wards. Crit Care Med 2012;40:2349-61. [Crossref] [PubMed]
- Subbe CP, Duller B, Bellomo R. Effect of an automated notification system for deteriorating ward patients on clinical outcomes. Crit Care 2017;21:52. [Crossref] [PubMed]
- Taenzer AH, Pyke JB, McGrath SP, et al. Impact of pulse oximetry surveillance on rescue events and intensive care unit transfers: a before-and-after concurrence study. Anesthesiology 2010;112:282-7. [Crossref] [PubMed]
- Michard F, Bellomo R, Taenzer A. The rise of ward monitoring: opportunities and challenges for critical care specialists. Intensive Care Med 2019;45:671-3. [Crossref] [PubMed]
- Michard F, Sessler DI, Saugel B. Non-invasive arterial pressure monitoring revisited. Intensive Care Med 2018;44:2213-5. [Crossref] [PubMed]
Cite this article as: Hoppe P, Kouz K, Saugel B. Perioperative hypotension: clinical impact, diagnosis, and therapeutic approaches. J Emerg Crit Care Med 2020;4:8.


