
“Blow with the high flow” an updated algorithm
An algorithm of nasal high flow (NHF) application in patients with acute hypoxemic respiratory failure was proposed by our clinic a few years ago (1). Since then, a number of recent studies have been published, thus the aim of the present letter is to provide an update to the already published algorithm regarding the optimum initiation flow rate, the introduction of indices of sufficient prognostic value for monitoring the patient, as well as the suggestion of a new strategy to intubate after NHF failure (Figure 1).
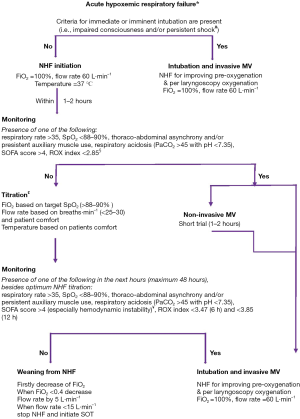
As previously described, the first concern in a patient with clinical signs of acute respiratory distress of almost any cause and hypoxemic respiratory failure detected by arterial blood gas analysis is to check if endotracheal intubation and invasive ventilation are indicated. If so, intubation must not be delayed and NHF could be used for pre-oxygenation and apneic oxygenation during laryngoscopy with maximum settings [flow rate 60 L·min–1, 100% fraction of inspired oxygen (FiO2)] (2,3). Nasal application of high flow oxygen during intubation extends apnoeic window by maintaining oxygen saturation within safer values (>90%) for a longer period with concomitant lesser rate of carbon dioxide rise (0.15 kPa·min–1) (3). It has been demonstrated that NHF minimizes intubation related adverse events such as rate and level of desaturation, arrhythmia and cardiac arrest.
If endotracheal intubation and invasive ventilation are not indicated, NHF must be applied as soon as possible with 100% FiO2, flow rate 50–60 L·min–1 and temperature 37 °C. The best initially set flow rate remains a matter of debate. Lower flow rates (35–40 L·min–1) are better tolerated (4), while a maximum flow of 60 L·min–1 can rapidly relieve dyspnea, improve oxygenation and prevent muscle fatigue (5). A recent physiologic randomized study in patients with hypoxemic respiratory failure showed that the optimum flow rate can be different depending on the respiratory target (i.e., oxygenation index, minute ventilation, work of breathing, etc.) (6). Hence, the authors concluded that it should be individualized. However, in clinical practice, it seems more reasonable to select the highest flow (60 L·min–1) for NHF initiation in order to achieve rapid improvement and then titrate according to the therapeutic targets and patient’s comfort (6). Therefore, we still recommend the application of high flow rates at the initial settings.
Soon after NHF initiation (within 1–2 h), negative prognostic respiratory parameters, such as pulse oximetry of oxygen (SpO2) <88–90%, respiratory rate >35 breaths·min–1, thoraco-abdominal asynchrony and auxiliary respiratory muscle use must be checked (7). The aforementioned parameters should improve early after NHF initiation and thus if any of these are present, they identify patients that do not respond to NHF. Based on the most significant prognostic indices, respiratory rate and SpO2, respiratory rate-oxygenation (ROX) index has been proposed recently as the ratio of SpO2/FiO2 to respiratory rate (8). It has been demonstrated that ROX index <2.85 at 2 hours after NHF initiation is a predictor of failure thus NHF must be discontinued. In that case non-invasive ventilation (NIV) application could be considered. Beyond respiratory parameters, the presence of additional organ dysfunction as expressed by a SOFA score >4, mainly hemodynamic failure, has also been significantly associated with NHF failure, indicating a more severe group of patients (9).
If negative prognostic respiratory parameters are not met, NHF can be continued. Initial settings must be adjusted according to patient’s respiratory rate (<25–30 breaths·min–1), SpO2 (>88–90%), comfort and tolerance. Flow rate should be decreased by 5–10 L·min–1 every 1–2 h. However, if flow rate is less than 60 L·min–1 and respiratory targets of SpO2 and respiratory rate are not achieved, an increase of flow rate by 5–10 L·min–1 is suggested instead of raising FiO2, since entrainment of room air during inspiration will decrease, leading to a more reliable FiO2. Higher flow rates will also linearly increase airway pressure, recruiting more alveolar units. If SpO2 remains low, an increase in FiO2 is necessary (4).
The abovementioned prognostic respiratory parameters and the presence of organ dysfunction, especially hemodynamic instability and the ROX index (cut-of values for NHF failure at 6 and 12 hours are 3.47 and 3.85 respectively) must be ideally monitored every 1–2 hours (7-9). Sustaining NHF for respiratory support in an unresponsive patient can result in undesired respiratory and cardiac complications. Kang et al. , suggested that intubating a patient who had failed on NHF therapy within 48 h of starting NHF is associated with lower overall intensive care unit (ICU) mortality compared to those who were intubated after 48 h (10). However, we recommend that instead of a specific time frame after NHF initiation as a criterion for early or late intubation, the presence of negative prognostic indices and the inability to reverse them within 1 or 2 hours after NHF settings titration should be considered as more accurate. In such a case, invasive mechanical ventilation (MV) should be initiated and NHF can be maintained throughout the intubation period to improve pre-oxygenation and apneic oxygenation.
If the patient’s clinical status and arterial blood gases gradually improve, firstly FiO2 must be decreased to 40–50%, followed by a stepwise decrease in flow rate by 5–10 L·min–1 with intervals based on the patient’s respiratory parameters. NHF can be weaned off and delivery of oxygen with a venturi mask or low flow nasal prongs can be started, if the patient remains stable for 1–2 h with FiO2 40% and flow rate <15 L·min–1.
Despite the abundant literature up to date regarding NHF use in hypoxemic respiratory failure, more studies are needed in order to build specific algorithms based on the unique pathophysiological mechanism of each underlying disease. However, since no other algorithm exists in the current literature, we propose the use of our revised algorithm by clinicians.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Ischaki E, Pantazopoulos I, Zakynthinos S. Nasal high flow therapy: a novel treatment rather than a more expensive oxygen device. Eur Respir Rev 2017. [Crossref] [PubMed]
- Miguel-Montanes R, Hajage D, Messika J, et al. Use of high-flow nasal cannula oxygen therapy to prevent desaturation during tracheal intubation of intensive care patients with mild-to-moderate hypoxemia. Crit Care Med 2015;43:574-83. [Crossref] [PubMed]
- Patel A, Nouraei SA. Transnasal humidified rapid-insufflation ventilatory exchange (THRIVE): a physiological method of increasing apnoea time in patients with difficult airways. Anaesthesia 2015;70:323-9. [Crossref] [PubMed]
- Spoletini G, Alotaibi M, Blasi F, et al. Heated humidified high-flow nasal oxygen in adults: mechanisms of action and clinical implications. Chest 2015;148:253-61. [Crossref] [PubMed]
- Frat JP, Thille AW, Mercat A, et al. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med 2015;372:2185-96. [Crossref] [PubMed]
- Mauri T, Alban L, Turrini C, et al. Optimum support by high-flow nasal cannula in acute hypoxemic respiratory failure: effects of increasing flow rates. Intensive Care Med 2017;43:1453-63. [Crossref] [PubMed]
- Sztrymf B, Messika J, Bertrand F, et al. Beneficial effects of humidified high flow nasal oxygen in critical care patients: a prospective pilot study. Intensive Care Med 2011;37:1780-6. [Crossref] [PubMed]
- Roca O, Caralt B, Messika J, et al. An index combining respiratory rate and oxygenation to predict outcome of nasal high-flow therapy. Am J Respir Crit Care Med 2019;199:1368-76. [Crossref] [PubMed]
- Kim WY, Sung H, Hong SB, et al. Predictors of high flow nasal cannula failure in immunocompromised patients with acute respiratory failure due to non-HIV pneumocystis pneumonia. J Thorac Dis 2017;9:3013-22. [Crossref] [PubMed]
- Kang BJ, Koh Y, Lim CM, et al. Failure of high-flow nasal cannula therapy may delay intubation and increase mortality. Intensive Care Med 2015;41:623-32. [Crossref] [PubMed]
Cite this article as: Ischaki E, Pantazopoulos I. “Blow with the high flow” an updated algorithm. J Emerg Crit Care Med 2019;3:61.


