
Diagnosis of invasive pulmonary aspergillosis in the intensive care unit: what we should concern and how to do better
Background
Aspergillus is a large sort of saprophytic fungi which are present almost everywhere in the environment and more azole-resistant Aspergillus fumigatus have been detected in intensive care unit (ICU) patients these years (1-4). Invasive pulmonary aspergillosis (IPA) may lead to fatal illness if timely diagnosis and appropriate management is not done. What is more, it cannot be denied that IPA has been on the rise in the ICU during the last two decades due to the increased number of immunocompromised hosts admitted and the application of invasive treatment procedures (5,6). However, data regarding the incidence of IPA in the ICU is limited and IPA diagnosis accurately is a quite challenging job, because of the non-specific symptoms or signs, which may lead to the misdiagnosis of bacterial pneumonia or even non-infectious diseases (7-9).
For various reasons, the true incidence of IPA among critical ill patients is variable (10). First, biopsy is hard to perform in ICU due to the potential adverse effects like bleeding or pneumothorax (11,12). Second, postmortem examinations are not routinely performed in most of medical institutions (13,14). Third, it remains challenging to discriminate between infection and colonization with positive Aspergillus species culture results. Forth, the sensitivity and specificity of biomarkers for diagnosing IPA varies a lot, especially in the nonneutropenic ICU patients.
The current recommended criteria by European Society for Clinical Microbiology and Infectious Diseases (ESCMID) and the Infectious Diseases Society of America (IDSA) still suggest collecting risk factors, chest imaging and biological data to support the diagnosis of IPA with a strong presumption (15,16). Recently, with the development of detecting technologies, non-culture-based methodologies are widely used in diagnosing IPA (17). Thus, we present the various arguments for the diagnosis of IPA in ICU patients, with a particular emphasis on the novel biomarkers and exploration techniques.
Epidemiology and risk factors of IPA in ICU
Previously, IPA was mostly observed in immunocompromised host in the department of hematology or oncology. It was once reported that the incidence of IPA could be as high as 59.2% in patients with hematopoietic stem-cell transplantation recipients (HSCT) whereas the incidence was relatively lower (1–6%) in those with solid organ transplantation (SOT) (18,19), even if the lung transplantation recipients are more at risk due to the early exposure of the spores in the contaminating environmental.
In the department of critical care setting, Aspergillus can harbour in the water and ventilation systems, as well as in various types of equipment. However, it is difficult to discriminate between true infection and colonization when Aspergillus is isolated from the lower respiratory tract specimens. The incidence of IPA in ICU ranges from 0.3% to 5.8% (20,21), and the overall mortality rate is over 80% (22,23).
The “classis” risk factor known as neutropenia was only observed in 10%-15% ICU patients while other factors were recognized in nonneutropenic ICU patients these years (24). Critical ill patients in ICU are subjected to multiple therapies to treat the diseases or even to maintain life (25,26). The therapeutic schedule may include insertion of central venous catheters (CVC), invasive mechanical ventilation (IMV), continuous renal replacement therapy (CRRT), parenteral nutrition (PN) or broad-spectrum antibiotics application that may affect the immunological defence system (27,28).
Chronic obstructive pulmonary disease (COPD) may lead to hypercapnia and sometimes IMV is need. Furthermore, COPD has become a considerable risk factor for IPA in ICU patients (29,30). It seems that Aspergillus colonization can easily be detected in the lower tract of airway which may lead to IPA due to lung structure alterations, reduced mucociliary clearance, impaired immunological response and mucosal lesions (31). Both compensated and decompensated cirrhosis have been described as risk factors for IA, and impaired phagocytosis has been proposed as a possible explanation for heightened risk in these groups (32). Diabetes has been observed as another risk factor due to the impaired innate and acquired immunity caused by hyperglycemia (33). Other risk factors like influenza and extracorporeal membrane oxygenation (ECMO) were also noted in the lecture (34-37).
IPA definition in ICU
Improve diagnostic accuracy of IPA in ICU is quite challenging. The criteria of IPA include proven, probable and possible diagnosis (38) which was only validated in immunocompromised host like HSCT or SOT.
Positive Aspergillus culture in respiratory tract samples is not uncommon in ICU and the upward tendency has been observed these years (39). However, to discriminate colonization and real infection remains a vexed question. To overcome this obstacle, the AspICU clinical algorithm has been proposed to discriminate IPA from Aspergillus colonization in ICU patients with higher diagnostic utility than existing tests (40) (Table 1). In Blot’s multicenter observational study (40), this algorithm revealed 92% sensitivity and 61% specificity among 115 IPA proven patients. Although it seems to be an effective method to discriminate disease from colonization in critical ill patients, there still a number of shortages. This clinical algorithm includes at least one positive Aspergillus culture in a respiratory tract specimen as an essential inclusion criterion, while positive results are obtained for only approximately 50% of patients with IPA (41) that leads to misdiagnosis or missed diagnosis of IPA patients with negative culture results. Another flaw is that the algorithm does not include any antigen or DNA testing. Schroeder M tried to modify the algorithm by adding BALF GM value (42), and the study revealed that it could increase the diagnostic sensitivity for IPA in ICU patients. Anyhow, items of the algorithm really need to be modified since the development of new testing methods, and it is still a long way to go (43).

Full table
Diagnostic methodologies of IPA
Histological detection
Direct histopathological identification in lung tissue biopsies remains the gold standard for diagnosing IPA which is performed by CT-guided transthoracic biopsies, convex endobronchial ultrasound transbronchial needle aspiration or transbronchial biopsies in most cases (44-46) (Figure 1). According to Hoffer’s retrospective study (47), 28 percutaneous biopsies revealed that IPA was diagnosed with 100% (18/18) specificity and 100% (10/10) sensitivity in the cohort of immunosuppressed children.
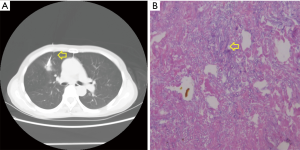
As is well-known, lung biopsy is not without risk in critical ill patients who are suspected IPA, especially in those with respiratory failure or even need invasive mechanical ventilation. According to Libby’s meta analysis, the surgical complication rate of lung biopsy was about 22% which might lead to an overall 44% mortality rate (48). In patients with invasive mechanical ventilation, the incidence of complication could be as high as approximate to 60% (49).
Air leak and pneumothorax are the most common lung biopsy complication while procedure related bleeding or hypoxia can also be observed in some patients (50-53). The question as to whether to perform lung biopsy in patients suspected IPA does not have a clear answer till now (54,55). The benefit of this invasive procedure must be weighed carefully against both the inherent risk and the risk of a result which might influence IPA therapy.
Direct microscopic examination and culture
The biological sampling is targeted to the anatomic site of Aspergillar disease development. The deeper and closer to the suspicious lesion initially observed on imaging sampling is made, the more it is pertinent (56). Direct microscopic examination is always performed on a fresh sample like bronchoalveolar lavage fluid (BALF), in a wet medium between a glass slide and cover slip (57-59). The morphological characteristics of Aspergillus spp. are the presence of hyaline and septate hyphae with dichotomous branches at angles of 45° and with uniform width (3–6 µm) (60,61). The direct examination is sometimes performed after fluorescent marking, which can increase the sensitivity of hyphae detection. Sometimes Aspergillus crown could be observed in KOH direct compression (Figure 2). However, it is really a hard work or impossible to distinguish the species of Aspergillus because of the difficulty in distinguishing the morphology of the different fungi species (62,63). What is more, Pseudallescheria boydii and Fusarium were reported to have the same morphological characteristics (64).
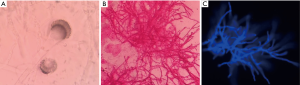
Sabouraud’s dextrose-agar is the most used medium in culture method. The global yield of the positive culture results are rather moderate, including for IPA proven, at 50% (65). Aspergillus spp. usually grows in 2 to 5 days, or less for some species if the culture medium is kept at 37 °C (66). In addition, the experience and knowledge of the microbiologist is of great importance to identify the colonies correctly.
Bronchoscopy
The development of bronchoscopy and bronchoalveolar lavage (BAL) has led to an increased use in ICU, where their applications for differential diagnosis of pulmonary diseases make them indispensable instruments for intensivists (67-69). Invasive aspergillus tracheobronchitis (IATB) is no longer rare only in immunocompromised host or those with malignancy in ICU which may be an early period of IPA (70). It was reported that lung parenchyma was usually involved together with IATB which led to a poor prognosis and high mortality (71).
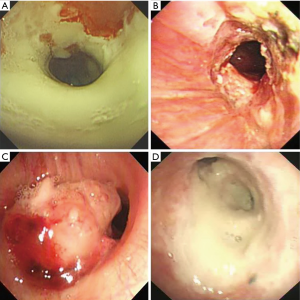
Intraluminal lesions could be divided into four types according to the relevant bronchoscopic features (Figure 3). (I) Full-layer involvement type: tracheobronchial lesions infiltrating through the matrix layer of bronchi with extensive pseudomembrane formation; (II) ulcerative type: superficial ulcer and inflammatory infiltration in tracheobronchial mucosa; (III) occlusion type: bronchus obstruction >50% of the original caliber which caused by extensive pseudomembrane formation or necrotic tissues and (IV) mixed type: two or more different forms of typical bronchoscopic features coexisting in one patient which sometimes might lead to tracheoesophageal fistula (72-74). As the most direct and effective method in IATB diagnosis, bronchoscopy plays a crucial part in differential diagnosis of immunocompromised patients in ICU. Early detection of airway lesions might improve the prognosis of those with IATB (75).
Chest imaging
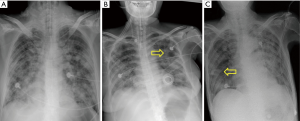
Over the past few decades, chest radiographs and thoracic computed tomography (CT) scan have become the most important diagnostic tools for lung diseases since they are non-invasive procedures (76). However, chest radiography is nonspecific in ICU patients with IPA, especially in nonneutropenic patients (Figure 4). The classic air crescent sign and halo sign are less often present and their sensitivity is relatively low (less than 25%) than in neutropenic patients in ICU (77). Moreover, even if the halo sign is seen in the chest CT, the specific of IPA is not satisfactory. For example, the halo sign may be observed in pneumonia due to highly virulent bacteria like pseudomonas aeruginosa, community-acquired methicillin-resistant staphylococcus aureus or klebsiella pneumoniae. Sometimes it can be seen in other pathological states due to neoplasm metastasis, Wegener’s granulomatosis or dermatomyositis (78-80). What is more, dual infection is not uncommon in ICU patients which may complicate the clinical diagnosis of IPA based on chest CT alone (Figure 5).
Besides the classic halo sign and air crescent sign, other findings from chest CT have been reported with various frequencies, especially in nonneutropenic patients in ICU: cavitary lesions, pleural effusion, pulmonary macro-nodules (>1 cm) and micro-nodules (<1 cm), areas of alveolar consolidation with or without infarction, etc. (81-83). According to Caillot et al., the multiple lesions of lung seems to be one significant prognosis factor of 90-day mortality in patients with IPA (RR =4.9, P=0.001) (84).
[18F]FDG-PET has been applied to improve the radiological detection of IPA. However, up taking of the tracer during IPA is indistinguishable from those seen during bacterial infections, inflammatory reactions, or even during cancer. The specificity of PET for diagnosing IPA has been dramatically improved these years through the application of Aspergillus siderophores, and monoclonal antibodies conjugated to radionuclides. What we should note is that all these studies to date have been conducted only in animal models of IPA (85).
Biomarkers detection
Pentraxin 3 (PTX3)
PTX3 is a soluble pattern recognition receptor (PRR) which is produced by nonimmune cells and phagocytes at the sites of injury or inflammation (86). Therefore, it has been recognized as a biomarker of sepsis in the last decade (87,88). Some recent studies showed that PTX3 had a non-redundant role in modulating various effect or pathways involved in immune resistance to Aspergillus fumigatus, including activating innate immune cells and driving protective adaptive immunity (89).
As Kabbani reported (90), the concentrations of PTX3 in BALF was higher in IPA patients compared with those in the state of Aspergillus colonization [439.20 (IQR, 168.18–778.90) vs. 68.93 (IQR 13.67–156.74) pg/mL, P<0.001]. What is more, it might help to identify false-positive galactomannan (GM) values in BALF samples. Another study by Li revealed that BALF PTX3 had a satisfactory diagnostic efficacy (91). When the cutoff value of BALF PTX3 was set at 1.9 ng/mL, the sensitivity was 86.3% while the specificity was 82.5%, respectively.
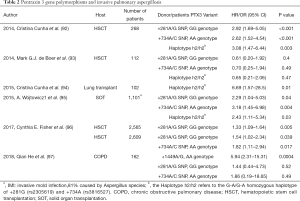
PTX3 plays an important role in antifungal immunity and it was once thought that genetic PTX3 deficiency might be a reliable method to predict the incidence of IPA. However, the real effect is still controversial. The genetic deficiency in homozygous haplotype (h2/h2) of PTX3 was first reported by Cristina Cunha in 2014 (92). In 268 patients with HSCT, it revealed that PTX3 gene polymorphisms associated with an increased risk of IPA in the discovery study (adjusted HR, 3.08; P=0.003) and the confirmation study (adjusted HR, 2.78; P=0.03). It was presumed that the instability of messenger RNA might lead to impaired phagocytosis and clearance of the aspergillus. However, the results could not be confirmed by de Boer (93). Forty-four patients with HSCT and 68 controls were enrolled in their case-control study. No association of donor PTX3 variants (+281A→G SNP, GG genotype; +734A→C SNP, AA genotype; Haplotype h2/h2) with the risk of IPA was found during their study. What is more, many other studies about different hosts have come to different conclusions these years (94-97) (Table 2). This creates challenges in generalizability. It is recognized that many single-nucleotide polymorphisms (SNPs) in innate immune genes have been linked to an increased risk of IPA, but the disease probably may not develop unless other immune deficiencies are present. Whether the genetic deficiency of PTX3 contributes to the incidence of IPA needs further study, and a systems-biology approach may help in understanding the clinical consequences of genetic polymorphisms affecting innate immunity.
Full table
Triacetylfusarinine C (TAFC)
In order to overcome iron restriction by the host and obtain iron for growth, Aspergillus species always secrete low-molecular mass iron chelators, which is called siderophores. The main category of siderophore of A. fumigatus is triacetylfusarinine C (TAFC) which is produced only by actively growing cells and cannot be detected in conidia (98). It is reported that TAFC can be tested in serum and BALF samples in patients with IPA using ultra performance liquid chromatography tandem mass spectrometry (LC MS/MS) (99,100). Meanwhile, TAFC is also a small molecule (905,323 g/mol) with a relatively short half-life in blood which leads to a rapid clearance (99). Thus, it can be detected in kidneys, bladder as well as urine samples. It has been demonstrated in Martin’ study that TAFC/creatinine index determination in urine had a high sensitivity (86%) and specificity (88%) in diagnosing IPA which may be a promising biomarker because of the non-invasive sampling (101).
Metabolites detection
A great large number of secondary metabolites can be produced by Aspergillus fumigatus, and almost all of them are volatile (102). Thus, they might be detected as biomarkers like volatile organic compounds (VOCs) in exhaled breath. It seems to be a promising diagnostic method due to the non-invasive techniques and the demand rates of BAL or lung biopsy could be decreased.
On the other hand, it has been demonstrated that a great variety of characteristic VOCs could be produce by A. fumigatus in vitro. The metabolites may vary a lot depending on the antifungal treatment or respiratory microbiota. Therefore, a set of metabolites rather than a very specific one is much preferred as the “breath-signature” of IPA. Four metabolites (trans-geranylacetone, α-trans-bergamotene, β-trans-bergamotene and β-vatirenene) have been proven to diagnose IPA with a higher sensitivity (94%) and specificity (93%) (103). Another study by Chambers revealed that 2-pentylfuran might be a relatively specific marker of IPA (104). Anyhow, further investigation needs to be carried out and the specific set of metabolites should be reintegration.
Proteins detection
Matrix assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS) has become a rapid and accurate technology for the identification of bacteria as well as yeast and mold species in recent years. Filamentous fungi can be identified using MALDI-TOF MS, although Aspergillus species demonstrate variable phenotypes as a result of which protein spectra may vary (105). However, MALDI-TOF MS application for the identification of Aspergillus species is limited till now due to the insufficient fungal databases and a good quality of mass spectra is hard to obtain. Fungal cell wall is thicker and more robust than that of bacteria which also makes it more difficult to use this technology (106,107). Anyhow, the novel method will be a great advance in reducing the time of IPA diagnosis in ICU.
What the future holds
In the last century, identification of Aspergillus species was routinely based on the morphological characteristic from direct microscopic examination. Although it requires expertise and with low sensitivity, it can detect the Aspergillus hyphae in less than 15 minutes. Thus, it should not be forgotten as a traditional technique with high feasibility.
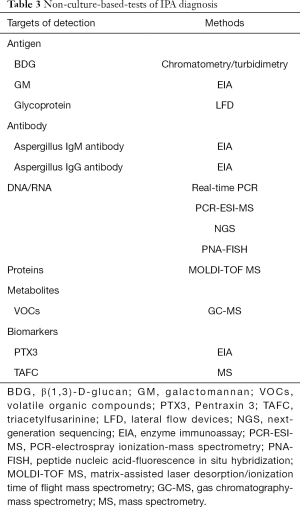
A great number of novel non-culture-based-tests of IPA diagnosis have been well developed in the last few years (Table 3). Detection technology has entered the era of molecules and genes. However, there is no perfect diagnostic method for IPA till now and each technique has its own limitation which must consequently be confronted in the future. Combination of detection methods is recommended to elevate the sensitivity and specificity so as to improve the prognosis of IPA patients in ICU.
Full table
Acknowledgments
Funding: This work was supported by the Scientific Research Project of Shanghai Municipal Health Bureau (201840006) and National Science Foundation of China (No. 81601745).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Hurst SF, Berkow EL, Stevenson KL, et al. Isolation of azole-resistant Aspergillus fumigatus from the environment in the south-eastern USA. J Antimicrob Chemother 2017;72:2443-6. [Crossref] [PubMed]
- Lavergne RA, Chouaki T, Hagen F, et al. Home Environment as a Source of Life-Threatening Azole-Resistant Aspergillus fumigatus in Immunocompromised Patients. Clin Infect Dis 2017;64:76-8. [Crossref] [PubMed]
- Onishi K, Muhammad Sarumoh B, Hagiwara D, et al. Azole-resistant Aspergillus fumigatus Containing a 34-bp Tandem Repeat in cyp51A Promoter is Isolated from the Environment in Japan. Med Mycol J 2017;58:E67-E70. [Crossref] [PubMed]
- Tangwattanachuleeporn M, Minarin N, Saichan S, et al. Prevalence of azole-resistant Aspergillus fumigatus in the environment of Thailand. Med Mycol 2017;55:429-35. [PubMed]
- Kaneko T, Ishigatsubo Y. Increasing incidence of invasive pulmonary aspergillosis and its early diagnosis. Intern Med 2001;40:1073-4. [Crossref] [PubMed]
- Sun KS, Tsai CF, Chen SC, et al. Galactomannan Testing and the Incidence of Invasive Pulmonary Aspergillosis: A 10-Year Nationwide Population-Based Study in Taiwan. PloS One 2016;11:e0149964. [Crossref] [PubMed]
- Abers MS, Ghebremichael MS, Timmons AK, et al. A Critical Reappraisal of Prolonged Neutropenia as a Risk Factor for Invasive Pulmonary Aspergillosis. Open Forum Infect Dis 2016;3:ofw036. [Crossref] [PubMed]
- Caffrey-Carr AK, Hilmer KM, Kowalski CH, et al. Host-Derived Leukotriene B4 Is Critical for Resistance against Invasive Pulmonary Aspergillosis. Front Immunol 2018;8:1984. [Crossref] [PubMed]
- Olaechea Astigarraga PM, Alvarez Lerma F, Zaldibar Enriquez E. Invasive pulmonary aspergillosis in the non-neutropenic critical patient: future challenges. Med Intensiva 2006;30:386-91. [Crossref] [PubMed]
- Bassetti M, Garnacho-Montero J, Calandra T, et al. Intensive care medicine research agenda on invasive fungal infection in critically ill patients. Intensive Care Med 2017;43:1225-38. [Crossref] [PubMed]
- Kim K, Lee MH, Kim J, et al. Importance of open lung biopsy in the diagnosis of invasive pulmonary aspergillosis in patients with hematologic malignancies. Am J Hematol 2002;71:75-9. [Crossref] [PubMed]
- Kita H, Kobayashi Y, Yamashita K, et al. Bronchial biopsy in allergic bronchopulmonary aspergillosis without clinical asthma. Nihon Kokyuki Gakkai Zasshi 2003;41:411-5. [PubMed]
- Peddada K, Khan NM, Rubin J, et al. Diagnosis of Vitreoretinal Aspergillosis with Transvitreal Retinochoroidal Biopsy. Case Rep Ophthalmol Med 2018;2018:8306163. [Crossref] [PubMed]
- Rantakokko-Jalava K, Laaksonen S, Issakainen J, et al. Semiquantitative detection by real-time PCR of Aspergillus fumigatus in bronchoalveolar lavage fluids and tissue biopsy specimens from patients with invasive aspergillosis. J Clin Microbiol 2003;41:4304-11. [Crossref] [PubMed]
- Ullmann AJ, Aguado JM, Arikan-Akdagli S, et al. Diagnosis and management of Aspergillus diseases: executive summary of the 2017 ESCMID-ECMM-ERS guideline. Clin Microbiol Infect 2018;24 Suppl 1:e1-e38. [Crossref] [PubMed]
- Patterson TF, Thompson GR 3rd, Denning DW, et al. Practice Guidelines for the Diagnosis and Management of Aspergillosis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis 2016;63:e1-e60. [Crossref] [PubMed]
- Moura S, Cerqueira L, Almeida A. Invasive pulmonary aspergillosis: current diagnostic methodologies and a new molecular approach. Eur J Clin Microbiol Infect Dis 2018;37:1393-403. [Crossref] [PubMed]
- Neofytos D, Horn D, Anaissie E, et al. Epidemiology and outcome of invasive fungal infection in adult hematopoietic stem cell transplant recipients: analysis of Multicenter Prospective Antifungal Therapy (PATH) Alliance registry. Clin Infect Dis 2009;48:265-73. [Crossref] [PubMed]
- Salman N, Torun SH, Budan B, et al. Invasive aspergillosis in hematopoietic stem cell and solid organ transplantation. Expert Rev Anti Infect Ther 2011;9:307-15. [Crossref] [PubMed]
- Garnacho-Montero J, Amaya-Villar R, Ortiz-Leyba C, et al. Isolation of Aspergillus spp. from the respiratory tract in critically ill patients: risk factors, clinical presentation and outcome. Crit Care 2005;9:R191-9. [Crossref] [PubMed]
- Meersseman W, Vandecasteele SJ, Wilmer A, et al. Invasive aspergillosis in critically ill patients without malignancy. Am J Respir Crit Care Med 2004;170:621-5. [Crossref] [PubMed]
- Taccone FS, Van den Abeele AM, Bulpa P, et al. Epidemiology of invasive aspergillosis in critically ill patients: clinical presentation, underlying conditions, and outcomes. Crit Care 2015;19:7. [Crossref] [PubMed]
- Vandewoude K, Blot S, Benoit D, et al. Invasive aspergillosis in critically ill patients: analysis of risk factors for acquisition and mortality. Acta Clin Belg 2004;59:251-7. [Crossref] [PubMed]
- Garnacho-Montero J, Olaechea P, Alvarez-Lerma F, et al. Epidemiology, diagnosis and treatment of fungal respiratory infections in the critically ill patient. Rev Esp Quimioter 2013;26:173-88. [PubMed]
- Bassetti M, Carnelutti A, Righi E. Issues in the management of invasive pulmonary aspergillosis in non-neutropenic patients in the intensive care unit: A role for isavuconazole. IDCases 2018;12:7-9. [Crossref] [PubMed]
- Dimopoulos G, Frantzeskaki F, Poulakou G, et al. Invasive aspergillosis in the intensive care unit. Ann N Y Acad Sci 2012;1272:31-9. [Crossref] [PubMed]
- Hedayati MT, Khodavaisy S, Alialy M, et al. Invasive aspergillosis in intensive care unit patients in Iran. Acta medica 2013;56:52-6. [PubMed]
- Kaziani K, Mitrakou E, Dimopoulos G. Improving diagnostic accuracy for invasive pulmonary aspergillosis in the intensive care unit. Ann Transl Med 2016;4:352. [Crossref] [PubMed]
- Agarwal R, Hazarika B, Gupta D, et al. Aspergillus hypersensitivity in patients with chronic obstructive pulmonary disease: COPD as a risk factor for ABPA? Med Mycol 2010;48:988-94. [Crossref] [PubMed]
- Dauchy C, Bautin N, Nseir S, et al. Emergence of Aspergillus fumigatus azole resistance in azole-naive patients with chronic obstructive pulmonary disease and their homes. Indoor Air 2018;28:298-306. [Crossref] [PubMed]
- Shahi M, Ayatollahi Mousavi SA, Nabili M, et al. Aspergillus colonization in patients with chronic obstructive pulmonary disease. Curr Med Mycol 2015;1:45-51. [Crossref] [PubMed]
- Prattes J, Hoenigl M, Krause R, et al. Invasive aspergillosis in patients with underlying liver cirrhosis: a prospective cohort study. Med Mycol 2017;55:803-12. [Crossref] [PubMed]
- Ghanaat F, Tayek JA. Weight loss and diabetes are new risk factors for the development of invasive aspergillosis infection in non-immunocompromized humans. Clinical practice 2017;14:296-301. [PubMed]
- Lamoth F, Calandra T. Let's add invasive aspergillosis to the list of influenza complications. The Lancet Respir Med 2018;6:733-5. [Crossref] [PubMed]
- Parcell BJ. Invasive pulmonary aspergillosis post extracorporeal membrane oxygenation support and literature review. Med Mycol Case Rep 2014;4:12-5. [Crossref] [PubMed]
- Rodriguez-Goncer I, Thomas S, Foden P, et al. Invasive pulmonary aspergillosis is associated with adverse clinical outcomes in critically ill patients receiving veno-venous extracorporeal membrane oxygenation. Eur J Clin Microbiol Infect Dis 2018;37:1251-7. [Crossref] [PubMed]
- Winiszewski H, Rougny AC, Lagoutte-Renosi J, et al. The pharmacokinetic challenge of treating invasive aspergillosis complicating severe influenzae assisted by extracorporeal membrane oxygenation. Crit Care 2018;22:355. [Crossref] [PubMed]
- Ascioglu S, Rex JH, de Pauw B, et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis 2002;34:7-14. [Crossref] [PubMed]
- Vandewoude KH, Vogelaers D, Blot SI. Aspergillosis in the ICU - The new 21st century problem? Med Mycol 2006;44:S71-S76. [Crossref] [PubMed]
- Blot SI, Taccone FS, Van den Abeele AM, et al. A clinical algorithm to diagnose invasive pulmonary aspergillosis in critically ill patients. Am J Respir Crit Care Med 2012;186:56-64. [Crossref] [PubMed]
- Wahidi MM, Herth FJ, Ernst A. State of the art: interventional pulmonology. Chest 2007;131:261-74. [Crossref] [PubMed]
- Schroeder M, Simon M, Katchanov J, et al. Does galactomannan testing increase diagnostic accuracy for IPA in the ICU? A prospective observational study. Crit Care 2016;20:139. [Crossref] [PubMed]
- Yu Y, Zhu C, Gao Y. Bronchoalveolar lavage fluid galactomannan as a diagnostic biomarker for IPA: still a long way to go. Crit Care 2016;20:280. [Crossref] [PubMed]
- García-Asensio S, Artigas JM, Barrena R. Optic nerve aspergillosis: report of a case diagnosed by fine-needle aspiration biopsy. Eur Radiol 2000;10:573-5. [Crossref] [PubMed]
- Kim K, Lee MH, Kim J, et al. Importance of open lung biopsy in the diagnosis of invasive pulmonary aspergillosis in patients with hematologic malignancies. Am J Hematol 2002;71:75-9. [Crossref] [PubMed]
- Özgün G, Tepeoglu M, Ozdemir BH, et al. Lung Biopsy Findings of Patients Who Have Undergone a Liver and Kidney Transplant. Exp Clin Transplant 2016;14:135-7. [PubMed]
- Hoffer FA, Gow K, Flynn PM, et al. Accuracy of percutaneous lung biopsy for invasive pulmonary aspergillosis. Pediatr Radiol 2001;31:144-52. [Crossref] [PubMed]
- Libby LJ, Gelbman BD, Altorki NK, et al. Surgical lung biopsy in adult respiratory distress syndrome: a meta-analysis. Ann Thorac Surg 2014;98:1254-60. [Crossref] [PubMed]
- Cronin CG, Sharma A, Digumarthy SR, et al. Percutaneous lung biopsy after pneumonectomy: factors for improving success in the care of patients at high risk. AJR Am J Roentgenol 2011;196:929-34. [Crossref] [PubMed]
- Patel SR, Karmpaliotis D, Ayas NT, et al. The role of open-lung biopsy in ARDS. Chest 2004;125:197-202. [Crossref] [PubMed]
- Rantakokko-Jalava K, Laaksonen S, Issakainen J, et al. Semiquantitative detection by real-time PCR of Aspergillus fumigatus in bronchoalveolar lavage fluids and tissue biopsy specimens from patients with invasive aspergillosis. J Clin Microbiol 2003;41:4304-11. [Crossref] [PubMed]
- Wong AK, Walkey AJ. Open Lung Biopsy Among Critically Ill, Mechanically Ventilated Patients. A Metaanalysis. Ann Am Thorac Soc 2015;12:1226-30. [PubMed]
- Lazguet Y, Maarouf R, Karrou M, et al. CT guided percutaneous needle biopsy of the chest: initial experience. Pan Afr Med J 2016;23:211. [Crossref] [PubMed]
- McSweeney SE, O'Regan KN, Mc Laughlin PD, et al. Evaluation of the efficacy and safety of percutaneous biopsy of lung. Open Respir Med J 2012;6:82-8. [Crossref] [PubMed]
- Palakshappa JA, Meyer NJ. Which patients with ARDS benefit from lung biopsy? Chest 2015;148:1073-82. [Crossref] [PubMed]
- Achar PN, Hermetz K, Rao S, et al. Microscopic studies on the Aspergillus flavus infected kernels of commercial peanuts in Georgia. Ecotoxicol Environ Saf 2009;72:2115-20. [Crossref] [PubMed]
- Das SK, Das AR, Guha AK. Adsorption behavior of mercury on functionalized aspergillus versicolor mycelia: atomic force microscopic study. Langmuir 2009;25:360-6. [Crossref] [PubMed]
- Yu Y, Zhu C, Zhang Z. Pneumocystis jiroveci pneumonia in patients with toxic epidermal necrolysis. J Emerg Crit Care Med 2018;2:37. [Crossref]
- Zhu C, Guo R, Zhou M, et al. Bloodstream infection caused by Cupriavidus gilardii in a systemic lupus erythematosus patient with invasive pulmonary aspergillosis. J Emerg Crit Care Med 2018;2:46. [Crossref]
- Gajjar DU, Pal AK, Ghodadra BK, et al. Microscopic evaluation, molecular identification, antifungal susceptibility, and clinical outcomes in fusarium, Aspergillus and, dematiaceous keratitis. Biomed Res Int 2013;2013:605308. [Crossref] [PubMed]
- Louis B, Waikhom SD, Roy P, et al. Invasion of Solanum tuberosum L. by Aspergillus terreus: a microscopic and proteomics insight on pathogenicity. BMC Res Notes 2014;7:350. [Crossref] [PubMed]
- Marfenina OE, Fomicheva GM, Gorlenko MV, et al. Ecological and physiological variations of saprotrophic and clinical isolates of the microscopic fungus Aspergillus sydowii (Bainier & Sartory) Thom & Church. Mikrobiologiia 2013;82:87-93. [PubMed]
- McClenny N. Laboratory detection and identification of Aspergillus species by microscopic observation and culture: the traditional approach. Med Mycol 2005;43 Suppl 1:S125-8. [Crossref] [PubMed]
- Hayden RT, Isotalo PA, Parrett T, et al. In situ hybridization for the differentiation of Aspergillus, Fusarium, and Pseudallescheria species in tissue section. Diagn Mol Pathol 2003;12:21-6. [Crossref] [PubMed]
- Boch T, Reinwald M, Spiess B, et al. Detection of invasive pulmonary aspergillosis in critically ill patients by combined use of conventional culture, galactomannan, 1-3-beta-D-glucan and Aspergillus specific nested polymerase chain reaction in a prospective pilot study. J Crit Care 2018;47:198-203. [Crossref] [PubMed]
- Guegan H, Robert-Gangneux F, Camus C, et al. Improving the diagnosis of invasive aspergillosis by the detection of Aspergillus in broncho-alveolar lavage fluid: Comparison of non-culture-based assays. J Infect 2018;76:196-205. [Crossref] [PubMed]
- Al-Qadi MO, Cartin-Ceba R, Kashyap R, et al. The Diagnostic Yield, Safety, and Impact of Flexible Bronchoscopy in Non-HIV Immunocompromised Critically Ill Patients in the Intensive Care Unit. Lung 2018;196:729-36. [Crossref] [PubMed]
- Greenstein YY, Shakespeare E, Doelken P, et al. Defining a Ventilation Strategy for Flexible Bronchoscopy on Mechanically Ventilated Patients in the Medical Intensive Care Unit. J Bronchology Interv Pulmonol 2017;24:206-10. [Crossref] [PubMed]
- Sandoval Gutiérrez JL. Fiberoptic bronchoscopy in the Intensive Care Unit. Certification, our challenge. Med Intensiva 2013;37:215. [PubMed]
- Wu N, Huang Y, Li Q, et al. Isolated invasive Aspergillus tracheobronchitis: a clinical study of 19 cases. Clin Microbiol Infect 2010;16:689-95. [Crossref] [PubMed]
- Huang HD, Li Q, Huang Y, et al. Pseudomembranous necrotizing tracheobronchial aspergillosis: an analysis of 16 cases. Chin Med J (Engl) 2012;125:1236-41. [PubMed]
- Liu WL, Lai CC. Pseudomembranous necrotizing tracheobronchial aspergillosis. QJM 2013;106:81. [Crossref] [PubMed]
- Majima S, Okachi S, Asano M, Wakahara K, Hashimoto N, Sato M, et al. Pseudomembranous Invasive Tracheobronchial Aspergillosis with Fulminant Hepatitis and Hemophagocytic Syndrome. Intern Med 2018;57:2371-5. [Crossref] [PubMed]
- Yu Y, Zhu C, Qian X, et al. Tracheoesophageal fistula induced by invasive pulmonary aspergillosis. Ann Transl Med 2016;4:345. [Crossref] [PubMed]
- Cao Y, Shao C, Song Y. Analysis of the clinical features of invasive bronchopulmonary aspergillosis. Clin Respir J 2018;12:1635-43. [Crossref] [PubMed]
- Alves GR, Marchiori E, Irion K, et al. The halo sign: HRCT findings in 85 patients. J Bras Pneumol 2016;42:435-9. [Crossref] [PubMed]
- Dai Z, Zhao H, Cai S, et al. Invasive pulmonary aspergillosis in non-neutropenic patients with and without underlying disease: a single-centre retrospective analysis of 52 subjects. Respirology 2013;18:323-31. [Crossref] [PubMed]
- Desoubeaux G, Bailly E, Chandenier J. Diagnosis of invasive pulmonary aspergillosis: updates and recommendations. Med Mal Infect 2014;44:89-101. [Crossref] [PubMed]
- Hauggaard A, Ellis M, Ekelund L. Early chest radiography and CT in the diagnosis, management and outcome of invasive pulmonary aspergillosis. Acta Radiol 2002;43:292-8. [Crossref] [PubMed]
- Imburgia C, Buscemi C, Corrao S. Pulmonary infiltrate with characteristic "reversed halo" and "air crescent" signs. Eur J Intern Med 2017;41:e3-e4. [Crossref] [PubMed]
- Karam GH, Griffin FM Jr. Invasive pulmonary aspergillosis in nonimmunocompromised, nonneutropenic hosts. Rev Infect Dis 1986;8:357-63. [Crossref] [PubMed]
- Prasad A, Agarwal K, Deepak D, et al. Pulmonary Aspergillosis: What CT can Offer Before it is too Late! J Clin Diagn Res 2016;10:TE01-5. [PubMed]
- Raju S, Ghosh S, Mehta AC, Chest CT. Signs in Pulmonary Disease: A Pictorial Review. Chest 2017;151:1356-74. [Crossref] [PubMed]
- Caillot D, Mannone L, Cuisenier B, et al. Role of early diagnosis and aggressive surgery in the management of invasive pulmonary aspergillosis in neutropenic patients. Clin Microbiol Infect 2001;7 Suppl 2:54-61. [Crossref] [PubMed]
- Thornton CR. Molecular Imaging of Invasive Pulmonary Aspergillosis Using ImmunoPET/MRI: The Future Looks Bright. Front Microbiol 2018;9:691. [Crossref] [PubMed]
- Garlanda C, Hirsch E, Bozza S, et al. Non-redundant role of the long pentraxin PTX3 in anti-fungal innate immune response. Nature 2002;420:182-6. [Crossref] [PubMed]
- Hamed S, Behnes M, Pauly D, et al. Pentraxin-3 Predicts Short- and Mid-term Mortality in Patients with Sepsis and Septic Shock During Intensive Care Treatment. Clin Lab 2018;64:999-1011. [Crossref] [PubMed]
- Lee YT, Gong M, Chau A, et al. Pentraxin-3 as a marker of sepsis severity and predictor of mortality outcomes: A systematic review and meta-analysis. J Infect 2018;76:1-10. [Crossref] [PubMed]
- Moalli F, Doni A, Deban L, et al. Role of complement and Fc{gamma} receptors in the protective activity of the long pentraxin PTX3 against Aspergillus fumigatus. Blood 2010;116:5170-80. [Crossref] [PubMed]
- Kabbani D, Bhaskaran A, Singer LG, et al. Pentraxin 3 levels in bronchoalveolar lavage fluid of lung transplant recipients with invasive aspergillosis. J Heart Lung Transplant 2017;36:973-9. [Crossref] [PubMed]
- Li H, Liu L, Zhou W, et al. Pentraxin 3 in bronchoalveolar lavage fluid and plasma in non-neutropenic patients with pulmonary aspergillosis. Clin Microbiol Infect 2019;25:504-10. [Crossref] [PubMed]
- Cunha C, Aversa F, Lacerda JF, et al. Genetic PTX3 deficiency and aspergillosis in stem-cell transplantation. N Engl J Med 2014;370:421-32. [Crossref] [PubMed]
- de Boer MG, Halkes CJ, van de Vosse E. PTX3 deficiency and aspergillosis. N Engl J Med 2014;370:1665-6. [Crossref] [PubMed]
- Cunha C, Monteiro AA, Oliveira-Coelho A, et al. PTX3-Based Genetic Testing for Risk of Aspergillosis After Lung Transplant. Clin Infect Dis 2015;61:1893-4. [Crossref] [PubMed]
- Wójtowicz A, Lecompte TD, Bibert S, et al. PTX3 Polymorphisms and Invasive Mold Infections After Solid Organ Transplant. Clin Infect Dis 2015;61:619-22. [Crossref] [PubMed]
- Fisher CE, Hohl TM, Fan W, et al. Validation of single nucleotide polymorphisms in invasive aspergillosis following hematopoietic cell transplantation. Blood 2017;129:2693-701. [Crossref] [PubMed]
- He Q, Li H, Rui Y, et al. Pentraxin 3 Gene Polymorphisms and Pulmonary Aspergillosis in Chronic Obstructive Pulmonary Disease Patients. Clin Infect Dis 2018;66:261-7. [Crossref] [PubMed]
- Szigeti ZM, Szaniszlo S, Fazekas E, et al. Optimization of triacetylfusarinine C and ferricrocin productions in Aspergillus fumigatus. Acta Microbiol Immunol Hung 2014;61:107-19. [Crossref] [PubMed]
- Haas H. Fungal siderophore metabolism with a focus on Aspergillus fumigatus. Nat Prod Rep 2014;31:1266-76. [Crossref] [PubMed]
- Orasch T, Prattes J, Faserl K, et al. Bronchoalveolar lavage triacetylfusarinine C (TAFC) determination for diagnosis of invasive pulmonary aspergillosis in patients with hematological malignancies. J Infect 2017;75:370-3. [Crossref] [PubMed]
- Hoenigl M, Orasch T, Faserl K, et al. Triacetylfusarinine C: A urine biomarker for diagnosis of invasive aspergillosis. J Infect 2019;78:150-7. [Crossref] [PubMed]
- Sethi S, Nanda R, Chakraborty T. Clinical application of volatile organic compound analysis for detecting infectious diseases. Clin Microbiol Rev 2013;26:462-75. [Crossref] [PubMed]
- Koo S, Thomas HR, Daniels SD, et al. A breath fungal secondary metabolite signature to diagnose invasive aspergillosis. Clin Infect Dis 2014;59:1733-40. [Crossref] [PubMed]
- Chambers ST, Syhre M, Murdoch DR, et al. Detection of 2-pentylfuran in the breath of patients with Aspergillus fumigatus. Med Mycol 2009;47:468-76. [Crossref] [PubMed]
- Brandão IS, Oliveira-Moraes H, Souza Motta CM, et al. Elastin increases biofilm and extracellular matrix production of Aspergillus fumigatus. Braz J Microbiol 2018;49:675-82. [Crossref] [PubMed]
- Li Y, Wang H, Zhao YP, et al. Evaluation of the Bruker Biotyper Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry System for Identification of Aspergillus Species Directly from Growth on Solid Agar Media. Front Microbiol 2017;8:1209. [Crossref] [PubMed]
- Vidal-Acuña MR, Ruiz-Perez de Pipaon M, Torres-Sanchez MJ, et al. Identification of clinical isolates of Aspergillus, including cryptic species, by matrix assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS). Med Mycol 2018;56:838-46. [Crossref] [PubMed]
Cite this article as: Shen H, Zhang Z, Yu Y. Diagnosis of invasive pulmonary aspergillosis in the intensive care unit: what we should concern and how to do better. J Emerg Crit Care Med 2019;3:54.


