
Setting intraoperative positive end expiratory pressure: how to be protective
Introduction
General anesthesia is nowadays considered as a safe procedure, with a relatively low mortality due to the improvement of anesthesiology and perioperative care, even though population is becoming elderly and with more co-morbidities. Despite an increase in popularity of locoregional techniques, general anesthesia and mechanical ventilation are still necessary in many surgical procedures; therefore several observational studies and randomized controlled trials have been conducted to better understand and improve ventilator management in different populations and surgical procedures (1-4). The role of modern anesthesiologists includes preventing postoperative complications, to improve the outcome of surgical patients, decreasing hospital length of stay and health-care systems costs.
During anesthesia, several mechanisms converge in reducing the functional residual capacity and impair gas exchange; while this mechanism has been traditionally considered the main determinant of postoperative pulmonary complications (PPCs) (5), research in the last two decades suggested that PPCs are the result of a complex interaction between patient-, anesthesia-, ventilation- and surgery-related factors (6). The occurrence of PPCs is estimated around 5–10% of patients (1), and it is associated with increased mortality and length of hospital stay (7); therefore, several scoring systems have been proposed to stratify patients’ risk. Among these, the ‘Assess Respiratory Risk in Surgical Patients in Catalonia risk score for PPCs’ (ARISCAT) score has been developed (8) and externally validated (9). Considering the high number of surgical procedures which require invasive ventilation—more than 200 million per year—identifying strategies to reduce the incidence of PPCs can have a great impact on patient’s outcomes and healthcare resources consumption. Protective mechanical ventilation strategies with low tidal volumes (Vt) set at 6 mL/kg of predicted body weight (10), and the use of positive end expiratory pressure (PEEP) are the standard of care in patients with acute respiratory distress syndrome (ARDS) (11,12). Many study groups hypothesized that low tidal volume and PEEP could be applied beneficially also in patients undergoing surgical procedures, even without pulmonary lesions. While the associations between higher plateau and driving (plateau-PEEP) pressures and PPCs have been demonstrated, the role of PEEP is still debated (13). The large observational trial LAS VEGAS (1) provided a snapshot on clinical practice in several countries, observing that two fixed levels of PEEP are typically used: 0 cmH2O or 5 cmH2O, also in patients with an increased risk of PPCs.
The aim of this review is to summarize the effects of PEEP in the surgical patient, to provide a review on the evidence regarding its use and to highlight gray areas warranting further research.
Pathophysiology: rationale of PEEP
Effects of general anesthesia on the respiratory system
Several functional changes occur in the respiratory system following the induction of general anesthesia, which result in respiratory mechanics alterations and gas exchange impairment; among these airway closure, atelectasis formation with formation of true shunt play a major role (14). Several mechanisms have been proposed for atelectasis formation during general anesthesia, including small airway collapse, lung structures compression, impairment of surfactant function and gas resorption. The application of PEEP can revert these effects and limit the formation of atelectasis (15). However, these advantages do not necessarily translate into improved clinical outcome (16).
Effects of PEEP in healthy lungs
Decades of research illustrated several potentially beneficial effects of PEEP during mechanical ventilation, especially in experimental studies in models of lung injury. In healthy lungs, the effects of PEEP on atelectasis reversal are mainly mediated by increased end-expiratory lung volume and improved ventilation/perfusion ratio (17). However, in patients with healthy lungs these beneficial effects seem to be more relevant in patients with major lung collapse following induction, as is the case of obese patients (18). The formation of atelectasis can persist in the post-operative period and could contribute to the development of PPCs through different mechanisms, e.g. inflammatory processes in the non-aerated lung regions (19). However, whether these theoretical advantages have a clinical impact is still debated and challenged by the findings of recent trials.
Pulmonary mechanics with PEEP application
Lung aeration starts early after induction of general anesthesia and, ideally, could be prevented by positive-pressure pre-oxygenation during the induction phase (20). This loss of aeration results in atelectasis formation and reduction of lung end-expiratory volume. The execution of a recruitment maneuver and the application of PEEP can revert this mechanism and restore lung aeration, also in patients at higher risk such as obese patients (21). However, compared to zero PEEP the application of a moderate PEEP level alone without recruitment maneuvers was sufficient to minimize atelectasis in a recent study in non-abdominal surgery (22). Therefore, PEEP and lung recruitment should maximize the lung volume available for ventilation, improving lung compliance (23). However, studies suggest that in many patients PEEP levels as low as 2 cmH2O can prevent lung collapse (24). Despite these theoretical advantages, an experimental study recently suggested that, in healthy lungs ventilated at low protective Vt, elevated PEEP levels increasing lung volume close to inspiratory capacity are injurious per se (25). This might explain why the advantages of protective intraoperative ventilation were observed when higher PEEP was used in conjunction with Vt reduction (26), but not when used alone at the same low Vt (3,4).
Hemodynamic effects of PEEP
After PEEP application, changes occur in the hemodynamic function. Overall, these effects are mediated by the action of airway pressure on the venous return, which according to Guyton is determined by compliance of veins, stressed volume, venous resistance and right atrial pressure (27). Lung volume change increases the difference between pleural and airway pressure, that could lead to vessels collapse and increase of pulmonary vascular resistances, reducing right ventricular injection. This phenomenon could be considered as a PEEP-induced capillary de-recruitment. On the contrary, reduction in end-expiratory lung volume determines alveolar collapse and increases vasomotor tone by pulmonary vasoconstriction (28). Patients with a low vascular reserve, as surgical patients that are often fasting before surgery, might be particularly sensitive to hypotensive effects (29). To counterbalance PEEP-induced hypotension fluids and vasoactive drugs might be required, but this might result in post-operative fluid overload.
Methods for setting PEEP in the operating room
Fixed PEEP levels
The commonest method for setting PEEP in the operating room is applying a fixed level to all patients (1), chosen by the clinician based on the characteristics of the patient and his own expertise and training. As mentioned above, in the general surgical population the most commonly used values are 0 and 5 cmH2O (1), while only slightly higher levels are used in particular cohort of patients, e.g., obese patients (30). It must be stressed that many anesthesia machines based on the bellows-in-bottle design, for technical reasons, cannot deliver an actual PEEP of 0 cmH2O, but a minimum level of 1–2 cmH2O: actual zero-PEEP conditions are rarely achieved in the clinical practice. These values typically ensure acceptable gas exchange in most patients. However, in patients with stiffer chest wall, such as in obesity or intra-abdominal hypertension, higher PEEP levels could be considered.
A recent hospital-based registry retrospective study (31) suggested that a PEEP level of 5 cmH2O was associated with a lower rate of PPCs compared with values higher or lower than 5. A large randomized multicenter trial compared two fixed PEEP strategies at the same protective Vt: 2 cmH2O without recruitment maneuvers versus PEEP of 12 cmH2O with recruitment maneuvers (4). The study included non-obese patients scheduled for open abdominal surgery at intermediate to high risk of developing PPCs. The higher fixed PEEP strategy allowed to maximize lung aeration, as reflected by an improvement of the dynamic compliance of the respiratory system, suggesting alveolar recruitment during mechanical ventilation for open abdominal surgery. A similar strategy was applied in another large randomized trial in patients with body mass index above 35 kg/m2 comparing PEEP of 4 versus 12 cmH2O, observing improved intraoperative oxygenation and compliance (2). Despite these findings concerning respiratory mechanics data, outcome of patients did not differ between the two study groups and the incidence of PPCs was similar, but hemodynamic impairment occurred more frequently in the high PEEP groups of both studies. This suggests that other mechanisms beside atelectasis could be involved in PPCs and morbidity after major surgery. Overall, the results of these two studies underline that a fixed PEEP level strategy is feasible, and that a low-moderate level is preferable in most patients.
Driving pressure (ΔP) or ‘open lung’ techniques
The ΔP is the difference between plateau pressure and PEEP, and has been proposed as a parameter to help anesthesiologist to set mechanical ventilation (32). It represents the dynamic strain of lung fibers and reflects the compliance of the respiratory system (33). After observing an association between high ΔP and the occurrence of PPCs (13), authors started proposing to titrate PEEP to the level corresponding to the minimum ΔP, namely to the highest respiratory system compliance. In this case, the level of PEEP can be set during a decremental PEEP test after a recruitment maneuver. PEEP increases the aerated volume both recruiting collapsed alveoli and causing hyperdistention; collapse and hyperdistention cause an increase of the ΔP, thus titrating PEEP based on this value could theoretically help balancing between these two unwanted phenomena (32). However, a recent physiological study challenged this assumption, observing that during ventilation of healthy lungs in surgical patients ΔP reflects lung aeration status only when no hyperinflation above the physiological functional residual capacity occurs, as is often the case when high PEEP levels are used (24).
A large randomized trial compared an ‘open lung’ PEEP titration strategy plus postoperative CPAP (3) in non-obese abdominal surgery patients at intermediate-high risk of PPCs. In this trial, PEEP titrated according to the lowest ΔP was compared with a fixed PEEP strategy with 5 cmH2O. The study did not find any difference in a composite of postoperative complications between the individualized strategies and standard, fixed-PEEP, lung-protective mechanical ventilation. A secondary outcome of PPCs was slightly favoring individualized intraoperative PEEP and postoperative CPAP.
The negative results of this trial do not make monitoring ΔP less important, as values above 13 cmH2O were associated with PPCs (13). Titration of PEEP has unclear ability to reduce this risk when identified but the clinician should keep in mind that if a PEEP increase translates in ΔP it means that harmful hyperdistention occurred, and PEEP should be lowered. An elevated ΔP is a marker of increased risk of PPC, and one method to lower ΔP and potentially the injuriousness of ventilation could be further reducing Vt (32).
While no clear superiority of titrated over fixed-PEEP strategies is demonstrated so far, further larger randomized trials could explore the possibility of further improving patient outcome through personalized ventilation.
Transpulmonary pressure (PL)
The pressure which is actually applied to alveoli during controlled positive pressure ventilation is the PL, that represents the difference between airways pressure and the pleural pressure. PL cannot be directly measured in the clinical practice, but it can be estimated measuring esophageal pressure (PES), a surrogate of pleural pressure (34,35). The measurement and interpretation of PES has been debated, but many physiological studies conducted in animal models and cadavers suggest that it is a reliable indicator of the absolute pleural pressures applied across the horizontal plane of the balloon. Titrating PEEP to achieve a positive end-expiratory PL improves oxygenation and compliance of the respiratory system in patients with ARDS thus could be a way to guide mechanical ventilation (36), but failed to show mortality benefits (37). The titration of PEEP based on PES can be applied in clinical practice for setting the optimal PEEP in critically ill patients but in the operating room it is limited to research purposes and physiologic mechanical respiratory system measurements (35).
Clinical recommendations
Every of the described methods has a clear physiologic rationale. The studies conducted so far, resumed in Table 1 (2-4,10,26,38-42), did not demonstrate the superiority of none of them. In absence of clear evidence, a fixed moderate-low PEEP between 2 and 5 cmH2O could be a reasonable initial value for most patients, while slightly higher values could be considered in specific subgroups of patients, as described below.
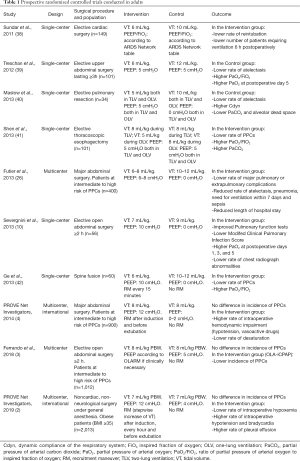
Full table
Grey areas
Some clusters of patients and types of surgical procedures have unique characteristics and specific features which might influence the choice of the PEEP level. In these fields, clinical evidence is particularly scarce and more clinical trials are required.
Laparoscopic and robotic surgery
Laparoscopic procedures are widely spread since they are less invasive compared to open surgery, but they present some features that can further impair the respiratory mechanics of patients. A pneumoperitoneum has to be induced by carbon dioxide inflation, with an increased abdominal pressure and cranial shift of the diaphragm. Then, many of these procedures require Trendelenburg positioning, that may worsen atelectasis formation in the dependent lung regions (43). The recent availability of robotic surgery makes positioning even more challenging. A PEEP level of 5 cmH2O applied to patients during laparoscopic procedures seems to be beneficial, leading to alveolar recruitment and improvement of chest wall and lung elastance (35). Higher levels could be considered in very long interventions and extreme positionings, as is the case of robotic surgery.
Duration of surgery
All the large randomized trials on intraoperative PEEP setting included procedures with an average duration around 3 hours. The role of PEEP in longer interventions is unclear; as the risk of lung collapse increases with the duration of invasive mechanical ventilation, higher PEEP levels might be considered in these patients to reduce atelectasis and possibly the incidence of PPCs.
Obese patients
Obese patients with an elevated body mass index present an increased intraabdominal pressure, with decreased chest wall elastance and lung volume, so they can experience a more severe gas exchange deterioration after anesthesia induction (44). Moreover, airway ΔP poorly reflects lung mechanics when chest wall compliance is reduced and intraabdominal pressure increases (45). Literature suggested how PEEP may improve respiratory mechanics in obese patients (18) and PEEP levels required to achieve lung recruitment might be proportional to the degree of obesity, as suggested by imaging-based studies (46). A large randomized trial was recently conducted, where the study group aimed to determine if this enhancement of respiratory function would also have a clinical effect on patients’ outcome (2). Around 2,000 patients with BMI ≥35 kg/m2 were randomized in two groups: higher level of PEEP (12 cmH2O with alveolar recruitment maneuvers) and low level of PEEP (4 cmH2O); the set tidal volume was the same in both groups. Remarkably, the data of this study demonstrate how an improvement of respiratory function due to higher levels of PEEP is not reflected by a reduction of PPCs. These findings suggest that setting a PEEP level around 12 cmH2O in obese patients is not routinely suitable, since the reduction of intraoperative ΔP and atelectasis are not related to a clinical enhancement.
Neurosurgical patients
Patients undergoing neurosurgical procedures and neurologically ill patients undergoing surgery (e.g., trauma patients) are more sensitive to hypercapnia because it may cause cerebral vasodilation with consequent detrimental effects on cerebral perfusion pressure and intracranial pressure. Application of PEEP in neurologically ill patients was traditionally considered harmful because of the reduction of the venous outflow (47), while recent evidence demonstrate that the application of moderate PEEP levels in both pediatric and adult population undergoing neurosurgical procedures may be safe, if an adequate arterial pressure and cerebral perfusion pressure are preserved (48-51). However, there is paucity of literature concerning the intraoperative ventilatory management of these patients, and caution should be applied when setting the PEEP level.
One lung ventilation (OLV)
During thoracic surgery, OLV is often required to allow surgeons to access the lung, chest wall or mediastinum. One lung is non-aerated and collapsed, with consequent potential alveolar inflammation and acting as shunt. The other lung must provide gas exchange for the organism during the intraoperative period. Incidence of PPCs is higher than in other surgical scenarios. A meta-analysis based on 22 studies suggests that a PEEP level ≥5 cmH2O may have a beneficial effect in reducing PPCs during one-lung ventilation (52). A large randomized controlled trial is ongoing (53) to investigate the effects of high PEEP (10 cmH2O plus recruitment maneuvers) and low PEEP (5 cmH2O) on PPCs during one-lung ventilation for thoracic surgery. In this setting, it must be remembered that when the non-dependent lung is excluded from ventilation, applying elevated PEEP to the dependent lung could divert the pulmonary blood flow to the excluded lung, increasing shunt and worsening oxygenation.
Conclusions
Adjusting PEEP levels influences gas exchange, hemodynamics and lung mechanics, together with the other ventilation parameters and the mechanical properties of the lung and chest wall to which ventilation is applied. A minimal value of PEEP between 2 and 5 cmH2O can be appropriate for most patients, to avoid atelectasis and compensate the loss of volume due to body positioning and loss of muscular tone. Since an ‘open lung’ approach doesn’t seem to improve outcome, an approach comprising a certain degree of ‘permissive atelectasis’ might be considered. Level of PEEP, as well as the other ventilation parameters, has to be adapted to specific surgical settings such as laparoscopic procedures, obese patients, one-lung ventilation, neurologically ill patients and long-duration procedures. A reasoned approach to PEEP titration is warranted, based on clinical practice and scientific evidence.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- LAS VEGAS investigators. Epidemiology, practice of ventilation and outcome for patients at increased risk of postoperative pulmonary complications: LAS VEGAS-an observational study in 29 countries. Eur J Anaesthesiol 2017;34:492-507. [Crossref] [PubMed]
- Writing Committee for the PROBESE Collaborative Group of the PROtective VEntilation Network (PROVEnet) for the Clinical Trial Network of the European Society of Anaesthesiology, Bluth T, Serpa Neto A, et al. Effect of Intraoperative High Positive End-Expiratory Pressure (PEEP) With Recruitment Maneuvers vs Low PEEP on Postoperative Pulmonary Complications in Obese Patients: A Randomized Clinical Trial. JAMA 2019;321:2292-305. [Crossref] [PubMed]
- Ferrando C, Soro M, Unzueta C, et al. Individualised perioperative open-lung approach versus standard protective ventilation in abdominal surgery (iPROVE): a randomised controlled trial. Lancet Respir Med 2018;6:193-203. [Crossref] [PubMed]
- PROVE Network Investigators for the Clinical Trial Network of the European Society of Anaesthesiology, Hemmes SN, Gama de Abreu M, et al. High versus low positive end-expiratory pressure during general anaesthesia for open abdominal surgery (PROVHILO trial): a multicentre randomised controlled trial. Lancet 2014;384:495-503. [Crossref] [PubMed]
- Hedenstierna G, Edmark L, Perchiazzi G. Postoperative lung complications: have multicentre studies been of any help? Br J Anaesth 2015;114:541-3. [Crossref] [PubMed]
- Ball L, Costantino F, Orefice G, et al. Intraoperative mechanical ventilation: state of the art. Minerva Anestesiol 2017;83:1075-88. [PubMed]
- Serpa Neto A, Hemmes SN, Barbas CS, et al. Incidence of mortality and morbidity related to postoperative lung injury in patients who have undergone abdominal or thoracic surgery: a systematic review and meta-analysis. Lancet Respir Med 2014;2:1007-15. [Crossref] [PubMed]
- Canet J, Gallart L, Gomar C, et al. Prediction of postoperative pulmonary complications in a population-based surgical cohort. Anesthesiology 2010;113:1338-50. [Crossref] [PubMed]
- Mazo V, Sabaté S, Canet J, et al. Prospective external validation of a predictive score for postoperative pulmonary complications. Anesthesiology 2014;121:219-31. [Crossref] [PubMed]
- Severgnini P, Selmo G, Lanza C, et al. Protective mechanical ventilation during general anesthesia for open abdominal surgery improves postoperative pulmonary function. Anesthesiology 2013;118:1307-21. [Crossref] [PubMed]
- Briel M, Meade M, Mercat A, et al. Higher vs lower positive end-expiratory pressure in patients with acute lung injury and acute respiratory distress syndrome: Systematic review and meta-analysis. JAMA 2010;303:865-73. [Crossref] [PubMed]
- Fan E, Del Sorbo L, Goligher EC, et al. An Official American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine Clinical Practice Guideline: Mechanical Ventilation in Adult Patients with Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med 2017;195:1253-63. [Crossref] [PubMed]
- Neto AS, Hemmes SN, Barbas CS, et al. Association between driving pressure and development of postoperative pulmonary complications in patients undergoing mechanical ventilation for general anaesthesia: a meta-analysis of individual patient data. Lancet Respir Med 2016;4:272-80. [Crossref] [PubMed]
- Rothen HU, Sporre B, Engberg G, et al. Airway closure, atelectasis and gas exchange during general anaesthesia. Br J Anaesth 1998;81:681-6. [Crossref] [PubMed]
- Hedenstierna G, Brismar B, Strandberg Å, et al. New aspects on atelectasis during anaesthesia. Clin Physiol 1985;5:127-31. [Crossref] [PubMed]
- Pelosi P, Ball L, de Abreu MG, et al. Better Physiology does not Necessarily Translate Into Improved Clinical Outcome. Turk J Anaesthesiol Reanim 2016;44:165-6. [Crossref] [PubMed]
- Rusca M, Proietti S, Schnyder P, et al. Prevention of atelectasis formation during induction of general anesthesia. Anesth Analg 2003;97:1835-9. [Crossref] [PubMed]
- Pelosi P, Ravagnan I, Giurati G, et al. Positive end-expiratory pressure improves respiratory function in obese but not in normal subjects during anesthesia and paralysis. Anesthesiology 1999;91:1221-31. [Crossref] [PubMed]
- van Kaam AH, Lachmann RA, Herting E, et al. Reducing atelectasis attenuates bacterial growth and translocation in experimental pneumonia. Am J Respir Crit Care Med 2004;169:1046-53. [Crossref] [PubMed]
- Futier E, Constantin JM, Pelosi P, et al. Noninvasive ventilation and alveolar recruitment maneuver improve respiratory function during and after intubation of morbidly obese patients: a randomized controlled study. Anesthesiology 2011;114:1354-63. [Crossref] [PubMed]
- Reinius H, Jonsson L, Gustafsson S, et al. Prevention of atelectasis in morbidly obese patients during general anesthesia and paralysis: a computerized tomography study. Anesthesiology 2009;111:979-87. [Crossref] [PubMed]
- Östberg E, Thorisson A, Enlund M, et al. Positive End-expiratory Pressure Alone Minimizes Atelectasis Formation in Nonabdominal Surgery: A Randomized Controlled Trial. Anesthesiology 2018;128:1117-24. [Crossref] [PubMed]
- Ferrando C, Suarez-Sipmann F, Tusman G, et al. Open lung approach versus standard protective strategies: Effects on driving pressure and ventilatory efficiency during anesthesia-A pilot, randomized controlled trial. PLoS One 2017;12:e0177399. [Crossref] [PubMed]
- Grieco DL, Russo A, Romanò B, et al. Lung volumes, respiratory mechanics and dynamic strain during general anaesthesia. Br J Anaesth 2018;121:1156-65. [Crossref] [PubMed]
- Protti A, Andreis DT, Milesi M, et al. Lung anatomy, energy load, and ventilator-induced lung injury. Intensive Care Med Exp 2015;3:34. [Crossref] [PubMed]
- Futier E, Constantin JM, Paugam-Burtz C, et al. A trial of intraoperative low-tidal-volume ventilation in abdominal surgery. N Engl J Med 2013;369:428-37. [Crossref] [PubMed]
- Guyton AC, Lindsey AW, Abernathy B, et al. Venous return at various right atrial pressures and the normal venous return curve. Am J Physiol 1957;189:609-15. [Crossref] [PubMed]
- Mahmood SS, Pinsky MR. Heart-lung interactions during mechanical ventilation: the basics. Ann Transl Med 2018;6:349. [Crossref] [PubMed]
- Magder S. Bench-to-bedside review: An approach to hemodynamic monitoring-Guyton at the bedside. Crit Care 2012;16:236. [Crossref] [PubMed]
- Ball L, Hemmes SNT, Serpa Neto A, et al. Intraoperative ventilation settings and their associations with postoperative pulmonary complications in obese patients. Br J Anaesth 2018;121:899-908. [Crossref] [PubMed]
- Ladha K, Vidal Melo MF, McLean DJ, et al. Intraoperative protective mechanical ventilation and risk of postoperative respiratory complications: hospital based registry study. BMJ 2015;351:h3646. [Crossref] [PubMed]
- Pelosi P, Ball L. Should we titrate ventilation based on driving pressure? Maybe not in the way we would expect. Ann Transl Med 2018;6:389. [Crossref] [PubMed]
- Ball L, Costantino F, Fiorito M, et al. Respiratory mechanics during general anaesthesia. Ann Transl Med 2018;6:379. [Crossref] [PubMed]
- Marini JJ. Should we titrate positive end-expiratory pressure based on an end-expiratory transpulmonary pressure? Ann Transl Med 2018;6:391. [Crossref] [PubMed]
- Cinnella G, Grasso S, Spadaro S, et al. Effects of recruitment maneuver and positive end-expiratory pressure on respiratory mechanics and transpulmonary pressure during laparoscopic surgery. Anesthesiology 2013;118:114-22. [Crossref] [PubMed]
- Kassis EB, Loring SH, Talmor D. Should we titrate peep based on end-expiratory transpulmonary pressure?-yes. Ann Transl Med 2018;6:390. [Crossref] [PubMed]
- Beitler JR, Sarge T, Banner-Goodspeed VM, et al. Effect of Titrating Positive End-Expiratory Pressure (PEEP) With an Esophageal Pressure-Guided Strategy vs an Empirical High PEEP-Fio2 Strategy on Death and Days Free From Mechanical Ventilation Among Patients With Acute Respiratory Distress Syndrome: A Randomized Clinical Trial. JAMA 2019;321:846-57. [Crossref] [PubMed]
- Sundar S, Novack V, Jervis K, et al. Influence of low tidal volume ventilation on time to extubation in cardiac surgical patients. Anesthesiology 2011;114:1102-10. [Crossref] [PubMed]
- Treschan TA, Kaisers W, Schaefer MS, et al. Ventilation with low tidal volumes during upper abdominal surgery does not improve postoperative lung function. Br J Anaesth 2012;109:263-71. [Crossref] [PubMed]
- Maslow AD, Stafford TS, Davignon KR, et al. A randomized comparison of different ventilator strategies during thoracotomy for pulmonary resection. J Thorac Cardiovasc Surg 2013;146:38-44. [Crossref] [PubMed]
- Shen Y, Zhong M, Wu W, et al. The impact of tidal volume on pulmonary complications following minimally invasive esophagectomy: a randomized and controlled study. J Thorac Cardiovasc Surg 2013;146:1267-74. [Crossref] [PubMed]
- Ge Y, Yuan L, Jiang X, et al. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2013;38:81-5. [Effect of lung protection mechanical ventilation on respiratory function in the elderly undergoing spinal fusion]. [PubMed]
- Valenza F, Chevallard G, Fossali T, et al. Management of mechanical ventilation during laparoscopic surgery. Best Pract Res Clin Anaesthesiol 2010;24:227-41. [Crossref] [PubMed]
- Pelosi P, Croci M, Ravagnan I, et al. The effects of body mass on lung volumes, respiratory mechanics, and gas exchange during general anesthesia. Anesth Analg 1998;87:654-60. [PubMed]
- Cortes-Puentes GA, Keenan JC, Adams AB, et al. Impact of Chest Wall Modifications and Lung Injury on the Correspondence Between Airway and Transpulmonary Driving Pressures. Crit Care Med 2015;43:e287-95. [Crossref] [PubMed]
- Pereira SM, Tucci MR, Morais CCA, et al. Individual Positive End-expiratory Pressure Settings Optimize Intraoperative Mechanical Ventilation and Reduce Postoperative Atelectasis. Anesthesiology 2018;129:1070-81. [Crossref] [PubMed]
- Shapiro HM, Marshall LF. Intracranial pressure responses to PEEP in head-injured patients. J Trauma 1978;18:254-6. [Crossref] [PubMed]
- Robba C, Bragazzi L, Bertuccio A, et al. Effects of Prone Position and Positive End-Expiratory Pressure on Noninvasive Estimators of ICP: A Pilot Study. J Neurosurg Anesthesiol 2017;29:243-250. [Crossref] [PubMed]
- Pulitanò S, Mancino A, Pietrini D, et al. Effects of positive end expiratory pressure (PEEP) on intracranial and cerebral perfusion pressure in pediatric neurosurgical patients. J Neurosurg Anesthesiol 2013;25:330-4. [Crossref] [PubMed]
- Caricato A, Conti G, Della Corte F, et al. Effects of PEEP on the intracranial system of patients with head injury and subarachnoid hemorrhage: the role of respiratory system compliance. J Trauma 2005;58:571-6. [Crossref] [PubMed]
- Mascia L, Grasso S, Fiore T, et al. Cerebro-pulmonary interactions during the application of low levels of positive end-expiratory pressure. Intensive Care Med 2005;31:373-9. [Crossref] [PubMed]
- Liu Z, Liu X, Huang Y, et al. Intraoperative mechanical ventilation strategies in patients undergoing one-lung ventilation: a meta-analysis. Springerplus 2016;5:1251. [Crossref] [PubMed]
- Kiss T, Wittenstein J, Becker C, et al. Protective ventilation with high versus low positive end-expiratory pressure during one-lung ventilation for thoracic surgery (PROTHOR): study protocol for a randomized controlled trial. Trials 2019;20:213. [Crossref] [PubMed]
Cite this article as: Iachi A, Battaglini D, Robba C, Pelosi P, Ball L. Setting intraoperative positive end expiratory pressure: how to be protective. J Emerg Crit Care Med 2020;4:7.


