
Which type of fluid to use perioperatively?
Which type of fluid to use perioperatively?
Fluid management is a fundamental part of the daily clinical practice of anesthesiologists and intensivists. Careful consideration should be given to fluid management with amongst others evaluation of type of fluid, dosing, administration strategy, and potential side-effects. This review focuses on these important aspects of fluid management in perioperative care. First, a short history will be presented and basic physiology and indications for fluid therapy will be discussed. Second, different types of fluid will be discussed, with the exception of blood products. Third, different types of strategies to help guide fluid management will be addressed.
Physiology
History
The British physician William B. O’Shaughnessy (1809–1889) was the first to describe that cholera patients lost a lot of water and electrolytes (“neutral saline ingredients”) from their blood, and he suggested to administer fluids intravenously to restore the normal composition of blood (1). His idea was put to clinical practice by Dr. Thomas A. Latta (1796–1833) who was the first to administer intravenous fluids to a patient (2). During the cholera epidemic in London in 1831, Latta tried to replace the lost fluids and salts from one of his patients by injecting fluids into the larger intestine of the patient and letting the patient drink from time to time. Unfortunately, this act did not appear to result in any benefit for the patient. So, Latta decided to administer the (unsterile!) fluids directly into the circulation of the patient. The patient was given several boluses of a fluid, which was similar to what we now would call ½ Ringer’s lactate, via the basilic vein. During the fluid administration the breathing of the patient immediately became less labored, the pulse became stronger and less frequent, and the patient improved clinically. The woman exclaimed afterwards that she was ‘free from all uneasiness’ and Latta declared her cured (2-5).
In the 19th century the British clinician, physiologist and pharmacologist Sidney Ringer (1835–1910) developed a solution as a fluid replacement for blood loss (6), and late in the 19th century the first sterile fluids were developed (7). In the early 1900’s a 0.9% NaCl solution (also known as “normal” or physiological saline) was developed by Hartog J. Hamburger (1859–1924), a physiological chemist (7). All of these fluids were classified as crystalloids, i.e., solutions containing small molecules (electrolyte salts or glucose) dissolved in water. In 1913 a 0.9% NaCl solution was combined with ‘the purest gelatin’, a colloid, and this solution was administered for the first time to a trauma patient with a successful outcome (8). The theoretical reason for adding colloids to the replacement fluids was that these larger molecules were thought to stay longer within the vasculature and did not vanish into the extravascular space as easily as water and electrolytes.
Developments in fluids were ongoing and in 1932 a lactate buffer was added to Ringer’s solution by Alexis F. Hartmann, pediatrician and chemical pathologist, and Milton J.E. Senn, pediatric psychiatrist (9). They added this buffer to the solution in order to make the composition more similar to that of blood and reduce the amount of chloride, since it became apparent that chloride caused acidosis (10). It took up until the second world war before colloids were frequently used to replace blood losses. Albumin solutions became frequently used replacement fluids for soldiers with trauma on the battlefield (11).
Over the past 200 years the general concept of resuscitating with fluids has stayed the same. Nowadays a differentiation is made between maintenance and replacement fluid therapy. The latter should be given preferably guided by hemodynamic variables, i.e., goal-directed.
Physiology
The goals of perioperative fluid management are to optimize fluid balance, optimize plasma constitution, secure sufficient circulation of blood and thus sufficient oxygen delivery to organs in order to prevent organ dysfunction (12,13). Throughout the perioperative period a broad range of events can lead to insufficient oxygen being delivered to the organs. Overall, the indications to give fluids can be divided into two categories; maintenance and replacement therapy. Maintenance therapy is given to compensate for fasting, i.e., absence of the normal fluid intake. Replacement therapy is needed when hemorrhagic or non-hemorrhagic causes (e.g., fluid shifts or vasodilation) result in absolute or relative volume depletion (14). When resuscitation fluid is administered a cascade of reactions takes place, which will eventually prevent deterioration of organ function. Yet, at every step physiological or pathological mechanisms can obstruct or prevent this process from happening. Figure 1 shows a schematic representation of this process.
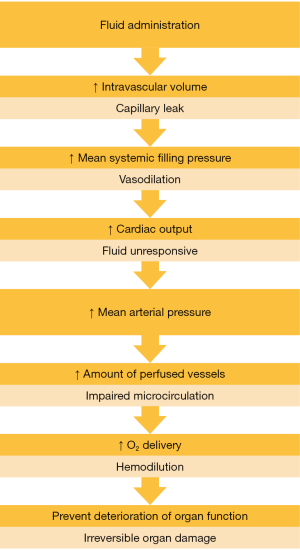
The first step in the cascade begins immediately after the administration of fluids with an increase in intravascular volume. However, capillary leakage can prevent administration of a fluid bolus form increasing intravascular volume (15). The glycocalyx is a layer consisting of proteoglycans on the vascular endothelium that contributes to the barrier function of the endothelium, which plays a very important role in vascular permeability (16). Ischemia and reperfusion, hypoxia, hypervolemia, sepsis, inflammatory activation and hyperglycemia can all cause a loss of integrity of the glycocalyx (17).
When there is no or minimal capillary leakage the intravascular volume increases after fluid administration. The intravascular volume can be separated in stressed and unstressed vascular volume. Unstressed vascular volume is the volume needed to fill the vessels without putting pressure on the vascular wall. The stressed blood volume is the volume which stretches the vascular wall and determines the mean systemic filling pressure (18). When vasodilatation occurs the unstressed part of the vascular volume increases and less volume remains to stretch the vascular wall, which will lead to a decreased mean systemic filling pressure (13).
An increase in mean systemic filling pressure, the driving force behind venous return and thus right ventricular preload, can lead to an increase in cardiac output (CO) if the heart acts on the steep part of the Frank-Starling curve and can cope with the increased preload. The patient is in a preload responsive state which is also called fluid responsive. However, if the heart acts on the horizontal part of the Frank-Starling curve or is failing, CO will not increase following a fluid bolus and the patient is in a preload unresponsive state (19,20). Figure 2 shows a visualization of the Frank-Starling curve for both a normal and a failing ventricle.

An increase in CO has a direct and an indirect effect on the microcirculation. First, an increase in CO directly increases flow through these small vessels, and second CO increases mean arterial pressure, which increases the amount of perfused vessels (21), when there is no microcirculatory impairment. An increased proportion of perfused vessels and increased flow will result in an increased amount of oxygen delivery to the organs (DO2). Yet, this effect might be attenuated by hemodilution caused by the infused fluids (22). As a result of an increased DO2 organ function should be prevented from deterioration, unless hypoxia has been ongoing and has caused structural and irreversible damage to the organ tissue.
Types of fluid
Crystalloids
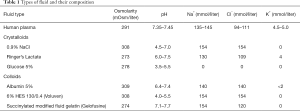
Crystalloid solutions contain minerals or other water-soluble molecules dissolved in water. Frequently used crystalloids are 0.9% NaCl, Ringer’s lactate or glucose 5% (Table 1). Crystalloids and colloids can be balanced or unbalanced. In balanced fluids chloride is partially replaced by other anions. Also, potassium is added to make these solutions more equal to the composition of blood (23). Several studies have assessed the effects of balanced versus unbalanced crystalloid solutions. Unbalanced fluids, especially 0.9% NaCl, can cause hyperchloremia (24) and can lead to hyperchloremic metabolic acidosis (25,26). Hyperchloremia has been associated with increased risk of mortality (OR 2.05; 95% CI: 1.62–2.59) (27) and hyperchloremic acidosis potentially leads to adverse effects such as renal impairment (28) and immune dysfunction (29). These effects can be more pronounced in patients suffering from impaired renal function, due to difficulties in excreting chloride. 0.9% NaCl was also associated with postoperative infection, renal replacement therapy (RRT), blood transfusion and electrolyte imbalances compared to balanced crystalloid solutions, whereas balanced crystalloids had an overall tendency towards a reduced in-hospital mortality (30,31). A study performed in healthy volunteers showed that a fluid bolus of two liters of 0.9% NaCl led to a decrease in renal blood flow and renal cortical perfusion compared to balanced crystalloid solutions (32). Another study showed in addition to these adverse effects that the amount of transfused blood volume was higher and mechanical ventilation times were longer in patients receiving 0.9% NaCl compared to low-chloride containing fluids (24). Two recent trials, the SMART (33) and the SALT-ED (34) trial compared balanced crystalloids and 0.9% NaCl in critically ill and in noncritically ill patients. Both trials showed a lower incidence in major adverse kidney events (MAKE) at 30 days when balanced crystalloids were used (OR 0.91; 95% CI: 0.84–0.99 and OR 0.76; 95% CI: 0.64–0.91 for SMART and SALT-ED, respectively). MAKE includes death from any cause, new RRT, or persistent renal dysfunction. Given the beneficial results, balanced crystalloids are preferred over 0.9% NaCl, as they are more similar to the composition of human plasma (see Table 1) (35,36).
Full table
Colloids
Colloids are mainly used for replacement and not for maintenance of fluid balance. Colloids contain larger molecules than crystalloids such as gelatin, hydroxyethyl starch, dextran or albumin (23) (Table 1). These molecules do not dissolve in plasma so that colloids stay intravascularly longer than crystalloids. When the glycocalyx is intact, it is difficult for colloids to pass this barrier, while crystalloids can pass freely (37,38). In theory, most of the colloids administered will stay intravascular, whereas only 18% of administered balanced crystalloids will (39). Compared to crystalloids, the volume effect of colloids is also about 2–5 times larger (40,41). In addition, there is a difference in colloid osmotic pressure between colloids and crystalloids. Crystalloids generally lower the colloid osmotic pressure and thus decrease the intravascular volume by inducing extravasation, whereas some colloids raise the colloid osmotic pressure and thus increase intravascular volume by causing fluid absorption from extravascular spaces.
Albumin is made using pooled fractionated blood from donors added to a crystalloid solution. This is a very time-consuming and expensive process depending completely on human blood donors and thus semisynthetic colloids were developed. Albumin comes in different solutions: including a 4% hypo-oncotic, a 5% iso-oncotic and a 20% hyper-oncotic solution, the latter with a fivefold higher volume effect (plasma expander). The SAFE trial randomized 7,000 ICU patients in need of fluid replacement to receive either albumin or saline and found no difference in survival at 28 days (42). More recently, the ALBIOS trial reproduced this finding when albumin was compared to crystalloids in patients with sepsis (43).
Hydroxy-ethyl starch (HES) is an example of semi-synthetic colloids (44). HES is derived from corn starch or potato starch, and has undergone several modifications (from hetastarch via pentastarch to tetrastarch) with an improved safety profile while maintaining its efficient volume effect. Modern HES containing products are also available as balanced solutions. HES products were widely used in the perioperative setting as well as in the ICU for volume replacement. However, several large randomized controlled trials (RCT) performed in critically ill patients with sepsis raised concerns about the safety of HES (45-47). These studies showed an increase in acute kidney injury (AKI), RRT, adverse events and mortality after the use of HES, as well as disturbances in coagulation (45-47). As a consequence of these large studies, the indication for HES was redefined. Furthermore, although all of these studies were performed in critically ill patients, the safety and efficacy of HES products has to be re-evaluated in the perioperative setting.
Gelatins are one of the oldest synthetic colloids (8). Since they were developed a long time ago, proper safety studies have never been conducted. Authors of a systematic review with meta-analysis performed in 2012 concluded that “despite over 60 years of clinical practice, the safety and efficacy of gelatin cannot be reliably assessed”, and the authors recommended to investigate and establish such safety (48).
An in vitro study showed that administration of different colloids led to an increase in clotting time and a decrease in clot firmness compared to administration of crystalloids (49). Possible reasons include the “coating” of platelets and hemodilution. 6% HES 130/0.4 showed higher reduction in fibrinogen and thrombin concentrations. Patients receiving HES compared to albumin and gelatin solutions had overall worse outcomes (50). A systematic review with meta-analysis assessed the difference in outcomes between crystalloids and colloids for critically ill, trauma and surgical patients and showed no increased risk for patients undergoing cardiac surgery which had received colloids. A sub-analysis of the group of patients which underwent general surgery also showed no increased risk of mortality in the HES group (51).
Since multiple studies showed adverse events after the use of HES, the European Medicines Agency (EMA) restricted the use of HES in 2013 and redefined the indication to hypovolemia due to blood loss, which meant that there no longer was an indication for HES products in critically ill patients. The EMA ordered the conduct of a drug utilization study which showed that HES was still sometimes given to critically ill patients which led them to consider to take HES from the market completely. An expert meeting recommended not to withdraw HES for several reasons: (I) withdrawing HES from the market might cause unmet medical needs, especially when replacement with crystalloids alone is insufficient, (II) the use of albumin will increase which is very costly and dependent on human plasma donors (52). The EMA decided to add further risk minimization measures to prevent the use of HES in patients at risk. An e-learning on the use of HES according to the European Product Information has been developed and is available online at the ESA Academy. Furthermore, on request of the EMA, the PHOENICS and TETHYS trials are currently being conducted and aim to look at safety and efficacy of use of HES in patients undergoing high-risk abdominal surgery (NCT03278548) and trauma patients (NCT03338218), respectively.
Fluid management
Hypovolemic versus fluid overload
Both hypovolemia and hypervolemia are associated with postoperative complications, although it is very difficult to strictly maintain normovolemia as there is no clear definition of and no easy way to monitor normovolemia (53). Hypovolemia is associated with organ hypoperfusion, systemic inflammatory response syndrome, sepsis, multi-organ failure and even mortality (53). Hypervolemia on the other hand is associated with tissue edema, ileus, postoperative nausea and vomiting, pulmonary complications and an increase in cardiac demand (53). A proof-of-concept study conducted in 48 surgical patients who were postoperatively admitted to the ICU showed that weight gain during surgery was associated with increased mortality. The group with less than 10% body weight gain (n=29) had a mortality rate of 10%, the group that gained 10–20% body weight (n=16) had a mortality of 19%, and in the group that gained more than 20% body weight (n=3) all patients died (54). A study including patients undergoing cardiac surgery also showed that fluid overload was associated with mortality (OR 1.59; 95% CI: 1.18–2.14) (55).
A large retrospective study of 90,000 patients undergoing non-cardiac surgery showed an association between perioperatively administered fluid volume and postoperative outcomes. The outcomes 30-day mortality, postoperative respiratory complications and AKI all showed a U-shaped curve pattern indicating that there might be an optimal level (sweet spot) of fluid administration. The same results were found for the outcomes length of hospital stay and total hospital cost (56).
Hypovolemia as well as fluid overload cause adverse postoperative outcomes. Several strategies for perioperative fluid management exist and numerous studies have shown that postoperative outcomes are influenced by perioperative fluid management strategies (57-60). Current strategies for fluid management include restrictive fluid therapy, liberal fluid therapy, and perioperative goal-directed therapy (PGDT).
Restrictive versus liberal fluid management
Since 1961 a liberal fluid strategy was promoted due to the observation that during major surgery a reduction in extracellular fluid occurred, presumably by internal fluid redistribution (61). In the following years no proper evaluation of liberal fluid management was performed and it took a few decades before the adverse outcomes of liberal fluid management were realized (62). More knowledge of the harmful effects of fluid overload caused several comparison studies to assess if a restrictive fluid strategy would be more beneficial for patient outcomes. One of the difficulties of comparing liberal and restrictive fluid therapy is the fact that these terms are not clearly defined, i.e., what is considered to be liberal fluid therapy in one study could be considered restrictive fluid therapy in another. A systematic review with meta-analysis assessed the difference in postoperative morbidity between a restrictive and a liberal fluid therapy approach and found a range of 998 to 2,740 mL for a restrictive approach, whereas a liberal approach ranged from 2,750 to 5,388 mL (53). In this meta-analysis three studies showed a beneficial outcome when a restrictive approach was used in major abdominal surgery: length of stay, return of bowel function and overall decrease in complications (63-65). Two studies included in the meta-analysis showed no difference in outcome between a restrictive or a liberal approach (66,67). One study, conducted in orthopedic patients, showed improved pulmonary function and coagulation in the patient group receiving the liberal approach (68). Another study found improved pulmonary function, as well as improved post-operative hypoxemia and reduced vasoactive hormonal response in the liberal approach, and the total number of complications was reduced with the liberal approach (69). Another systematic review with meta-analysis included twelve studies comparing liberal and restrictive fluid management (70). The included studies overlap with the abovementioned meta-analysis (53), however, an additional five studies were included and the primary outcome was mortality instead of postoperative morbidity (71-75). Pneumonia and pulmonary edema occurred more often in the group receiving liberal fluid therapy, as length of hospital stay and return of bowel function took longer in the liberal fluid management groups (70).
In the RELIEF trial, 3,000 patients undergoing abdominal surgery were included and allocated to a restrictive or a liberal fluid therapy (76). The restrictive group received a fluid bolus of 5 mL kg-1 during induction of anesthesia, after which the dose was reduced to 5 mL kg-1 h-1 until the end of surgery and up until 24 h after surgery a dose of 0.8 mL kg-1 h-1 was given. For the liberal fluid strategy, the dosages were 10 mL kg-1 h-1, 8 mL kg-1 h-1 and 1.5 mL kg-1 h-1, respectively. No difference was found for the primary outcome, which was disability free-survival one year after surgery. Furthermore, the incidence of AKI was slightly higher within the restrictive strategy, as well as RRT and surgical site infections.
In summary, restrictive and liberal fluid strategies are hard to compare since there are no clear definitions of how much fluid is administered in each of these strategies, leading to a lack of clear cut-off values separating both regimens. The optimal amount of volume needs to be administered at the right moments and only when a patient is in need of fluids, which can be detected by advanced hemodynamic monitoring. For this purpose, PGDT was developed, aiming at a more personalized approach.
Perioperative goal-directed therapy
The principle of PGDT is to assess fluid responsiveness and cardiac function using advanced hemodynamic monitoring in order to optimize oxygen delivery to the tissues (77). The obtained hemodynamic variables are used in an algorithm, which helps the anesthesiologist to make sure that the hemodynamic status of the patient is optimized. In these algorithms, fluid management is usually the primary intervention, which is however often combined with titration of vasopressors and inotropes. Figure 3 shows an example of a PGDT algorithm.
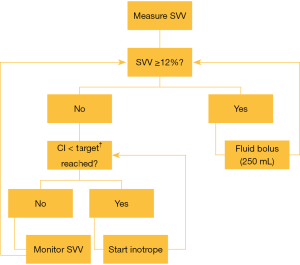
The introduction of the pulmonary artery catheter (PAC) in the 1980s led to the first trials using hemodynamic variables to guide perioperative therapy. Researchers noticed that survivors of trauma surgery often had ‘supranormal’ values of hemodynamic values such as cardiac index (CI) and DO2 (78). An early systematic review with meta-analysis showed that obtaining these ‘supranormal’ hemodynamic values was only beneficial when started before the onset of organ failure and in patients with an expected mortality >20% (79). After decades of research and after development of less invasive hemodynamic monitors, dynamic hemodynamic variables such as stroke volume variation (SVV) and pulse pressure variations (PPV) were implemented in PGDT strategies as targets to help guiding fluid therapy.
Over the last couple of decades, a large number of studies have shown beneficial patient outcomes using PGDT. A recent systematic review with meta-analysis, including 95 RCTs, showed that PGDT reduced mortality (OR 0.66; 95% CI: 0.50–0.87). However, no mortality benefit was found in fluid-based-only algorithms, in cardiac surgery patients, or in non-elective surgeries. PGDT did also decrease the risk of AKI, pneumonia, wound infection and the hospital length of stay (80). Another systematic review with meta-analysis, including 37 RCTs, was conducted with the aim to assess the difference in PGDT using only fluids guided by dynamic variables versus standard (i.e., unmonitored) fluid therapy. The group where PGDT using only fluids was used showed a lower serum lactate than the standard fluid therapy group. Yet, no difference was found in short-term mortality, morbidity, and length of stay. When, however, the use of fluids was combined with additional optimization goals such as CO or CI, there was a reduction in short-term mortality, morbidity, serum lactate concentration, cardiopulmonary complications, heart failure/cardiovascular dysfunction, acute lung injury/acute respiratory distress syndrome, pneumonia, and length of stay in the ICU, and length of hospital stay (81).
Although there is a considerable evidence in favor of PGDT, it is still not commonly used in clinical practice. One of the reasons is heterogeneity in PGDT studies and all components of the PGDT algorithms (82). Therefore, the 5 ‘T’s’ have been suggested to help implement PGDT in clinical practice: target population, timing of the intervention, type of intervention, target variable, and target value (83). PGDT should be aimed at high-risk patients as target population. High risk patients can have patient specific features that increase risk of complications or are scheduled for a high-risk procedure. PGDT should be started early in the perioperative phase, preferably before anesthetic induction (80). The right choice of intervention should be made. This review aimed to focus solely on perioperative fluid use. Nonetheless, in order to provide best practice a complete PGDT algorithm should contain vasopressors and inotropes as well. The right variable should be targeted, preferably hemodynamic variables reflecting blood flow (81). Within the target variable the right target values must be pursued, and this goal might be quite difficult to achieve since the use of dynamic variables is limited to certain circumstances. Finally, blood pressure and CO targets should be individualized to patient specific baseline values (84).
Currently two large RCTs are conducted to enrich the evidence on PGDT. In the first one, the Fluid Optimisation in Emergency LAparatomy trial (FLO-ELA), nearly 8,000 patients will be included who are undergoing emergency bowel surgery in order to assess mortality when a cardiac-output guided hemodynamic therapy strategy is used (www.floela.org). In the second one, Optimisation of Peri-operaTive CardIovascular Management to Improve Surgical outcome II trial (OPTIMISE-II), 2,502 patients will be included who are undergoing elective major gastro-intestinal surgery. In this study, a cardiac-output guided hemodynamic maximization strategy with infusion of low-dose inotropes is compared to standard care (www.optimiseii.org). A third study on the effect of early goal-directed therapy on complications, quality of life and healthcare costs after high-risk abdominal surgery has recently stopped recruiting patients and is currently under analysis (85).
Future perspectives
Closed-loop hemodynamic management
One of the most important future directions of perioperative fluid management is the development of closed-loop fluid administration systems. Closed-loop fluid management makes use of dynamic hemodynamic variables, which aid in the assessment of fluid responsiveness. Minimally or non-invasive hemodynamic monitors can be used to assess fluid responsiveness and the closed-loop systems then delivers a fluid bolus if the patient is considered fluid responsive. The hemodynamic effect of this fluid bolus is determined and this information is used in the decision to give another fluid bolus (86). Several studies have been performed using this concept and a novel closed-loop system was first tested using a computer simulation of hemorrhage scenarios (87,88), followed by an animal study, after which the system was studied in patients (89). These studies showed that closed-loop systems were more precise and performed more consistently compared to anesthesiologists. The closed-loop system was tested in moderate and high-risk patients for compliance to a PGDT strategy. The closed-loop system was successful in keeping patients in a preload independent state (PPV <13%) and with a CI >2.5 L min-1 m-2 for at least 85% of the time (90,91).
Personalized hemodynamic management
A general problem of current fluid management is that patients are treated according to a one-size-fits-all approach. However, there is a considerable amount of variability between individual patients and thus PGDT should be individualized with the use of hemodynamic monitoring in order to assess fluid responsiveness and optimize blood flow. Subsequently, PGDT should be personalized with personal targets adjusted to several biometric factors (84). Age, bodyweight and height are independently associated with CO (92) and stroke volume decreases with age (93,94). Hemodynamic management could be optimized when individual ‘normal’ baseline values are obtained for each patient preoperatively.
Conclusions
There are currently no universal standards for volume management. Crystalloids belong to the first-line agents for fluid therapy (both maintenance and replacement). Colloids are indicated for the treatment of severe hypovolemia/hypovolemic shock (e.g., due to major bleeding) and can be used safely for short terms in the perioperative period when the dose limitations are respected. It is very complex to determine the best strategy for fluid administration and current evidence remains inconclusive. Most important, fluids should be administered with the same caution that is used with any intravenous drug (95).
Acknowledgments
None.
Footnote
Conflicts of Interest: TWLS received research grants and honoraria from Edwards Lifesciences (Irvine, CA, USA) and Masimo Inc. (Irvine, CA, USA) for consulting and lecturing and from Pulsion Medical Systems SE (Feldkirchen, Germany) for lecturing. Other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- O’Shaughnessy WB. Experiments on the blood in cholera. Lancet 1831;1:490. [Crossref]
- Latta TA. Relative to the treatment of cholera by the copius injection of aqueous and saline fluids into the veins. Lancet 1832;2:274-7.
- Baskett TF. William O’Shaughnessy, Thomas Latta and the origins of intravenous saline. Resuscitation 2002;55:231-4. [Crossref] [PubMed]
- MacGillivray N. Dr. Latta of Leith: pioneer in the treatment of cholera by intravenous saline infusion. J R Coll Physicians Edinb 2006;36:80-5. [PubMed]
- Marik PE. Fluid therapy in 2015 and beyond: the mini-fluid challenge and mini-fluid bolus approach. Br J Anaesth 2015;115:347-9. [Crossref] [PubMed]
- Ringer S. Regarding the action of hydrate of soda, hydrate of ammonia, and hydrate of potash on the ventricle of the frog’s heart. J Physiol 1882;3:195-202. [Crossref] [PubMed]
- Awad S, Allison SP, Lobo DN. The history of 0.9% saline. Clin Nutr 2008;27:179-88. [Crossref] [PubMed]
- Hogan J. The intravenous use of colloidal (gelatin) solutions in shock. JAMA 1915;64:721. [Crossref]
- Hartmann AF, Senn MJE. Studies in the metabolism of sodium r-lactate. I. Response of normal human subjects to the intrevenous injection of sodium r-lactate. J Clin Invest 1932;11:327-35. [Crossref] [PubMed]
- Lee JA. Sydney Ringer (1834-1910) and Alexis Hartmann (1898-1964). Anaesthesia 1981;36:1115-21. [Crossref] [PubMed]
- McClelland DB. Human albumin solutions. BMJ 1990;300:35-7. [Crossref] [PubMed]
- Voldby AW, Brandstrup B. Fluid therapy in the perioperative setting - a clinical review. J Intensive Care 2016;4:27. [Crossref] [PubMed]
- Monnet X, Teboul JL. My patient had received fluid. How to assess its efficacy and side effects? Ann Intensive Care 2018;8:54. [Crossref] [PubMed]
- Hoorn EJ. Intravenous fluids: balancing solutions. J Nephrol 2017;30:485-92. [Crossref] [PubMed]
- Aya HD, Rhodes A, Chis Ster I, et al. Hemodynamic effect of different doses of fluids for a fluid challenge: a quasi-randomized controlled study. Crit Care Med 2017;45:e161-8. [Crossref] [PubMed]
- Reitsma S, Slaaf DW, Vink H, et al. The endothelial glycocalyx: composition, functions, and visualization. Pflugers Arch 2007;454:345-59. [Crossref] [PubMed]
- Becker BF, Jacob M, Leipert S, et al. Degradation of the endothelial glycocalyx in clinical settings: searching for the sheddases. Br J Clin Pharmacol 2015;80:389-402. [Crossref] [PubMed]
- Magder S. Volume and its relationship to cardiac output and venous return. Crit Care 2016;20:271. [Crossref] [PubMed]
- Sequeira V, van der Velden J. Historical perspective on heart function: the Frank-Starling Law. Biophys Rev 2015;7:421-47. [Crossref] [PubMed]
- Patterson SW, Starling EH. On the Mechanical Factors Which Determine the Output of the Ventricles. J Physiol 1914;48:357-79. [Crossref] [PubMed]
- Ospina-Tascon G, Neves AP, Occhipinti G, et al. Effects of fluids on microvascular perfusion in patients with severe sepsis. Intensive Care Med 2010;36:949-55. [Crossref] [PubMed]
- Monnet X, Julien F, Ait-Hamou N, et al. Lactate and venoarterial carbon dioxide difference/arterial-venous oxygen difference ratio, but not central venous oxygen saturation, predict increase in oxygen consumption in fluid responders. Crit Care Med 2013;41:1412-20. [Crossref] [PubMed]
- Boer C, Bossers SM, Koning NJ. Choice of fluid type: physiological concepts and perioperative indications. Br J Anaesth 2018;120:384-96. [Crossref] [PubMed]
- Krajewski ML, Raghunathan K, Paluszkiewicz SM, et al. Meta-analysis of high- versus low-chloride content in perioperative and critical care fluid resuscitation. Br J Surg 2015;102:24-36. [Crossref] [PubMed]
- Myles PS, Andrews S, Nicholson J, et al. Contemporary Approaches to Perioperative IV Fluid Therapy. World J Surg 2017;41:2457-63. [Crossref] [PubMed]
- Burdett E, Dushianthan A, Bennett-Guerrero E, et al. Perioperative buffered versus non-buffered fluid administration for surgery in adults. Cochrane Database Syst Rev 2012;12:CD004089. [PubMed]
- McCluskey SA, Karkouti K, Wijeysundera D, et al. Hyperchloremia after noncardiac surgery is independently associated with increased morbidity and mortality: A propensity-matched cohort study. Anesth Analg 2013;117:412-21. [Crossref] [PubMed]
- Hadimioglu N, Saadawy I, Saglam T, et al. The effect of different crystalloid solutions on acid-base balance and early kidney function after kidney transplantation. Anesth Analg 2008;107:264-9. [Crossref] [PubMed]
- Kellum JA, Song M, Li J. Science review: Extracellular acidosis and the immune response: Clinical and physiologic implications. Crit Care 2004;8:331-6. [Crossref] [PubMed]
- Orbegozo Cortés D, Rayo Bonor A, Vincent JL. Isotonic crystalloid solutions: A structured review of the literature. Br J Anaesth 2014;112:968-81. [Crossref] [PubMed]
- Shaw AD, Bagshaw SM, Goldstein SL, et al. Major complications, mortality, and resource utilization after open abdominal surgery: 0.9% saline compared to plasma-lyte. Ann Surg 2012;255:821-9. [Crossref] [PubMed]
- Chowdhury AH, Cox E, Francis S, et al. A randomized, controlled, double-blind crossover study on the effects of 2-L infusions of 0.9% salin and plasma-lyte 148 on renal blood flow velocity and renal cortical tissue perfusion in healthy volunteers. Ann Surg 2012;256:18-24. [Crossref] [PubMed]
- Semler MW, Self W, Wanderer J, et al. Balanced crystalloid versus saline in critically ill adults. N Engl J Med 2018;378:829-39. [Crossref] [PubMed]
- Self WH, Semler M, Wanderer J, et al. Balanced crystalloid versus saline in noncritically ill adults. N Engl J Med 2018;378:819-28. [Crossref] [PubMed]
- Miller TE, Roche AM, Mythen M. Fluid management and goal-directed therapy as an adjunct to Enhanced Recovery After Surgery (ERAS). Can J Anesth 2015;62:158-68. [Crossref] [PubMed]
- Feldheiser A, Pavlova V, Bonomo T, et al. Balanced crystalloid compared with balanced colloid solution using a goal-directed haemodynamic algorithm. Br J Anaesth 2013;110:231-40. [Crossref] [PubMed]
- Rehm M, Haller M, Orth V, et al. Changes in Intravascular Volume During Acute Normovolemic Hemodilution and Intraoperative Retransfusion in Patients with Radical Hysterectomy. Anesthesiology 2000;92:657-64. [Crossref] [PubMed]
- Chappell D, Jacob M. Role of the glycocalyx in fluid management: Small things matter. Best Pract Res Clin Anaesthesiol 2014;28:227-34. [Crossref] [PubMed]
- Frost P. Intravenous fluid therapy in adult inpatients. BMJ 2015;350:g7620. [Crossref] [PubMed]
- McIlroy DR, Kharasch E. Acute intravascular volume expansion with rapidly administered crystalloid or colloid in the setting of moderate hypovolemia. Anesth Analg 2003;96:1572-7. [Crossref] [PubMed]
- Jacob M, Chappell D, Hofmann-Kiefer K, et al. The intravascular volume effect of Ringer’s lactate is below 20%: A prospective study in humans. Crit Care 2012;16:R86. [Crossref] [PubMed]
- Finfer S, Bellomo R, Boyce N, et al. A Comparison of Albumin and Saline for Fluid Resuscitation in the Intensive Care Unit. N Engl J Med 2004;350:2247-56. [Crossref] [PubMed]
- Caironi P, Tognoni G, Masson S, et al. Albumin replacement in patients with severe sepsis or septic shock. N Engl J Med 2014;370:1412-21. [Crossref] [PubMed]
- Finfer S, Liu B, Taylor C, et al. Resuscitation fluid use in critically ill adults: an international cross-sectional study in 391 intensive care units. Crit Care 2010;14:R185. [Crossref] [PubMed]
- Brunkhorst FM, Engel C, Bloos F, et al. Intensive insulin therapy and pentastarch resuscitation in severe sepsis. N Engl J Med 2008;358:125-39. [Crossref] [PubMed]
- Perner A, Haase N, Guttormsen A, et al. Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med 2012;367:124-34. [Crossref] [PubMed]
- Myburgh JA, Finfer S, Bellomo R, et al. Hydroxyethyl Starch or Saline for Fluid Resuscitation in Intensive Care. N Engl J Med 2012;367:1901-11. [Crossref] [PubMed]
- Thomas-Rueddel DO, Vlasakov V, Reinhart K, et al. Safety of gelatin for volume resuscitation-a systematic review and meta-analysis. Intensive Care Med 2012;38:1134-42. [Crossref] [PubMed]
- Fenger-Eriksen C, Tønnesen E, Ingerslev J, et al. Mechanisms of hydroxyethyl starch-induced dilutional coagulopathy. J Thromb Haemost 2009;7:1099-105. [Crossref] [PubMed]
- Niemi TT, Kuitunen AH. Artificial colloids impair haemostasis. An in vitro study using thromboelastometry coagulation analysis. Acta Anaesthesiol Scand 2005;49:373-8. [Crossref] [PubMed]
- Qureshi SH, Rizvi SI, Patel NN, et al. Meta-analysis of colloids versus crystalloids in critically ill, trauma and surgical patients. Br J Surg 2016;103:14-26. [Crossref] [PubMed]
- Annane D, Fuchs-Buder T, Zoellner C, et al. EMA recommendation to suspend HES is hazardous. Lancet 2018;391:736-8. [Crossref] [PubMed]
- Bundgaard-Nielsen M, Secher NH, Kehlet H. “Liberal” vs. “restrictive” perioperative fluid therapy - A critical assessment of the evidence: Review Article. Acta Anaesthesiol Scand 2009;53:843-51. [Crossref] [PubMed]
- Lowell JA, Schifferdecker C, Driscoll D, et al. Postoperative fluid overload: Not a beningn problem. Crit Care Med 1990;18:728-33. [Crossref] [PubMed]
- Stein A, de Souza LV, Belettini CR, et al. Fluid overload and changes in serum creatinine after cardiac surgery: Predictors of mortality and longer intensive care stay. A prospective cohort study. Crit Care 2012;16:R99. [Crossref] [PubMed]
- Shin CH, Long ADR, Mclean AD, et al. Effects of Intraoperative Fluid Management on Postoperative Outcomes. Ann Surg 2018;267:1084-92. [Crossref] [PubMed]
- Holte K, Kehlet H. Fluid Therapy and Surgical Outcomes in Elective Surgery: A Need for Reassessment in Fast-Track Surgery. J Am Coll Surg 2006;202:971-89. [Crossref] [PubMed]
- Chappell D, Hofmann-Kiefer K, Conzen P, et al. A Rational Approach to Perioperative Fluid Management. Anesthesiology 2008;109:723-40. [Crossref] [PubMed]
- Brandstrup B. Fluid therapy for the surgical patient. Best Pract Res Clin Anaesthesiol 2006;20:265-83. [Crossref] [PubMed]
- Holte K, Kehlet H. Compensatory fluid administration for preoperative dehydration - does it improve outcome? Acta Anaesthesiol Scand 2002;46:1089-93. [Crossref] [PubMed]
- Shires T, Williams J, Brown F. Acute change in extracellular fluids associated with major surgical procedures. Ann Surg 1961;154:803-10. [Crossref] [PubMed]
- Schol PBB, Terink IM, Lancé MD, et al. Liberal or restrictive fluid management during elective surgery: a systematic review and meta-analysis. J Clin Anesth 2016;35:26-39. [Crossref] [PubMed]
- Lobo DN, Bostock KA, Neal KR, et al. Effect of salt and water balance on recovery of gastrointestinal function after elective colonis resection: a randomised controlled trial. Lancet 2002;359:1812-8. [Crossref] [PubMed]
- Brandstrup B, Tønnesen H, Beier-Holgersen R, et al. Effects of Intravenous Fluid Restriction on Postoperative Complications: Comparison of Two Perioperative Fluid Regimens - A Randomized Assessor-Blinded Multicenter Trial. Ann Surg 2003;238:641-8. [Crossref] [PubMed]
- Nisanevich V, Felsenstein I, Almogy G, et al. Effect of Intraoperative Fluid Management on Outcome After Intraabdominal Surgery. Anesthesiology 2005;103:25-32. [Crossref] [PubMed]
- Kabon B, Sessler DI, Kurz A, et al. Effect of Intraoperative Goal-directed Balanced Crystalloid versus Colloid Administration on Major Postoperative Morbidity: A Randomized Trial. Anesthesiology 2019;130:728-44. [Crossref] [PubMed]
- MacKay G, Fearon K, McConnachie A, et al. Randomized clinical trial of the effect of postoperative intravenous fluid restriction on recovery after elective colorectal surgery. Br J Surg 2006;93:1469-74. [Crossref] [PubMed]
- Holte K, Kristense B, Valentiner L, et al. Liberal versus restrictive fluid management in knee arthroplasty: a randomized, double-blind study. Anesth Analg 2007;105:465-74. [Crossref] [PubMed]
- Holte K, Foss NB, Andersen J, Valentiner L, Lund C, Bie P, et al. Liberal or restrictive fluid administration in fast-track colonic surgery: A randomized, double-blind study. Br J Anaesth 2007;99:500-8. [Crossref] [PubMed]
- Corcoran T, Rhodes JEJ, Clarke S, et al. Perioperative fluid management strategies in major surgery: A stratified meta-analysis. Anesth Analg 2012;114:640-51. [Crossref] [PubMed]
- González-Fajardo JA, Mengibar L, Brizuela JA, et al. Effect of Postoperative Restrictive Fluid Therapy in the Recovery of Patients with Abdominal Vascular Surgery. Eur J Vasc Endovasc Surg 2009;37:538-43. [Crossref] [PubMed]
- McArdle GT, McAuley DF, McKinley A, et al. Preliminary results of a prospective randomized trial of restrictive versus standard fluid regime in elective open abdominal aortic aneurysm repair. Ann Surg 2009;250:28-34. [Crossref] [PubMed]
- Vermeulen H, Hofland J, Legemate DA, et al. Intravenous fluid restriction after major abdominal surgery: A randomized blinded clinical trial. Trials 2009;10:50. [Crossref] [PubMed]
- Muller S, Zalunardo MP, Hubner M, et al. A Fast-Track Program Reduces Complications and Length of Hospital Stay After Open Colonic Surgery. Gastroenterology 2009;136:842-7. [Crossref] [PubMed]
- Hübner M, Schäfer M, Demartines N, et al. Impact of restrictive intravenous fluid replacement and combined epidural analgesia on perioperative volume balance and renal function within a fast track program. J Surg Res 2012;173:68-74. [Crossref] [PubMed]
- Myles PS, Bellomo R, Corcoran T, et al. Restrictive versus Liberal Fluid Therapy for Major Abdominal Surgery. N Engl J Med 2018;378:2263-74. [Crossref] [PubMed]
- Kaufmann T, Saugel B, Scheeren TWL. Perioperative goal-directed therapy - what is the evidence? Best Pract Res Clin Anaesthesiol 2019. Epub ahead of print. [Crossref] [PubMed]
- Shoemaker WC, Appel PL, Kram HB, et al. Prospective trial of supranormal values of survivors as therapeutic goals in high-risk surgical patients. Chest 1988;94:1176-86. [Crossref] [PubMed]
- Kern JW, Shoemaker WC. Meta-analysis of hemodynamic optimization in high-risk patients. Crit Care Med 2002;30:1686-92. [Crossref] [PubMed]
- Chong MA, Wang Y, Berbenetz NM, et al. Does goal-directed haemodynamic and fluid therapy improve peri-operative outcomes? Eur J Anaesthesiol 2018;35:469-83. [PubMed]
- Deng QW, Tan WC, Zhao BC, et al. Is goal-directed fluid therapy based on dynamic variables alone sufficient to improve clinical outcomes among patients undergoing surgery? A meta-analysis. Crit Care 2018;22:298. [Crossref] [PubMed]
- Kaufmann T, Clement RP, Scheeren TWL, et al. Perioperative goal-directed therapy: A systematic review without meta-analysis. Acta Anaesthesiol Scand. 2018;62:1340-55. [Crossref] [PubMed]
- Saugel B, Kouz K, Scheeren TWL. The ‘5 Ts’ of perioperative goal-directed haemodynamic therapy. Br J Anaesth 2019;123:103-7. [Crossref] [PubMed]
- Saugel B, Vincent JL, Wagner JY. Personalized hemodynamic management. Curr Opin Crit Care 2017;23:334-41. [Crossref] [PubMed]
- Montenij L, De Waal E, Frank M, et al. Influence of early goal-directed therapy using arterial waveform analysis on major complications after high-risk abdominal surgery: Study protocol for a multicenter randomized controlled superiority trial. Trials 2014;15:360. [Crossref] [PubMed]
- Rinehart J, Liu N, Alexander B, et al. Closed-loop systems in anesthesia: Is there a potential for closed-loop fluid management and hemodynamic optimization? Anesth Analg 2012;114:130-43. [Crossref] [PubMed]
- Rinehart J, Chung E, Canales C, et al. Intraoperative stroke volume optimization using stroke volume, arterial pressure, and heart rate: Closed-loop (learning intravenous resuscitator) versus anesthesiologists. J Cardiothorac Vasc Anesth 2012;26:933-9. [Crossref] [PubMed]
- Rinehart J, Alexander B, Manach YL, et al. Evaluation of a novel closed-loop fluid-administration system based on dynamic predictors of fluid responsiveness: An in silico simulation study. Crit Care 2011;15:R278. [Crossref] [PubMed]
- Rinehart J, Lee C, Canales C, et al. Closed-Loop Fluid Administration COmpared to Anesthesiologist Management for Hemodynamic Optimization and Resuscitation During Surgery. Anesth Analg 2013;117:1119-29. [Crossref] [PubMed]
- Joosten A, Huynh T, Suehiro K, et al. Goal-Directed fluid therapy with closed-loop assistance during moderate risk surgery using noninvasive cardiac output monitoring: A pilot study. Br J Anaesth 2015;114:886-92. [Crossref] [PubMed]
- Rinehart J, Le Manach Y, Douiri H, et al. First closed-loop goal directed fluid therapy during surgery: A pilot study. Ann Fr Anesth Reanim 2014;33:e35-41. [Crossref] [PubMed]
- Saugel B, Mair S, Götz SQ, et al. Indexation of cardiac output to biometric parameters in critically ill patients: A systematic analysis of a transpulmonary thermodilution-derived database. J Crit Care 2015;30:957-62. [Crossref] [PubMed]
- Cain PA, Ahl R, Hedstrom E, et al. Age and gender specific normal values of left ventricular mass, volume and function for gradient echo magnetic resonance imaging: A cross sectional study. BMC Med Imaging 2009;9:2. [Crossref] [PubMed]
- Kou S, Caballero L, Dulgheru R, et al. Echocardiographic reference ranges for normal cardiac chamber size: Results from the NORRE study. Eur Heart J Cardiovasc Imaging 2014;15:680-90. [Crossref] [PubMed]
- Myburgh JA, Mythen MG. Resuscitation Fluids. N Engl J Med 2013;369:1243-51. [Crossref] [PubMed]
Cite this article as: de Keijzer IN, Kaufmann T, Scheeren TW. Which type of fluid to use perioperatively? J Emerg Crit Care Med 2019;3:51.


