Infections in elderly intensive care unit patients
Introduction
Elderly population is increasing in the developed world, both in terms of absolute numbers and proportion of the entire population. In the UK, from 1982 to 2002, life expectancy at the age of 80 increased from 5.8 years to 7.2 years in men and from 7.5 to 8.7 years in women. Trends are similar in other countries where elderlies represent more than 10% of ICU admissions (1-3). Infection, either as a cause of admission to intensive care units (ICU) or as complication during the ICU stay, represents a major health-care issue. It is estimated that elderly adults account for 60% of all ICU days, indicating a prolonged and frequently complicated length of stay (LOS) in an environment with increased rates of nosocomial infections owing to the severity of illness, multiplicity of invasive procedures and complexity of antibiotic and non-antibiotic treatments (3-6). Large epidemiological studies on outcomes of elderly adults after ICU admission are limited. Most data derive from small reports usually from single centers, describing a particular infection or syndrome, while few studies are available on ICU acquired infections (7-14). The most frequent infections in the ICU are respiratory infections, followed by urinary tract (UTIs) and bloodstream infections (BSIs) including catheter related infections (CRIs) (15,16). The definition of elderly patient is a matter of debate. The adult of 65 years old is considered elderly due to insurance reasons. However, aging is a continuous and host-specific process, therefore the subgroups of 65–74 (young-old elderlies), 75–84 (old elderlies) and >85 (old-old or oldest old elderlies) have been used (4,17-30).
Immune function in elderlies
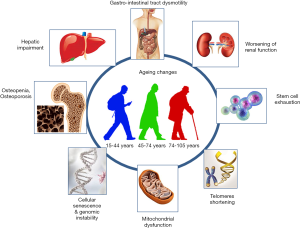
The reasons why elderly individuals are vulnerable to life-threatening sepsis are multifactorial, including the pathophysiology of aging, the age-related reduction of organ function as well as the comorbidities and the polypharmacy (Table 1, Figure 1) (31). The modern definition of modifications in the immune system related to growing age, is described as immunosenescence, a process including anatomical changes (skin/vaginal atrophy, prostate hypertrophy, incomplete bladder emptying, diminished mucous production etc.) and cellular modifications (decreased chemotaxis/phagocytosis by macrophages/monocytes and neutrophils, superoxide production, alterations in both the expression of cell surface receptors and the signal transduction (32-34). The stimulation of macrophages toll-like receptor results in decreased production of tissue necrosis factor-a (TNFa) and interleukin-6 (IL6), while diminished expression of major histocompatibility complex (MHC) II allows to a reduced capacity for antigen presentation, mainly concerning dendritic cells (33,34). Finally the absolute number of natural killer (NK) cells increase in order to counterbalance a shift to a modified phenotype, with reduced cytotoxic activity and cytokines production which diminish the activation of both DCs and adaptive system response (34,35). Beyond these, various stressors changes in gut microbiota (predominantly endogenous, such as cell debris-products of damaged cells/organelles) and cells senescence-associated secretory phenotype (SASP) (36-38) have been correlated with chronic low-grade inflammation, known as inflammaging (39). Recently, it was proposed that monocytes, after an epigenetic change, promoting metabolic shift towards anaerobic glycolysis, acquire a pro-inflammatory phenotype which also conveys a kind of memory to the innate immune system (trained immune system). This is speculated to be at the center of inflammaging (40).

Full table
Modifications in the adaptive immune system seem to be much more investigated. B cells number and diversity are depleted; while their functionality is also altered, due to mRNA instability, resulting in decreased class switching and antibodies production (41,42). T cells are generally divided into two great subpopulations; CD4+ T cells-regulator and memory cells- and CD8 T cells-memory and effector cells. Aging seems to result in a reduced production of their progenitor cells, naive T-cells, via a shift in the hematopoietic stem cell to myeloid lineage (43) in addition to the thymus involution (44). Furthermore, the increased turnover rate that has been reported for peripheral naive CD8+ T cells in elderly (CD4+ compartment is maintained) results in the accumulation of reverted and virtual memory cells, which are not truly naive, thus further compromising their population (45,46). In terms of terminally differentiated T-cells, especially CD8+, their proliferation is highly oligoclonal (47), something which is more evident with latent viral infections. Indeed CD8+ T-cells specific for Varicella Zoster virus (VZV) tend to decrease (increasing reactivation) while the opposite applies for Epstein-Barr virus (EBV) and Cytomegalovirus (CMV) (48). The latter was initially proposed to have deleterious effects but subsequent studies are controversial (49). Instead of that, T-cell receptor repertoire although decreasing with aging, still remains wide (50). The above-mentioned clones undergo various epigenetic changes and acquire either a senescent (low proliferation/maintained cytokines production) or an exhausted phenotype (low proliferation/cytokine production) (51). The sum of these changes in addition to the diminished survival and functionality of T-cells, caused by inflammaging (51), increase vulnerability to infections and limit vaccination effectiveness.
Risk factors, epidemiology and infections in elderly ICU population
Approximately 60% of patients with sepsis are >65 years old (30). The comorbidities in the elderly population have a major role for the development of ICU infections (52). A large study of 30,239 community-dwelling participants showed that incident sepsis in the ICU was associated with a baseline history of chronic lung disease (adjusted HR: 2.43; 95% CI: 2.05–2.86), peripheral artery disease (adjusted HR: 2.16; 95% CI: 1.58–2.95), chronic kidney disease (adjusted HR 1.99; 95% CI: 1.73–2.29), myocardial infarction (adjusted HR 1.79; 95% CI: 1.49–2.15), diabetes (adjusted HR 1.78; 95% CI: 1.53–2.07), stroke (adjusted HR 1.67; 95% CI: 1.34–2.07), deep vein thrombosis (adjusted HR 1.63; 95% CI: 1.29–2.06), coronary artery disease (adjusted HR: 1.61; 95% CI: 1.38–1.87), hypertension (adjusted HR 1.49; 95% CI: 1.29–1.74), atrial fibrillation (adjusted HR 1.48; 95% CI: 1.21–1.81) and dyslipidemia (adjusted HR: 1.16; 95% CI: 1.01–1.34). Risk of sepsis increased with the total number of chronic medical conditions (53). Immunosuppressive diseases (such as AIDS and cancer), immunosuppressive medications, diabetes, alcohol abuse, indwelling catheters or conditions involving altered skin integrity, predispose to ICU infections (54). A recent study from Australia, that examined 4,137 patients above 80 years admitted to the ICU for sepsis over a 15-year long period [2000–2015], demonstrated that the most prevalent comorbidity in these patients was chronic cardiovascular disease (9.5%), followed by chronic respiratory disease (7.3%) and diabetes mellitus type I (4.8%) (55).
When examining potential underlying mechanisms, longstanding diabetes mellitus may increase infection risk due to peripheral neuropathy, poor vasculature and delayed phagocytosis leading to decreased clearance of yeast and bacteria by neutrophils (56). Similar immune deficiencies contribute to infections in case of chronic kidney disease (57), whereas chronic liver failure causes impairment of complement factor formation and proliferation of cellular immunity. COPD patients may present impaired mucociliary clearance, alveolar macrophage dysfunction, and suppressed cough mechanism, thus increasing the risk for lower respiratory tract infections. Frailty is considered an indirect risk factor for elderly hospitalization and therefore in-hospital infections and ICU admission (58). Finally, it has also been recently shown that patients with septicaemia, especially of higher severity, seem to have a higher risk for developing dementia after hospitalization (59).
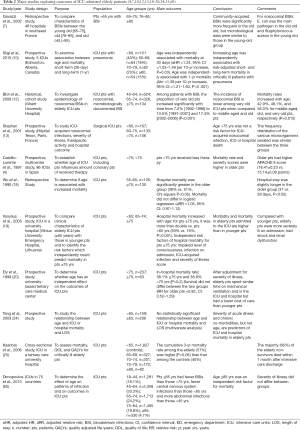
There are very few large-scale studies focused on ICU Acquired Infections in this population and the data are scarce and controversial. Most of them derive from single centers and target one type (4,7-17) of infection (18-32). The most frequent IAIs are respiratory infections, followed by UTIs and BSIs including (7-15) CRIs (4,16-29). The most important studies reporting on epidemiology and outcomes of elderly ICU patients are summarized in Table 2.
An analysis of data from an international, observational, point-prevalence study: Extended Prevalence of Infection in Intensive Care (EPIC II), stands out as one of the largest epidemiological studies in the field. The authors aimed to determine the effect of age on patterns of infection and on outcomes. The study encompassed 1,265 ICUs in 75 countries (66% mixed medical-surgical ICUs) and 13,796 adults of whom 7,087 (51.4%) had an infection. Among them, 1,713 (24.2%) were 65 to 74, 1,405 (19.8%) were 75 to 84, and 330 (4.7%) were aged 85 and older. Those aged 85 and older had proportionately more abdominal infections, fewer BSIs, more Gram-negative pathogens and higher mortality rates compared to younger counterparts (60). Quite striking in respect to the worldwide distribution of the prevalence study, surgical emergency predominated as admission cause across all age groups, however displaying a linear increase with age, ranging from 41% in young adults aged 18–44 years to 51% in adults aged ≥85 years (60). Maillet et al. studied a subgroup of overaged ICU population (>80 yrs old) and found that of the 343 IAI, 19% occurred in the elderly group (43% was respiratory infections, 24% UTIs, 21% BSIs, 4.5% CRIs and 0.5% others). Elderly and younger patients with ICU acquired infections were predominantly infected by Gram-negative organisms (56.6%) and had significantly prolonged ICU stay but ICU mortality rates were significantly higher for elderly compared with younger patients (38.6% vs. 25.0% respectively, P<0.001) (61). Sousa et al. in a study of 308 elderly, described respiratory tract infections in 49.7%, UTIs in 33.8%, BSIs in 21.1% and surgical site infections in 4.9%. In three age sub-groups: 60 to 69 years, 70 to 79 years and over 80 years, the mortality rate was 46.8%, 49.5% and 64.9% respectively and according to the Kaplan-Meier survival curve, hospital mortality in the elderly who developed ICU infection did not have large variations among age groups (62). In line with this, Hifumi et al. over a 7-year study period, retrospectively investigated patients with CAP who required mechanical ventilation (3 age groups: 65–74, 75–84, and ≥85 years) and concluded that age itself was not significantly associated with weaning from mechanical ventilation (P=0.59), in-hospital mortality (P=0.90), ventilator-free days (P=0.83) or ICU days (P=0.12) among the 3 age groups (63).
Critically ill elderly patients are particularly vulnerable to ICU-BSI, which is associated with prolonged LOS, resulting in excess costs and high mortality. In most cases ICU-BSIs are secondary to UTI, lung, soft tissue or intra-abdominal infections. Even if no causative was identified in over the half cases of sepsis, Gram-negative bacteria were 1.31 times more likely to be the cause of sepsis among patients 65 years or older, with respiratory and genitourinary sources being the most common causes (15). Staphylococcus aureus is the most common isolated in all age groups, but in the elderly group is more often drug-resistant (64). Apart from the methicillin-resistant Staphylococcus aureus (MRSA), there is data supporting that other Gram-negative and Gram-positive multi-drug resistant (MDR) are increasingly isolated in the elderly, including vancomycin-resistant enterococci (VRE) and extended spectrum b-lactamase (ESBL) Klebsiella spp strains (65,66). Moreover, elderly population is at higher risk of infections due to Pseudomonas aeruginosa because of frequent antimicrobial and/or steroid use (recurrent infections, COPD exacerbations, autoimmune diseases), structural lung diseases (bronchiectasis, in respiratory infections) and the great number of residents in nursing homes and assisted living facilities (67).
Enterobacteriaceae are the most frequent family of microorganisms isolated in UTIs according to recent epidemiological data from EARSS-NET, representing 45.3% of all isolated microorganisms with Candida spp ranking second. Although E. coli is the predominant in all ages, other Gram-negative bacteria, such as Proteus spp, Klebsiella spp, and Pseudomonas spp. are more frequent isolated in older population (68,69).
Elderly with fungal infections in the ICU
The incidence of candideamia in elderly critically ill patients has no differences compared to younger patients (60). However, in the elderly population there are differences regarding the outcome. In a post hoc analysis of a non-comparative, prospective, multicentre, phase IIIb study the global success rate and the incidence/profile of adverse events with IV anidulafungin treatment against confirmed candideamia were similar in elderly and non-elderly patients at EOT in mITT but the 28-days mortality was higher in elderlies because this population during the entry in the study had more comorbidities and higher APACHE II and SOFA scores (70). In patients with intraabdominal candidiasis, differences have been noted between elderlies and non-elderlies regarding the incidence and outcomes. In a post-hoc analysis of a retrospective multinational cohort study, 482 patients were included, 124 (25.7%) of them being elderly and 358 (74.3%) non-elderly. Mortality was significantly higher in the elderly group, while factors independently predicting mortality in elderly patients were end-stage renal disease and inadequate abdominal source control (71). Data regarding the epidemiology and diagnosis of invasive aspergillosis (IA) in the critically ill population are limited, with data regarding elderly patients (the critically ill populatioA prospective, international, multicenter observational study (AspICU study) including adult ICU patients, with a culture and/or direct examination and/or histopathological sample positive for Aspergillus spp. at any site reported that elderly and non-elderly ICU patients with IA differed in a number of characteristics, including comorbidities, clinical features of the disease, mycology testing, and radiological findings. No difference regarding mortality was found. Elderly patients had less diagnostic radiological findings and when these findings were present they were detected late in the disease course. All patients who were diagnosed with proven IA died (72).
Therapeutic considerations in elderly ICU patients
There are certain particularities concerning the treatment of critically ill elderly (Figure 2). Septic patients older than 65 years old are twice as likely to suffer from at least one chronic comorbidities and three times more likely to have a coincident diagnosis of coronary artery disease or congestive heart failure (15). Renal and hepatic impairment may also be present in the elderly and therefore potentially nephrotoxicity or hepatotoxicity of the medication used should be carefully considered.
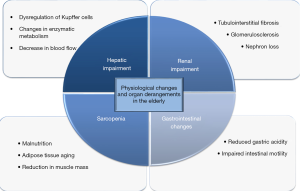
Renal aging is a complex process which plays an important role on pharmacokinetics. Nephron loss, glomerulosclerosis and tubulointerstitial fibrosis predispose to acute kidney injury in the elderly population especially when individuals are treated with nephrotoxic agents (73). Deterioration of renal function leads to accumulation of active metabolites of certain drugs which consequently results in vulnerability to a certain drug effect (74) and increased adverse drug interactions, especially as far as hydrophilic molecules is concerned. It is well documented that sarcopenia is an independent risk factor for mortality in elderly ICU patients (75). In such patients, measured serum creatinine may be falsely low due to the reduction in muscle mass (76) and this fact could probably lead to an overestimation of renal function. Even if age-adjusted equations are used for GFR estimation, the latter should be carefully interpreted (77), as they may represent frailty (78), malnutrition, cachexia or even inflammation (79). Furthermore, the high adipose tissue content in the elderly individuals and the consequent sarcopenia plays an important role in lipophilic and hydrophilic drugs metabolism and more specifically to certain classes of antibiotics (80).
Pseudocapillarisation of the liver sinusoidal endothelial cells, dysregulation of Kupffer cell activation and age-related changes in hepatic metabolism may explain the pharmacokinetics and pharmacodynamics of hepatic metabolism of the drugs concerning the elderly (81). Hepatic parenchymal mass and blood flow notably decrease in the elderly while changes in liver enzymes are present as well (82). All these changes do affect the administration of certain medications as it is suggested that the dosage of high clearance drugs should be reduced by approximately 40% in the elderly while the dosage of low clearance drugs should be reduced by approximately 30% (83). Finally, the reduced gastric acidity and the impaired intestinal motility may retard the dissolution and absorption rate of the enterically administered drugs or supplements (84). Older adults admitted to ICU are pretreated with an average of twelve different medications (85). As if this were not enough, prescription and nonprescription medications, namely over-the-counter medications, herbal and dietary supplements are now widely used by the elderly (86) and this could lead to unpredictable drug-to drug interactions (87). In a prospective study Doan et al. demonstrated that the probability of a drug-to-drug interaction increased with the number of medications and reached 100% when a patient was treated with 20 or more medications (88). Given the fact that more than 70% of ICU patients receive multiple antibiotics, usually empirically (89), those interactions seem more probable especially when referring to the elderly (90). Despite all the reservations mentioned above, clinicians should not withhold lifesaving treatment options for the sake of advanced age alone (91).
Conclusions
The elderly population is increasing in the developed world, therefore elderlies account for a considerable proportion of ICU admissions. As a definition “elderly” is a matter of debate, subgroups of 65–74 (young-old elderlies), 75–84 (old elderlies) and >85 (old-old or oldest old elderlies) have been used to explore ICU outcomes in this population in line with the continuous character of ageing process. Vulnerability of elderly people in infections and severe infections requiring advanced support has been thoroughly elucidated by physiological and functional alterations of the human body and organs, immunosenescence and inflammaging. Comorbidities associated with increasing age, such as diabetes mellitus, renal insufficiency and immunosuppressive conditions pose an additive risk for infections. Epidemiological differences of ICU infections in elderlies, compared to other adults include obscure presentation with predilection for intraabdominal foci, lower rates of microbiological confirmation and implication of MDR pathogens. MDR Enterobacteriaceae are very common, owing to multiple recognized risk factors for MDR infections in this age group, such as multiple antibiotic prescriptions, specific comorbidities (such as bronchiectasis or COPD), residence in long-term-care facilities and frequent hospitalisations, among others. Data from a large European database showed that Candida spp infections rank second, after Enterobacteriaceae among elderlies with ICU infections. Organ derangements and multiple concomitant medications may pose important implications in treatment decisions, while calling for vigilance for adverse events and toxicity. Despite all the above, and the heterogeneity of most studies, elderlies did not have disappointing ICU outcomes. In particular, most studies have shown a worse ICU outcome for the group of oldest-old elderlies, compared with young adults and elderlies in the range of 65 to 84 years of age. These data indicate that age per se may not represent a barrier in decisions concerning ICU admission. However, due to the epidemiological particularities of this age group rigorous identification of the site of infection and pathogen should be sought, enabling selection of appropriate antimicrobial treatment and overall clinical management, which ultimately will optimize patients’ outcomes.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Gastrell J. Annual update: Mortality statistics 2001: General. Health Stat Q 2004.67-9. [PubMed]
- de Rooij SE., Abu-Hanna A, Levi M, et al. Factors that predict outcome of intensive care treatment in very elderly patients: A review. Crit Care 2005;9:R307-14. [Crossref] [PubMed]
- Garrouste-Orgeas M, Timsit JF, Montuclard L, et al. Decision-making process, outcome, and 1-year quality of life of patients over 80 years referred for intensive care unit admission. Intensive Care Med 2006;32:e1045-51. [Crossref]
- Castillo-Lorente E, Rivera-Fernandez R, Vazquez-Mata G. Limitation of therapeutic activity in elderly critically ill patients. Project for the Epidemiological Analysis of Critical Care Patients. Crit Care Med 1997;25:1643-8. [Crossref] [PubMed]
- Groeger JS, Guntupalli KK, Strosberg M, et al. Descriptive analysis of critical care units in the United States: Patient characteristics and intensive care unit utilization. Crit Care Med 1993;21:279-91. [Crossref] [PubMed]
- Marik PE. Management of the critically ill geriatric patient. Crit Care Med 2006;34:S176-82. [Crossref] [PubMed]
- Gavazzi G, Krause KH. Ageing and infection. Lancet Infect Dis 2002;2:659-66. [Crossref] [PubMed]
- Smith PW. Nosocomial infections in the elderly. Infect Dis Clin North Am 1989;3:763-77. [PubMed]
- Saviteer SM, Samsa GP, Rutala WA. Nosocomial infections in the elderly. Increased risk per hospital day. Am J Med 1988;84:661-6. [Crossref] [PubMed]
- Sligl WI, Eurich DT, Marrie TJ, et al. Age still matters: Prognosticating short- and long-term mortality for critically ill patients with pneumonia. Crit Care Med 2010;38:2126-32. [Crossref] [PubMed]
- Rello J, Rodriguez R, Jubert P, et al. Severe community-acquired pneumonia in the elderly: Epidemiology and prognosis. Study Group for Severe Community-Acquired Pneumonia. Clin Infect Dis 1996;23:723-8. [Crossref] [PubMed]
- Blot S, Cankurtaran M, Petrovic M, et al. Epidemiology and outcome of nosocomial bloodstream infection in elderly critically ill patients: A comparison between middle-aged, old, and very old patients. Crit Care Med 2009;37:1634-41. [Crossref] [PubMed]
- Stéphan F, Cheffi A, Bonnet F. Nosocomial infections and outcome of critically ill elderly patients after surgery. Anesthesiology 2001;94:407-14. [Crossref] [PubMed]
- Avci M, Ozgenc O, Coskuner SA, et al. Hospital acquired infections (HAI) in the elderly: Comparison with the younger patients. Arch Gerontol Geriatr 2012;54:247-50. [Crossref] [PubMed]
- Martin GS, Mannino DM, Moss M. The effect of age on the development and outcome of adult sepsis. Crit Care Med 2006;34:15-21. [Crossref] [PubMed]
- Rowe T, Araujo KL, Van Ness PH, et al. Outcomes of older adults with sepsis at admission to an intensive care unit. Open Forum Infect Dis 2016;3:ofw010. [Crossref] [PubMed]
- McLean RF, McIntosh JD, Kung GY, et al. Outcome of respiratory intensive care for the elderly. Crit Care Med 1985;13:625-9. [Crossref] [PubMed]
- Wu AW, Rubin HR, Rosen MJ. Are elderly people less responsive to intensive care? J Am Geriatr Soc 1990;38:621-7. [Crossref] [PubMed]
- Vosylius S, Sipylaite J, Ivaskevicius J. Determinants of outcome in elderly patients admitted to the intensive care unit. Age Ageing 2005;34:157-62. [Crossref] [PubMed]
- Ely EW, Evans GW, Haponik EF. Mechanical ventilation in a cohort of elderly patients admitted to an intensive care unit. Ann Intern Med 1999;131:96-104. [Crossref] [PubMed]
- Bo M, Massaia M, Raspo S, et al. Predictive factors of in-hospital mortality in older patients admitted to a medical intensive care unit. J Am Geriatr Soc 2003;51:529-33. [Crossref] [PubMed]
- Dardaine V, Constans T, Lasfargues G, et al. Outcome of elderly patients requiring ventilatory support in intensive care. Aging (Milano) 1995;7:221-7. [PubMed]
- Dardaine V, Constans T. Outcome of elderly patients requiring ventilator support. Age Ageing 1997;26:238. [Crossref] [PubMed]
- Tang EY, Hsu LF, Lam KN, et al. Critically ill elderly who require mechanical ventilation: The effects of age on survival outcomes and resource utilisation in the medical intensive care unit of a general hospital. Ann Acad Med Singapore 2003;32:691-6. [PubMed]
- Kaarlola A, Tallgren M, Pettila V. Long-term survival, quality of life, and quality-adjusted life-years among critically ill elderly patients. Crit Care Med 2006;34:2120-6. [Crossref] [PubMed]
- Lee CC, Chen SY, Chang IJ, et al. Comparison of clinical manifestations and outcome of community-acquired bloodstream infections among the oldest old, elderly, and adult patients. Medicine (Baltimore) 2007;86:138-44. [PubMed]
- Reunes S, Rombaut V, Vogelaers D, et al. Risk factors and mortality for nosocomial bloodstream infections in elderly patients. Eur J Intern Med 2011;22:e39-44. [Crossref] [PubMed]
- Gavazzi G, Mallaret MR, Couturier P, et al. Bloodstream infection: Differences between young-old, old, and old-old patients. J Am Geriatr Soc 2002;50:1667-73. [Crossref] [PubMed]
- Rubin DB. Estimating causal effects from large data sets using propensity scores. Ann Intern Med 1997;127:757-63. [Crossref] [PubMed]
- Angus DC, Linde-Zwirble WT, Lidicker J, et al. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 2001;29:1303-10. [Crossref] [PubMed]
- Flevari A, Theodorakopoulou M, Velegraki A, et al. Clin Interv Aging 2013;8:1199-208. [PubMed]
- Bender BS. Infectious disease risk in the elderly. Immunol Allergy Clin North Am 2003;23:57-64. [Crossref] [PubMed]
- Linehan E, Fitzgerald DC. Ageing and the immune system: focus on macrophages. Eur J Microbiol Immunol (Bp) 2015;5:14-24. [Crossref] [PubMed]
- Solana R, Tarazona R, Gayoso I, et al. Innate immunosenescence: effect of aging on cells and receptors of the innate immune system in humans. Semin Immunol 2012;24:331-41. [Crossref] [PubMed]
- Hazeldine J, Lord JM. The impact of ageing on natural killer cell function and potential consequences for health in older adults. Ageing Res Rev 2013;12:1069-78. [Crossref] [PubMed]
- Franceschi C, Garagnani P, Vitale G, et al. Inflammaging and 'Garb-aging'. Trends Endocrinol Metab 2017;28:199-212. [Crossref] [PubMed]
- Biagi E, Candela M, Fairweather-Tait S, et al. Aging of the human metaorganism: the microbial counterpart. Age (Dordr) 2012;34:247-67. [Crossref] [PubMed]
- Byun HO, Lee YK, Kim JM, et al. From cell senescence to age-related diseases: differential mechanisms of action of senescence-associated secretory phenotypes. BMB Rep 2015;48:549-58. [Crossref] [PubMed]
- Franceschi C, Bonafè M, Valensin S, et al. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann N Y Acad Sci 2000.244-54. [PubMed]
- Fulop T, Larbi A, Dupuis G, et al. Immunosenescence and Inflamm-Aging As Two Sides of the Same Coin: Friends or Foes? Front Immunol 2018;8:1960. [Crossref] [PubMed]
- Frasca D, Diaz A, Romero M, et al. Age effects on B cells and humoral immunity in humans. Ageing Res Rev 2011;10:330-5. [Crossref] [PubMed]
- Gibson KL, Wu YC, Barnett Y, et al. B-cell diversity decreases in old age and is correlated with poor health status. Aging Cell 2009;8:18-25. [Crossref] [PubMed]
- Geiger H, de Haan G, Florian MC. The ageing haematopoietic stem cell compartment. Nat Rev Immunol 2013;13:376-89. [Crossref] [PubMed]
- Gui J, Mustachio LM, Su DM, et al. Thymus Size and Age-related Thymic Involution: Early Programming, Sexual Dimorphism, Progenitors and Stroma. Aging Dis 2012;3:280-90. [PubMed]
- Cicin-Sain L, Messaoudi I, Park B, et al. Dramatic increase in naive T cell turnover is linked to loss of naive T cells from old primates. Proc Natl Acad Sci USA 2007;104:19960-5. [Crossref] [PubMed]
- Nikolich-Žugich J. Aging of the T cell compartment in mice and humans: from no naive expectations to foggy memories J Immunol 2014;193:2622-9. [Crossref] [PubMed]
- Henson SM, Riddell NE, Akbar AN. Properties of end-stage human T cells defined by CD45RA re-expression. Curr Opin Immunol 2012;24:476-81. [Crossref] [PubMed]
- Khan N, Shariff N, Cobbold M, et al. Cytomegalovirus seropositivity drives the CD8 T cell repertoire toward greater clonality in healthy elderly individuals. J Immunol 2002;169:1984-92. [Crossref] [PubMed]
- Sansoni P, Vescovini R, Fagnoni FF, et al. New advances in CMV and immunosenescence. Exp Gerontol 2014;55:54-62. [Crossref] [PubMed]
- Qi Q, Liu Y, Cheng Y, et al. Diversity and clonal selection in the human T-cell repertoire. Proc Natl Acad Sci U S A 2014;111:13139-44. [Crossref] [PubMed]
- Goronzy JJ, Weyand CM. Successful and Maladaptive T Cell Aging. Immunity 2017;46:364-78. [Crossref] [PubMed]
- Nasa P, Juneja D, Singh O. Severe sepsis and septic shock in the elderly: An overview. World J Crit Care Med 2012;1:23-30. [Crossref] [PubMed]
- Wang HE, Shapiro NI, Griffin R, et al. Chronic Medical Conditions and Risk of Sepsis. PLoS One 2012;7:e48307. [Crossref] [PubMed]
- Cecconi M, Evans L, Levy M, et al. Sepsis and septic shock. Lancet 2018;392:75-87. [Crossref] [PubMed]
- Athari F, Hillman KM, Frost SA. The changing nature of the population of intensive-care patients. J Hosp Adm 2018;7:1. [Crossref]
- Zykova SN, Jenssen TG, Berdal M, et al. Altered cytokine and nitric oxide secretion in vitro by macrophages from diabetic type II-like db/db mice. Diabetes 2000;49:1451-8. [Crossref] [PubMed]
- Chonchol M. Neutrophil dysfunction and infection risk in end-stage renal disease. Semin Dial 2006;19:291-6. [Crossref] [PubMed]
- Clifford KM, Dy-Boarman E, Haase K, et al. Challenges with Diagnosing and Managing Sepsis in Older Adults. Expert Rev Anti Infect Ther 2016;14:231-41. [Crossref] [PubMed]
- Chou CH, Lee JT, Lin CC, et al. Septicemia is associated with increased risk for dementia: a population-based longitudinal study. Oncotarget 2017;8:84300-8. [Crossref] [PubMed]
- Dimopoulos G, Koulenti D, Blot S, et al. Extended Prevalence of Infection in Intensive Care Study Investigators. Critically ill elderly adults with infection: analysis of the extended prevalence of infection in intensive care study. J Am Geriatr Soc 2013;61:2065-71. [Crossref] [PubMed]
- Maillet JM, Guérot E, Novara A, et al. Comparison of intensive-care-unit-acquired infections and their outcomes among patients over and under 80 years of age. J Hosp Infect 2014;87:152-8. [Crossref] [PubMed]
- Sousa ÁFL, Queiroz AAFLN, Oliveira LB. Deaths among the elderly with ICU infections. Rev Bras Enferm 2017;70:733-9. [Crossref] [PubMed]
- Hifumi T, Jinbo I, Okada I, et al. The impact of age on outcomes of elderly ED patients ventilated due to community acquired pneumonia. Am J Emerg Med 2015;33:277-81. [Crossref] [PubMed]
- Girard TD, Ely EW. Bacteremia and sepsis in older adults. Clin Geriatr Med 2007;23:633-47. [Crossref] [PubMed]
- Levy MM, Dellinger RP, Townsend SR, et al. The Surviving Sepsis Campaign: results of an international guideline-based performance improvement program targeting severe sepsis. Crit Care Med 2010;38:367-74. [Crossref] [PubMed]
- Lineberry C, Stein DE. Infection, Sepsis, and Immune Function in the Older Adult Receiving Critical Care. Crit Care Nurs Clin North Am 2014;26:47-60. [Crossref] [PubMed]
- Arnold FW. How Antibiotics should be prescribed to hospitalized elderly patients with Community-Acquired Pneumonia. Drugs Aging 2017;34:13-20. [Crossref] [PubMed]
- Das R, Towle V, Van Ness PH, et al. Adverse outcomes in nursing home residents with increased episodes of observed bacteriuria. Infect Control Hosp Epidemiol 2011;32:84-6. [Crossref] [PubMed]
- ECDC SURVEILLANCE REPORT. Incidence and attributable mortality of healthcare-associated infections in intensive care units in Europe, 2008-2012.
- Dimopoulos G, Paiva JA, Meersseman W, et al. Efficacy and safety of anidulafungin in elderly, critically ill patients with invasive Candida infections: a post hoc analysis. Int J Antimicrob Agents 2012;40:521-6. [Crossref] [PubMed]
- Dimopoulos G, Matthaiou DK, Righi E, et al. Elderly versus non-elderly patients with intra-abdominal candidiasis in the ICU. Minerva Anestesiol 2017;83:1126-36. [PubMed]
- Matthaiou DK, Dimopoulos G, Taccone FS, et al. Elderly versus nonelderly patients with invasive aspergillosis in the ICU: a comparison and risk factor analysis for mortality from the AspICU cohort. Med Mycol 2018;56:668-78. [Crossref] [PubMed]
- Wang X, Bonventre JV, Parrish AR. The aging kidney: increased susceptibility to nephrotoxicity. Int J Mol Sci 2014;15:15358-76. [Crossref] [PubMed]
- Doogue MP, Polasek TM. Drug dosing in renal disease. Clin Biochem Rev 2011;32:69-73. [PubMed]
- Moisey LL, Mourtzakis M, Cotton BA, et al. Nutrition and Rehabilitation Investigators Consortium (NUTRIC). Skeletal muscle predicts ventilator-free days, ICU-free days, and mortality in elderly ICU patients. Crit Care 2013;17:R206. [Crossref] [PubMed]
- Corsonello A, Pedone C, Corica F, et al. Concealed renal insufficiency and adverse drug reactions in elderly hospitalized patients. Archives of Internal Medicine 2005;165:790-5. [Crossref] [PubMed]
- Garasto S, Fusco S, Corica F, et al. Estimating glomerular filtration rate in older people. Biomed Res Int 2014;2014:916542. [Crossref] [PubMed]
- Cox HJ, Bhandari S, Rigby AS, et al. Mortality at low and high estimated glomerular filtration rate values: a 'U' shaped curve. Nephron Clin Pract 2008;110:c67-72. [Crossref] [PubMed]
- Suzuki H, Asakawa A, Amitani H, et al. Ghrelin and cachexia in chronic kidney disease. Pediatr Nephrol 2013;28:521-6. [Crossref] [PubMed]
- Novak LP. Aging, total body potassium, fat-free mass, and cell mass in males and females between ages 18 and 85 years J Gerontol 1972;27:438-43. [Crossref] [PubMed]
- Mitchell SJ, Kane AE, Hilmer SN. Age-Related Changes in the Hepatic Pharmacology and Toxicology of Paracetamol Curr Gerontol Geriatr Res 2011;2011:624156. [PubMed]
- Kim IH, Kisseleva T, Brenner DA. Aging and liver disease. Curr Opin Gastroenterol 2015;31:184-91. [Crossref] [PubMed]
- Le Couteur DG, McLean AJ. The Aging Liver Clin Pharmacokinet 1998;34:359-73. [Crossref] [PubMed]
- Soenen S, Rayner CK, Jones KL, et al. The ageing gastrointestinal tract. Curr Opin Clin Nutr Metab Care 2016;19:12-8. [Crossref] [PubMed]
- Bell CM, Brener SS, Gunraj N, et al. Association of ICU or hospital admission with unintentional discontinuation of medications for chronic diseases. JAMA 2011;306:840-7. [Crossref] [PubMed]
- Qato DM, Alexander GC, Conti RM, et al. Use of prescription and over-the-counter medications and dietary supplements among older adults in the United States. JAMA 2008;300:2867-78. [Crossref] [PubMed]
- Qato DM, Wilder J, Schumm LP, et al. Changes in Prescription and Over-the-Counter Medication and Dietary Supplement Use Among Older Adults in the United States, 2005 vs. 2011. JAMA Intern Med 2016;176:473-82. [Crossref] [PubMed]
- Doan J, Zakrewski-Jakubiak H, Roy J, et al. Prevalence and risk of potential cytochrome p450- mediated drug-drug interactions in older hospitalized patients with polypharmacy. Ann Pharmacother 2013;47:324-32. [Crossref] [PubMed]
- Vincent JL, Rello J, Marshall J, et al. International Study of the Prevalence and Outcomes of Infection in Intensive Care Units. JAMA 2009;302:2323-9. [Crossref] [PubMed]
- Faulkner CM, Cox HL, Williamson JC. Unique aspects of antimicrobial use in older adults. Clin Infect Dis 2005;40:997-1004. [Crossref] [PubMed]
- Yu W, Ash AS, Levinsky NG, et al. Intensive care unit use and mortality in the elderly. J Gen Intern Med 2000;15:97-102. [Crossref] [PubMed]
Cite this article as: Poulakou G, Lagou S, Papadatos S, Anagnostopoulos I, Papatheodoridi M, Dimopoulos G. Infections in elderly intensive care unit patients. J Emerg Crit Care Med 2019;3:44.