
Antifungal drug dosing adjustment in critical patients with invasive fungal infections
Invasive fungal infections in the critically ill pediatric and adult patientOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
Invasive fungal diseases (IFD) are increasing both in incidence and in spectrum globally due to the intense application of aggressive immunosuppressive treatments and the development and administration of new immune modifying drugs (1). In addition, IFDs can complicate the clinical course of immunocompetent groups of patients, such as critically ill patients hospitalized in intensive care units (ICUs) (2). Associated mortality is high, and depends on the underlying medical conditions, the type of pathogen involved, the length of time prior to diagnosis, and the appropriateness of management (3,4).
Candidemia has been found to represent a significant proportion of catheter- and non-catheter- related bloodstream infections and to be the most common IFD in the ICU (5,6). The patients in the ICU with candidemia may significantly differ in terms of heterogeneity and predisposing factors as well as in terms of pathogen and organ involvement. Emergency surgery, underlying malignancy, presence of vascular access devices, prior use of broad-spectrum antibiotics and previous bacterial sepsis appear to significantly predispose to the emergence of invasive candidiasis in the ICU (7). Candidemia and intra-abdominal Candida infections represent the largest proportion of IFD Candida infections (2). Species distribution varies compared to other risk groups, with predominance of non-albicans Candida species in many places (8). Of note, some of these non-albicans Candida species are fluconazole-resistant.
IFDs due to Aspergillus spp. are also encountered in the ICU, with reports of increasing incidence of pneumonia due to Aspergillus, with predilection for immunocompromised patients and those with chronic pulmonary disease or even influenza (9,10). Predominant species involved include Aspergillus fumigatus, Aspergillus flavus and Aspergillus niger with varying contribution worldwide (5). Other opportunistic fungal infections in the setting of an ICU are rare (i.e., invasive mucormycosis in patients with hematological malignancies and Pneumocystis jirovecii in patients with kidney transplantation or HIV infection), affect specific groups of patients (i.e., Cryptococcus and P. jirovecii in HIV-infected patients), and/or are endemic in specific areas of the world (i.e., Histoplasma infections in USA or India) (5).
When comparing IFDs in terms of incidence, risk factors, microbiology, and clinical outcome among different age classes, critically ill neonates and children cannot be considered little adults (11,12). Younger patients have different predisposing factors; prematurity and long stay of very low-birth weight neonates in the ICU, as well as hematologic malignancy and neutropenia in children account for the majority of cases.
Treatment of IFD in critically ill patients is challenging as these patients are usually characterized by different demographic characteristics (age, weight, organ function) and a variety of co-morbidities and other co-administered medications. Critically ill patients are hallmarked by altered intravascular hemodynamics, clearance, tissue perfusion and hypoalbuminemia, causing unpredictable drug distribution within the different compartments of the organism (13). Pharmacokinetics and drug elimination are further complicated by kidney or liver insufficiencies, co-administration of other interacting medications and interventions such as continuous renal replacement therapy and extracorporeal membrane oxygenation, warranting adjustments in such patients.
Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerationsOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
Amphotericin B formulations
Amphotericin B preparations include amphotericin B deoxycholate (DAMB) and the lipid formulations: liposomal amphotericin B (LAMB) and amphotericin B lipid complex (ABLC). All preparations are administered intravenously, exhibit high protein binding (95–99%) in plasma and display non-linear pharmacokinetics. DAMB is eliminated by the kidney and bile (13). The elimination/metabolism of LAMB following intravenous administration has not yet been elucidated (13). The lipid formulations of amphotericin B are in vivo less potent than DAMB on a weight basis. However, due to their reduced nephrotoxicity they can be administered at significantly higher dosages than DAMB (14).
All amphotericin B preparations have concentration-dependent action, with Cmax/MIC being the pharmacodynamic (PD) index most predictive of efficacy. A Cmax/MIC ratio of 2–4 was associated with therapeutic efficacy of DAMB in animal models of invasive candidiasis and aspergillosis, with maximal efficacy at Cmax/MIC of 10 (15). Limited data in pediatric patients receiving LAMB suggest a mean Cmax/MIC of 68 associated with complete response (16). Amphotericin B preparations display a strong post-antifungal effect (PAFE) (17).
The usual dosage of DAMB for adults, children and neonates is 1 mg/kg/day (range: 0.6–1.5 mg/kg/day) administered in one dose. The usual dosage for the lipid formulations ranges between 3–5 mg/kg/day, also in one dose, except for treatment of invasive mucormycosis where a dose of ≥5 mg/kg/day is recommended. Currently available evidence does not support the routine use of therapeutic drug monitoring (TDM) for this class of antifungal agents (18).
Flucytosine
Flucytosine may be administered both intravenously and orally, as oral bioavailability is between 76–98%. It has low protein binding (3–4%) and is mostly (90%) eliminated by glomerular filtration (13). In animal models of invasive candidiasis treated with flucytosine, the PD index associated with net stasis was a T (Time) > MIC of 40% (19). Flucytosine has a narrow therapeutic index with toxicity associated with peak levels. Given its very short PAFE and efficacy associated with T > MIC, frequent (6 hourly) dosage is suggested for patients with normal renal function (15).
The usual dosage of flucytosine in adults and children ranges between 100–200 mg/kg/day divided into 4 doses. The recommended dosage for neonates is 100 mg/kg/day in 2 divided doses. TDM is strongly recommended, as peak concentrations >100 mg/L are associated with bone marrow suppression and hepatotoxicity, while trough levels <20–40 mg/L are associated with development of resistance (18).
Azoles
This class of antifungal agents comprises fluconazole, itraconazole and the triazoles voriconazole, posaconazole and isavuconazole. All of them are available as intravenous and oral formulations.
Fluconazole displays excellent oral bioavailability (>90%) and limited plasma protein binding (~12%). It is eliminated unchanged by the kidney, achieving very high concentrations in urine. Although it is not metabolized in the liver, it inhibits CYP3A4 and CYP2C9 activity; therefore, a number of interactions of fluconazole with other drugs, which are substrates of CYP3A4 or CYP2C9, may be observed (13). The usual dose of fluconazole is 200–400 mg once daily in adults depending on the indication (invasive or mucosal infection) and 12 mg/kg daily for children. For severe infections a loading dose of 800 mg for adults and 25 mg/kg for children may be considered. Neonates may be dosed at 12 mg/kg every 72 hours (if aged <14 days) or every 48 hours (if aged 14–28 days) (13).
Itraconazole oral formulations include capsules or suspension. Absorption however is highly variable and reduced when gastric acidity is decreased. Capsules are better absorbed with food; absorption of the suspension is better on empty stomach. Following oral or intravenous administration itraconazole exhibits very high plasma protein binding (>99%) (20). It is extensively metabolized in the liver, mostly by CYP3A4 enzyme. Numerous interactions with other drugs, which are CYP3A4 substrates, have been observed and complicate itraconazole treatment (18). In the intravenous formulation, the hydroxypropyl-β-cyclodextrin carrier is eliminated through glomerular filtration; this molecule may therefore be accumulated in patients with impaired renal function (13). The usual recommended dosage of intravenous itraconazole for adults is 200 mg once daily, with loading of 200 mg twice daily for the first 2 days. For children, corresponding dosage is 2.5 mg/kg daily with loading of 2.5 mg/kg twice daily for 2 days. Usual dosage for oral preparations ranges between 200–400 mg/day for adults and 2.5–5 mg/kg/day for children administered in 1 or 2 doses.
Voriconazole oral formulations (tablets, suspension) exhibit high bioavailability (~96%), which is not affected by gastric pH but may be reduced with high fat meals. Following oral or intravenous administration the plasma protein binding of voriconazole is approximately 58%. Voriconazole exhibits non-linear pharmacokinetics and is metabolized in the liver by P450 isoenzymes CYP2C19, CYP2C9 and CYP3A4 (21). Significant inter-patient variability in its clearance and exposure [area under the concentration-time curve (AUC)] may be observed, which is in part explained by genetic polymorphisms of CYP2C19 enzyme. In addition, a number of clinically significant interactions of voriconazole with other drugs (CYP450 substrates, inhibitors or inducers) have been observed (18,21). The intravenous formulation of voriconazole also contains a cyclo-dextrin carrier (hydroxypropylbetadex), which is eliminated in the urine and may be accumulated in patients with creatinine clearance <50 mL/min. The usual recommended dosage of intravenous voriconazole for adults is 6 mg/kg/12 hours for the 1st day and subsequently 4 mg/kg/12 hours. For children corresponding dosage is 9 mg/kg/12 hours and 8 mg/kg/12 hours, respectively. Oral formulations are dosed at 400 mg/12 hours (1st day) followed by 200 mg/12 hours for adults, and 9 mg/kg/12 hours for children (no loading recommended).
Posaconazole oral formulations also include gastro-resistant tablets and suspension; the tablets display improved bioavailability compared to the suspension, resulting in more than 3 times higher exposure (AUC) (22). Absorption is improved with fatty meals. Posaconazole is highly bound to plasma proteins (>98%). It is mainly excreted unchanged in the feces and only partially metabolized to multiple glucuronide conjugates. It inhibits CYP3A4 but not other CYP450 enzymes; numerous interactions with CYP3A4 substrates may therefore be observed. The intravenous preparation of posaconazole contains betadex sulfobutyl ether sodium as a vehicle, which is excreted by the kidney and may be accumulated in case of moderate or severe renal impairment (creatinine clearance <50 mL/min) (22). The usual recommended adult dosage for posaconazole intravenous and tablet formulations is 300 mg/12 hours the 1st day and subsequently 300 mg once daily. For the suspension, the dosage for invasive infections is 200 mg/6 hours.
Isavuconazole oral formulation (capsule) exhibits very high bioavailability (98%), which is not significantly affected by food. Isavuconazole is highly bound (>99%) to plasma proteins, mainly albumin. It is metabolized in the liver by CYP3A4, CYP3A5 and subsequently by uridine diphosphate-glucuronosyltransferases (UGT) (23). It is a moderate CYP3A4 inhibitor and many drug-drug interactions are likely to be observed (23). The usual recommended adult dosage for both intravenous and oral formulation of isavuconazole is 200 mg/8 hours for the 1st 2 days and subsequently 200 mg once daily. A recent open-label, phase I study in patients aged 1–18 years suggest that a loading dose of 10 mg/kg/8 hours for days 1 and 2 followed by 10 mg/kg once daily provides exposure equivalent to adults (24).
Several in vivo studies have suggested that the PD index associated with efficacy (50% maximal effect) of azole agents in Candida infections is a free-drug AUC/MIC ratio of 25–50. This practically means that drug concentrations near the MIC for a 24-hour period would likely achieve this target (15). The correspondent index (free-drug AUC/MIC ratio) for azole efficacy in aspergillosis animal models is significantly lower, ranging from 1.7–11 in various studies (25,26). However, in a murine cryptococcal meningitis model, efficacy (stasis) was associated with a fluconazole AUC/MIC ratio of 389 (27). The azole agents exhibit variable PAFE in vitro, which appears to be related to their non-covalent binding to lipophilic cytoplasmic components of fungal cells (for example significant PAFE for itraconazole but weak for fluconazole) (28). However, in vivo studies have demonstrated prolonged PAFEs for most azoles (29,30).
Clinical data for fluconazole and voriconazole in patients with invasive candidiasis suggest that the animal derived free-drug AUC/MIC ratio of 25–50 is indeed associated with optimal outcome (15,31). Studies on patients with invasive aspergillosis treated with azoles have mostly focused on the relationship of trough concentrations with outcome. For voriconazole, optimal outcome was associated with trough concentrations between 1–2 µg/mL (32). Higher trough levels of voriconazole (2–5 µg/mL) are recommended for severe IFD’s or when there are increased MICs. Similar results were obtained for itraconazole and posaconazole, for which optimal outcome was observed with trough concentrations ≥1 µg/mL (33). Based on available clinical data and considerable inter-patient pharmacokinetic variability, TDM is strongly recommended for patients with invasive fungal infections treated with itraconazole, voriconazole and posaconazole. The need for TDM in the case of isavuconazole has not yet been established (18).
Echinocandins
The echinocandin class of antifungal agents includes caspofungin, micafungin and anidulafungin, all available only as intravenous formulations.
Caspofungin is extensively bound to plasma proteins (~95%) and metabolised in the liver by spontaneous degradation and, further, hydrolysis and N-acetylation. All echinocandins are metabolized in the liver via pathways other than the CYP450 enzyme system (13). Since caspofungin metabolism is independent from cytochrome P450, drug interactions are much less compared to the azoles (34). The usual adult dosage of caspofungin is 70 mg once daily (1st day) and subsequently 50 mg (or 70 mg if body weight >80 kg) once daily. Patients aged 1–17 years should be dosed at 70 mg/m2 once daily (loading) followed by 50 mg/m2 (potentially increased up to 70 mg/m2). Neonates/infants <3 months of age and infants 3–11 months of age should be dosed at 25 mg/m2 and 50 mg/m2 once daily, respectively.
Micafungin is highly bound to plasma proteins (>99%), mainly albumin, and metabolized in the liver to several inactive metabolites. Although in vitro micafungin is a substrate for CYP3A, in vivo hydroxylation by CYP3A plays a minor role. Drug interactions are limited, as for caspofungin (34). The usual recommended therapeutic dosage for adults is 100 mg once daily (potentially increased to 200 mg), for children ≥4 months of age is 2 mg/kg once daily (potentially increased to 4 mg/kg) and for neonates/infants <4 months is 4–10 mg/kg once daily.
Anidulafungin also is extensively bound to plasma proteins (>99%). It is not metabolized in the liver but undergoes slow chemical degradation to a ring-opened peptide that lacks antifungal activity; the latter is excreted mainly in the bile. Anidulafungin is not an inducer or inhibitor of cytochrome P450 enzymes. No significant drug-drug interactions have been observed (35). The usual recommended dosage for adults is 200 mg once daily (1st day) and subsequently 100 mg once daily. The equivalent pediatric dose is 3 mg/kg once daily (1st day) and subsequently 1.5 mg/kg once daily (36).
The echinocandins exhibit concentration-dependent activity, both in vitro and in vivo, and prolonged PAFE, especially caspofungin and anidulafungin (37). For determination of their in vitro activity against Aspergillus isolates the minimum effective concentration (MEC) is used instead of the MIC. The MEC is the lowest drug concentration producing short, stubby and highly branched hyphae (38). The PD parameters associated with therapeutic and fungicidal efficacy are AUC/MIC for Candida spp. (AUC/MEC for Aspergillus) or, even better, Cmax/MIC for Candida (Cmax/MEC for Aspergillus) (15,34). In animal models of invasive C. albicans infection, free-drug Cmax/MIC >1 or AUC/MIC of 10–20 were associated with stasis. Lower ratios were needed for stasis in Candida parapsilosis and Candida glabrata infections (15,39). In vivo models of invasive aspergillosis have suggested that a Cmax/MEC ratio of 10–20 is associated with maximal reduction in pulmonary fungal burden (40). Limited PD data from clinical studies of echinocandins in patients with invasive candidiasis are in agreement with those derived from animal models (15). Their activity against Candida biofilms, which rapidly form on the surface of foreign devices, such as central venous catheters has been established (41).
Echinocandins are excreted to a comparatively very low amount as unchanged drug in the urine; despite anecdotal reports of successful treatment of Candida urinary tract infections they are not considered treatment of choice. Their accumulation in renal parenchyma, however, is high (42). There are currently insufficient data to support the routine use of TDM in patients treated with echinocandins (18).
Renal insufficiencyOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
When comparing patients with candidemia in the setting of ICU to patients with candidemia hospitalized in other hospital departments, ICU patients have more frequently chronic renal insufficiency as co-morbidity, as well as acute renal insufficiency treated by renal replacement therapy (43,44). In fact, among severely ill patients treated in the ICU, sepsis and renal replacement therapy are the only two parameters associated with a higher probability of invasive candidemia (9). ICU-acquired candidemia in chronic renal disease patients and especially developing in the context of acute renal function deterioration is associated with worse outcome and death (45). While data is not always consistent (46,47), dose adjustment may prove important (48).
Treatment of fungal infections in the context of renal impairment is challenging. Most of the antifungals (triazoles and echinocandins) are lipophilic and metabolized in the liver (49). As mentioned previously however, the use of the intravenous formulations of itraconazole, voriconazole and posaconazole is not without limitations due to accumulation of their solvent carriers. This side effect is of particular clinical importance in the case of voriconazole, a highly efficient drug against Aspergillus and other fungi. On the other hand, gastric absorption is severely compromised and unpredictable in critically ill patients worsening even more the situation of patients with renal insufficiency. Newer medications, such as isavuconazole have been designed to, among others, overcome these treatment infelicities. Broad-spectrum antifungals, e.g., conventional amphotericin B, with good penetration and availability in various body compartments, such as the renal parenchyma or the central nervous system have been associated with renal toxicity, driving research towards the development of less toxic formulations (50). Appropriate dosing of antifungals in the context of altered pharmacokinetics and pharmacodynamics in critically ill patients with renal impairment has yet to be fully addressed (51). In general, lipid formulations of amphotericin B do not need dose adjustment, nevertheless the probability of nephrotoxicity must be taken into account; flucytosine and fluconazole should be administered according to creatinine clearance and echinocandins do not require adjustment. During the methods of renal replacement such as continuous venovenous haemodiafiltration different antifungals require or not dose adjustment (47,48,52). Currently available renal function—adjusted dosing data are summarized in Table 1.

Full table
Hepatic insufficiencyOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
Impaired liver function represents a major issue in the everyday practice of ICU both in terms of safety of use of antifungals in critically ill patients, as well as in terms of pharmacokinetics and necessary dose adjustment of the antifungal agents. The presence of liver pathology in ICU patients developing IFD severely impairs outcome and appears to be an independent predictor of 28-day mortality (63). Critically ill patients are already characterized by impaired liver blood flow, biliary excretion and plasma protein binding and cannot tolerate further drug-induced hepatotoxicity (64). On the other hand, cirrhotic patients admitted to the ICU have a higher probability of becoming colonized with fungi and developing invasive fungal infections (65). Fungal infections usually follow bacterial infections and present preferentially in patients with advanced liver disease and organ failure (66).
All antifungal agents can induce liver damage ranging from mild asymptomatic abnormalities in liver function tests to acute liver failure. Drug-induced liver injury is categorized as hepatocellular, cholestatic and mixed injury, according to the ratio of alanine aminotransferase (ALT) to alkaline phosphatase (ALP) (67). The antifungal class associated with the most prominent toxic effect on liver function is the azoles, an effect that is not necessarily dose-dependent (68-70). Liver toxicity is observed frequently with their use; however, hepatic failure is uncommon (71). Main pattern of liver toxicity with this class of antifungals is liver enzymes elevation; itraconazole and posaconazole may induce cholestasis. Pre-existing liver disease is one of the most important predisposing factors. Among the triazoles used in ICU patients (fluconazole, voriconazole and posaconazole) dose reduction of 50% is recommended in the case of voriconazole; therapeutic drug monitoring is considered mandatory (64).
Polyenes are safer in patients with liver impairment; their use may be associated with mild hepatotoxicity regardless of the dose used, an effect that appears to be less frequent with the lipid formulations. They are excreted unchanged in the urine and feces (72). Elevation of liver function tests, however, was not associated with histopathologic changes in a series of 64 patients with hematological malignancies and invasive fungal infections (73).
As epidemiology of candidemia in critically ill patients is changing with the proportion of non-albicans Candida species especially fluconazole-resistant strains rising (74), echinocandins are administered as first line antifungal treatment in critically ill cirrhotic patients (75). Their pharmacodynamics includes good penetration in most tissues, except for the central nervous system/eye and the peritoneal cavity; their efficacy therefore in fungal peritonitis, the second most common fungal infection in critically ill cirrhotic patients, is questionable (76).
Among the three currently available members of echinocandin family, caspofungin is the one that warrants dose reduction in patients with severe liver impairment as the drug molecule becomes transformed in the liver and the recommended dose is 35 mg daily. Micafungin use in juvenile animals has been associated with the development of liver tumors; its use may therefore be restricted to cases where other therapeutic options are not available (74). However, no such toxicity has been observed after intense use in human neonates and children. For patients with severe hepatic insufficiency there are no data for the use of micafungin, and it should be avoided. Further studies need to be conducted to address the issue of altered pharmacokinetics and drug availability in critically ill patients and patient adjusted dosing. Initial data do not suggest however severely affected drug metabolism and levels. However, based on the aforementioned findings in experimental animals, EMA discourages against use of micafungin in patients with severe, advanced and potentially preneoplastic liver conditions (77). There is no need for dose adjustment for anidulafungin even in patients with severe hepatic insufficiency. The currently recommended dose adjustments of antifungal agents used for patients with hepatic insufficiency are summarized in Table 2 (64).
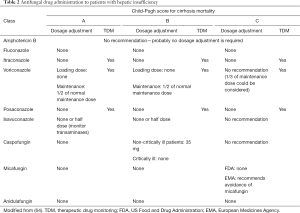
Full table
Age-dependent factorsOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
Invasive fungal infections show a predilection for elderly patients (78). More than half of the cases of candidemia occur in patients >65 years old with a significant associated mortality. Risk factors for invasive fungal infections in this age group, however, differ compared to other age and patient groups (79). Chronic pulmonary insufficiency, chronic kidney disease, diabetes mellitus and a higher Charlson comorbidity index are more frequently associated with the development of invasive fungal diseases in the elderly. Elderly people with invasive fungal diseases may present with more atypical features, and thus evade diagnosis (80). Moreover, physicians are usually more hesitant to apply invasive diagnostic procedures such as bronchoscopy or organ biopsies to patients in this age group. An additional contributing factor to the excess mortality associated with these infections in this age group is the increased resistance profile of the pathogens involved, due to different strain prevalence and previous exposure to antifungals. In general, the antifungal agents that are used in younger ages are also used in the elderly when they are hospitalized in the ICU, keeping in mind the higher probability of decreased renal and hepatic function that older patients in critical condition may have as well as a progressive reduction in total body water and lean body mass with advancing age, leading to a relative increase in body fat, thus affecting drug distribution (81).
However, one of the critical differences between these two extreme age groups when treated for IFIs appear to be the existing comorbidities, which are aggravated by critical illness, and the amount of other co-administered medications. Interactions of triazoles with co-administered medications may prove critical and need to be monitored. Co-medications in critically ill patients, whose levels may reach toxic levels (clinically relevant increase) through administration of triazoles include, among others, immunosuppressants, neuroleptic drugs, antiretroviral medications, acenocoumarol, and hypolipidemic drugs (82,83). With regard to the echinocandins, no particular interactions with other drugs are of clinical significance, with the exception of immunosuppressants, such as cyclosporine or tacrolimus, which may require increase of the administered dose.
Critically ill neonates especially premature neonates are prone to invasive fungal infections mainly candidiasis due to immaturity of immune system and multiple invasive procedures (11). Candidiasis in neonates with extremely low birth weight (ELBW) is frequent, is associated with high morbidity and may be followed by hematogenous Candida meningoencephalitis (HCME) found almost exclusively in this age group (84). Thus, in this age group and especially for ELBW neonates, antifungal fluconazole prophylaxis is justified as part of the antifungal prevention strategy.
The main antifungal agents commonly used in critically ill neonates include amphotericin B, fluconazole, caspofungin and micafungin because of differences in pathophysiology of invasive fungal infections as well as available data on antifungal drug PK/PD in this age group as compared to adults (85-87). In contrast to adults, DAMB is well tolerated by neonates and is recommended for neonatal candidiasis including HCME. Liposomal and lipid complex preparations of amphotericin B are recommended in neonatal invasive candidiasis based on limited PK data, whereas optimal dosing has not been established. Liposomal AmB is recommended at a dose of 3–5 mg/kg/d according to most recent IDSA guidelines (86). For the treatment of HCME in neonates, both DAMB and lipid formulations have shown similar efficacy based on few clinical studies as well as some preclinical models (86,88).
Fluconazole is used both for prophylaxis and treatment of invasive candidiasis in neonates (85,86). Fluconazole is in vitro active against most Candida species that are frequently isolated from neonates (C. albicans and C. parapsilosis) and resistance is low (<5%) at least in Europe and USA (89,90). It can be used as an alternative option for empiric treatment of invasive candidiasis in neonates who have not been exposed to antifungal prophylaxis with fluconazole and as a step-down option for the treatment of HCME (86).
For the treatment of invasive candidiasis in critically ill neonates and in contrast to critically ill adults, echinocandins are not first-line agents because of the lack of PK, safety and efficacy data (85,86,91). Micafungin is the only echinocandin that has been approved for use in neonates in Europe (92). It is used at higher doses (4–10 mg/kg/day) in neonates than older children especially for the treatment of HCME, for which micafungin is used at a dose of 10 mg/kg/day, according to preclinical data and very few clinical studies (93,94). The use of caspofungin in neonates is based on very limited data (95,96). Anidulafungin currently is not approved for use in neonates (36).
Beyond the neonatal period, general practices for the management of invasive fungal diseases in critically ill children are mainly derived from adults. However, significant differences exist in choosing optimal antifungal agents because of the limited clinical data and PK in critically ill children. For invasive candidiasis echinocandins are first-line agents and caspofungin is the most frequently used antifungal agent. In children caspofungin and micafungin have been approved for primary targeted therapy of invasive candidiasis in Europe (85). Pediatric data for the use of anidulafungin have just been published (36). A recent meta-analysis comparing echinocandins (mainly caspofungin and micafungin) to other antifungal agents for the treatment of invasive candidiasis in children showed no significant difference in terms of efficacy and adverse events. However, the use of echinocandins was related with fewer discontinuations due to adverse events (97).
Liposomal amphotericin B is approved as first line therapy of invasive fungal infections in children. As in adults, lipid preparations of amphotericin B have better toxicity profile than DAMB (98).
Fluconazole can be used in invasive candidiasis in children when fluconazole-susceptible species are suspected and patients are in a stable condition. Voriconazole is first line therapy of invasive aspergillosis, scedosporiosis and fusariosis in children except neonates (99-101). A loading dose of 9 mg/kg/dose ×2 followed by a maintenance dose of 8 mg/kg/dose ×2 is currently recommended for intravenous use in children aged 2–11 years old with voriconazole levels desired 1–5.5 µg/mL (102). Higher doses, or even more frequently administered are suggested for children aged less than 2 years of age, but there are limited safety data (103). A full list of all antifungal dosage schemes for neonates and children for treatment or prophylaxis of invasive fungal diseases is out of the scope of this review and can be found in a recent updated review (87).
Various underlying conditionsOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
Sepsis
In a multicenter European cohort study of critically ill patients the PK parameters of patients receiving fluconazole, anidulafungin and caspofungin were analyzed (Defining Antibiotic Levels in Intensive care, DALI study) (46). Fifteen patients received fluconazole, 9 anidulafungin and 6 caspofungin with a median SOFA score of 7, 6 and 3, respectively. Using a fix dose of 400 mg of fluconazole (4.9 mg/kg) the target PK/PD value (fAUCO-24/MIC ≥100) was achieved in 86%, 67% and 13% of patients when the assumed MIC was 1, 2 or 4 mg/L, respectively. Therefore, using a fix 400 mg dose of fluconazole in critically ill patients there is a 33% chance of achieving subtherapeutic levels. Adequate weight-based fluconazole dosing should be used in critically ill patients. In addition, high inter-individual variability was observed for the three antifungal agents included in this study (fluconazole, caspofungin and anidulafungin). Although the number of patients with caspofungin and anidulafungin was limited, in general antifungal exposure was found to be lower than healthy volunteers (46,104-107).
The jury is still out on whether the most adequate in terms of both cost and efficacy empirical antifungal treatment is fluconazole or an echinocandin in the management of a non-neutropenic critically ill patient. Apparently, there is more consensus on the need for a minimum maintenance dose of 6 mg/kg fluconazole, after the initial loading dose (12 mg/kg), a dose which is often not achieved with 400 mg/d (108). Where an echinocandin to be used, recently published data indicate variable PK/PD results among the different members of the class in the critically ill, with potentially better achieved targets in the case of caspofungin judging with better recorded clinical outcomes with its use; its use has limitations, especially in the context of co-existing liver impairment (104).
Obesity
The increasing proportion of critically ill patients with obesity has raised the question if variability of PK parameters commonly seen in critically ill patients may be heightened by the presence of obesity. Alobaid et al. studied population PK of fluconazole and analyzed the effect of body mass index (BMI) to PK parameters in 21 critically ill patients (109). Higher than recommended fixed fluconazole doses, adjusted to 12 mg/kg loading dose and 6 mg/kg/d maintenance dose and further adjusted for renal impairment if present, were needed to achieve better PK/PD targets for Candida strains with an MIC of 2 mg/L. A loading dose seems to improve PK/PD only when it is weight based rather than a fixed dose. Maintenance dose should also be based on mg/kg and be modified according to renal clearance. Similarly, population PK of micafungin in obese critically ill patients showed that a dose higher than recommended (100 mg/day) is needed to treat Candida infections (110). For C. albicans a fix dose of 150 mg/d was found adequate for patients weighing up to 115 kg and of 200 mg/d for those with weight more than 115 kg. Patients with C. glabrata a micafungin dose of 200 mg/d was adequate for obese patients weighing up to 115 kg (110).
Thermal injury
Patients with thermal injury have significant pathophysiological disturbances that may affect PK and PD parameters of antifungal agents (111,112). In most of the cases serum concentrations of antifungals may be subtherapeutic especially in the hypermetabolic phase of thermal injury. However, there are very limited data for each of antifungal agents (111). Most of the available data on antifungal PK parameters in patients with thermal injury are for fluconazole (113,114). In a small number of burn patients Boucher et al. found a 30% increase in fluconazole clearance during the first days of thermal insult (113). Using population pharmacokinetics, Han et al. found that fluconazole dose at 400 mg/day is sufficient in patients with major thermal injury for susceptible isolates (MIC equal or less than 2 µg/mL) (114). For Candida isolates with higher MICs, which may be frequent according to local epidemiology, higher daily doses are recommended, as mentioned previously (114). Among echinocandins, both micafungin and caspofungin have been studied in burn patients (115,116). In a case series of adult patients with severe burns, plasma concentrations of micafungin, was similar or slightly lower than healthy volunteers, when given a high dose of 200–300 mg/day (115,116). Jullien et al. studied PK parameters of caspofungin in two patients with severe burns and found that the patient that had severe hypoalbuminemia had lower caspofungin exposure (50%) of that observed in healthy volunteers (117). More data are needed for patients with thermal injury to optimize antifungal dosing.
Extracorporeal membrane oxygenation (ECMO)
There are few data on antifungal PK during ECMO. Patients on ECMO may experience therapeutic drug failure due to low plasma concentration (high clearance or high volume of distribution) or drug toxicity due to organ dysfunction and decreased clearance (118).
Fluconazole population PK has been studied in children supported with ECMO (119). In this study, which included mainly infants and very few children older than 2 yrs of age, a higher than recommended loading dose is suggested (35 mg/kg instead of 25 mg/kd) for treatment of candidemia followed by standard daily maintenance dose (12 mg/kg) for children supported by ECMO without renal insufficiency or replacement therapy. In addition, according to local practices and especially when invasive candidiasis is more than 10% these patients with ECMO may benefit from fluconazole prophylaxis which should also be administered at higher than recommended dose (12 mg/kg loading dose followed by 6 mg/kg per day) because of the high volume of distribution (119).
Micafungin has been studied in neonates and children aged less than two years of age receiving ECMO (120). In this study infants supported by ECMO had higher volume of micafungin distribution and clearance, and for this reason higher doses (2.5 mg/kg for prophylaxis and 5 mg/kg for treatment) were suggested. This study could not be extrapolated to preterm infants that may have HCME, and higher micafungin doses are used (as previously discussed). In an ex vivo study using different ECMO circuits, micafungin was found to be extracted by the circuit whereas fluconazole was not (121).
Caspofungin administration during ECMO resulted in subtherapeutic plasma levels, even when higher than recommended doses were used in a child (122). Similarly, an adult patient who was on ECMO and received voriconazole, caspofungin and amphotericin B was found to have low or even undetectable concentrations of both caspofungin and voriconazole (123). In contrast, Spriet et al. presented two patients on ECMO, who were on caspofungin and voriconazole and found that caspofungin levels were not affected (59). In these cases voriconazole was affected during ECMO and low levels could be explained by sequestration in the circuit together with altered pharmacokinetics (59). Similarly, in a child supported with ECMO and invasive aspergillosis high voriconazole doses based on TDM were required (124). However, voriconazole as previously described, have significant interindividual PK level variation and the possible effect of ECMO cannot be assessed.
Published data for anidulafungin use in patients on ECMO, are limited to only few case reports and plasma levels were measured only in 1 adult patient with no significant differences (125,126).
It seems that there is a high complexity of interactions between ECMO circuit and antifungal agents (121). More studies are needed on patients receiving ECMO and are treated with antifungal agents including latest ECMO technology/ newer circuits to optimize correct dosage schemes.
ConclusionsOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
As invasive fungal diseases, mostly invasive candidiasis, are a major disease burden in ICU regardless of age of patients, antifungal agents have to be administered optimally taking into account the many differences of the age, underlying conditions and co-existing factors that ICU patients have. Administration of these agents may be challenging due to various factors in patients hospitalized in the intensive care unit including extreme age, hemodynamic alterations and organ dysfunction of sepsis, underlying renal and hepatic insufficiency, thermal injury, excessive weight and modern organ function supporting measures. The physicians caring of these patients should be aware of the impact of such factors on pharmacokinetics and pharmacodynamics of antifungal agents as well as the drug interactions with other co-administered drugs. By having this knowledge they can adjust the regimens of the antifungal agents as well as those of other agents to achieve the best outcome of the critically ill patients.
AcknowledgmentsOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
None.
FootnoteOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
ReferencesOther Section
- Invasive fungal infections in the critically ill pediatric and adult patient
- Antifungal agents: pharmacokinetic, pharmacodynamic and dosage considerations
- Renal insufficiency
- Hepatic insufficiency
- Age-dependent factors
- Various underlying conditions
- Conclusions
- Acknowledgments
- Footnote
- References
- Benedict K, Richardson M, Vallabhaneni S, et al. Emerging issues, challenges, and changing epidemiology of fungal disease outbreaks. Lancet Infect Dis 2017;17:e403-11. [Crossref] [PubMed]
- Colombo AL, de Almeida JN Junior, Slavin MA, et al. Candida and invasive mould diseases in non-neutropenic critically ill patients and patients with haematological cancer. Lancet Infect Dis 2017;17:e344-56. [Crossref] [PubMed]
- Lortholary O, Renaudat C, Sitbon K, et al. Worrisome trends in incidence and mortality of candidemia in intensive care units (Paris area, 2002-2010). Intensive Care Med 2014;40:1303-12. [Crossref] [PubMed]
- Lewis RE, Cahyame-Zuniga L, Leventakos K, et al. Epidemiology and sites of involvement of invasive fungal infections in patients with haematological malignancies: a 20-year autopsy study. Mycoses 2013;56:638-45. [Crossref] [PubMed]
- Bajwa S, Kulshrestha A. Fungal infections in intensive care unit: challenges in diagnosis and management. Ann Med Health Sci Res 2013;3:238-44. [Crossref] [PubMed]
- Prowle JR, Echeverri JE, Ligabo EV, et al. Acquired bloodstream infection in the intensive care unit: incidence and attributable mortality. Crit Care 2011;15:R100. [Crossref] [PubMed]
- Papadimitriou-Olivgeris M, Spiliopoulou A, Fligou F, et al. Risk factors and predictors of mortality of candidaemia among critically ill patients: role of antifungal prophylaxis in its development and in selection of non-albicans species. Infection 2017;45:651-7. [Crossref] [PubMed]
- Pratikaki M, Platsouka E, Sotiropoulou C, et al. Epidemiology, risk factors for and outcome of candidaemia among non-neutropenic patients in a Greek intensive care unit. Mycoses 2011;54:154-61. [Crossref] [PubMed]
- Cavayas YA, Yusuff H, Porter R. Fungal infections in adult patients on extracorporeal life support. Crit Care 2018;22:98. [Crossref] [PubMed]
- Koehler P, Bassetti M, Kochanek M, et al. Intensive Care management of influenza-associated pulmonary aspergillosis. Clin Microbiol Infect 2019. [Epub ahead of print]. [Crossref] [PubMed]
- Hsu JF, Lai MY, Lee CW, et al. Comparison of the incidence, clinical features and outcomes of invasive candidiasis in children and neonates. BMC Infect Dis 2018;18:194. [Crossref] [PubMed]
- Blyth CC, Chen SC, Slavin MA, et al. Not just little adults: candidemia epidemiology, molecular characterization, and antifungal susceptibility in neonatal and pediatric patients. Pediatrics 2009;123:1360-8. [Crossref] [PubMed]
- Bellmann R, Smuszkiewicz P. Pharmacokinetics of antifungal drugs: practical implications for optimized treatment of patients. Infection 2017;45:737-79. [Crossref] [PubMed]
- Andes D, Safdar N, Marchillo K, et al. Pharmacokinetic-pharmacodynamic comparison of amphotericin B (AMB) and two lipid-associated AMB preparations, liposomal AMB and AMB lipid complex, in murine candidiasis models. Antimicrob Agents Chemother 2006;50:674-84. [Crossref] [PubMed]
- Lepak AJ, Andes D. Antifungal pharmacokinetics and pharmacodynamics. Cold Spring Harb Perspect Med 2014;5:a019653. [Crossref] [PubMed]
- Hong Y, Shaw PJ, Nath CE, et al. Population pharmacokinetics of liposomal amphotericin B in pediatric patients with malignant diseases. Antimicrob Agents Chemother 2006;50:935-42. [Crossref] [PubMed]
- Andes D, Stamsted T, Conklin R. Pharmacodynamics of amphotericin B in a neutropenic-mouse disseminated-candidiasis model. Antimicrob Agents Chemother 2001;45:922-6. [Crossref] [PubMed]
- Stott KE, Hope WW. Therapeutic drug monitoring for invasive mould infections and disease: pharmacokinetic and pharmacodynamic considerations. J Antimicrob Chemother 2017;72:i12-8. [Crossref] [PubMed]
- Andes D, van Ogtrop M. In vivo characterization of the pharmacodynamics of flucytosine in a neutropenic murine disseminated candidiasis model. Antimicrob Agents Chemother 2000;44:938-42. [Crossref] [PubMed]
- Caputo R. Itraconazole (Sporanox) in superficial and systemic fungal infections. Expert Rev Anti Infect Ther 2003;1:531-42. [Crossref] [PubMed]
- Sandherr M, Maschmeyer G. Pharmacology and metabolism of voriconazole and Posaconazole in the treatment of invasive aspergillosis: review of the literature. Eur J Med Res 2011;16:139-44. [Crossref] [PubMed]
- Moore JN, Healy JR, Kraft WK. Pharmacologic and clinical evaluation of posaconazole. Expert Rev Clin Pharmacol 2015;8:321-34. [Crossref] [PubMed]
- Wilson DT, Dimondi VP, Johnson SW, et al. Role of isavuconazole in the treatment of invasive fungal infections. Ther Clin Risk Manag 2016;12:1197-206. [Crossref] [PubMed]
- Arrieta A, Frangoul H, Steinbach W, et al., editors. An open-label, phase I, multi-centre study to evaluate the pharmacokinetic, safety and tolerability profile of intravenous isavuconazonium sulfate in paediatric patients. 29th ECCMID; 2019; Amsterdam, The Netherlands.
- Howard SJ, Lestner JM, Sharp A, et al. Pharmacokinetics and pharmacodynamics of posaconazole for invasive pulmonary aspergillosis: clinical implications for antifungal therapy. J Infect Dis 2011;203:1324-32. [Crossref] [PubMed]
- Lepak AJ, Marchillo K, Vanhecker J, et al. Isavuconazole (BAL4815) pharmacodynamic target determination in an in vivo murine model of invasive pulmonary aspergillosis against wild-type and cyp51 mutant isolates of Aspergillus fumigatus. Antimicrob Agents Chemother 2013;57:6284-9. [Crossref] [PubMed]
- Sudan A, Livermore J, Howard SJ, et al. Pharmacokinetics and pharmacodynamics of fluconazole for cryptococcal meningoencephalitis: implications for antifungal therapy and in vitro susceptibility breakpoints. Antimicrob Agents Chemother 2013;57:2793-800. [Crossref] [PubMed]
- Scheven M, Scheven C, Hahn K, et al. Post-antibiotic effect and post-expositional polyene antagonism of azole antifungal agents in Candida albicans: dependence on substance lipophilia. Mycoses 1995;38:435-42. [Crossref] [PubMed]
- Andes D, van Ogtrop M. Characterization and quantitation of the pharmacodynamics of fluconazole in a neutropenic murine disseminated candidiasis infection model. Antimicrob Agents Chemother 1999;43:2116-20. [Crossref] [PubMed]
- Andes D, Marchillo K, Stamstad T, et al. In vivo pharmacokinetics and pharmacodynamics of a new triazole, voriconazole, in a murine candidiasis model. Antimicrob Agents Chemother 2003;47:3165-9. [Crossref] [PubMed]
- Rodríguez-Tudela JL, Almirante B, Rodriguez-Pardo D, et al. Correlation of the MIC and dose/MIC ratio of fluconazole to the therapeutic response of patients with mucosal candidiasis and candidemia. Antimicrob Agents Chemother 2007;51:3599-604. [Crossref] [PubMed]
- Troke PF, Hockey HP, Hope WW. Observational study of the clinical efficacy of voriconazole and its relationship to plasma concentrations in patients. Antimicrob Agents Chemother 2011;55:4782-8. [Crossref] [PubMed]
- Andes D, Pascual A, Marchetti O. Antifungal therapeutic drug monitoring: established and emerging indications. Antimicrob Agents Chemother 2009;53:24-34. [Crossref] [PubMed]
- Kofla G, Ruhnke M. Pharmacology and metabolism of anidulafungin, caspofungin and micafungin in the treatment of invasive candidosis: review of the literature. Eur J Med Res 2011;16:159-66. [Crossref] [PubMed]
- Pfaller MA. Anidulafungin: an echinocandin antifungal. Expert Opin Investig Drugs 2004;13:1183-97. [Crossref] [PubMed]
- Roilides E, Carlesse F, Leister-Tebbe H, et al. A Prospective, Open-label Study to Assess the Safety, Tolerability and Efficacy of Anidulafungin in the Treatment of Invasive Candidiasis in Children 2 to <18 Years of Age. Pediatr Infect Dis J 2019;38:275-9. [Crossref] [PubMed]
- Smith RP, Baltch A, Bopp LH, et al. Post-antifungal effects and time-kill studies of anidulafungin, caspofungin, and micafungin against Candida glabrata and Candida parapsilosis. Diagn Microbiol Infect Dis 2011;71:131-8. [Crossref] [PubMed]
- Odds FC, Motyl M, Andrade R, et al. Interlaboratory comparison of results of susceptibility testing with caspofungin against Candida and Aspergillus species. J Clin Microbiol 2004;42:3475-82. [Crossref] [PubMed]
- Andes D, Diekema DJ, Pfaller MA, et al. In vivo comparison of the pharmacodynamic targets for echinocandin drugs against Candida species. Antimicrob Agents Chemother 2010;54:2497-506. [Crossref] [PubMed]
- Wiederhold NP, Kontoyiannis DP, Chi J, et al. Pharmacodynamics of caspofungin in a murine model of invasive pulmonary aspergillosis: evidence of concentration-dependent activity. J Infect Dis 2004;190:1464-71. [Crossref] [PubMed]
- Katragkou A, Roilides E, Walsh TJ. Role of Echinocandins in Fungal Biofilm-Related Disease: Vascular Catheter-Related Infections, Immunomodulation, and Mucosal Surfaces. Clin Infect Dis 2015;61 Suppl 6:S622-9. [Crossref] [PubMed]
- Felton T, Troke PF, Hope WW. Tissue penetration of antifungal agents. Clin Microbiol Rev 2014;27:68-88. [Crossref] [PubMed]
- Li C, Wang H, Yin M, et al. The Differences in the Epidemiology and Predictors of Death between Candidemia Acquired in Intensive Care Units and Other Hospital Settings. Intern Med 2015;54:3009-16. [Crossref] [PubMed]
- Ylipalosaari P, Ala-Kokko TI, Karhu J, et al. Comparison of the epidemiology, risk factors, outcome and degree of organ failures of patients with candidemia acquired before or during ICU treatment. Crit Care 2012;16:R62. [Crossref] [PubMed]
- Motta FA, Dalla-Costa LM, Muro MD, et al. Risk factors for candidemia mortality in hospitalized children. J Pediatr (Rio J) 2017;93:165-71. [Crossref] [PubMed]
- Sinnollareddy MG, Roberts JA, Lipman J, et al. Pharmacokinetic variability and exposures of fluconazole, anidulafungin, and caspofungin in intensive care unit patients: Data from multinational Defining Antibiotic Levels in Intensive care unit (DALI) patients Study. Crit Care 2015;19:33. [Crossref] [PubMed]
- Patel K, Roberts JA, Lipman J, et al. Population pharmacokinetics of fluconazole in critically ill patients receiving continuous venovenous hemodiafiltration: using Monte Carlo simulations to predict doses for specified pharmacodynamic targets. Antimicrob Agents Chemother 2011;55:5868-73. [Crossref] [PubMed]
- Sime FB, Stuart J, Butler J, et al. A pharmacokinetic case study of intravenous posaconazole in a critically ill patient with hypoalbuminaemia receiving continuous venovenous haemodiafiltration. Int J Antimicrob Agents 2018;52:506-9. [Crossref] [PubMed]
- Bassetti M, Garnacho-Montero J, Calandra T, et al. Intensive care medicine research agenda on invasive fungal infection in critically ill patients. Intensive Care Med 2017;43:1225-38. [Crossref] [PubMed]
- Alvarez C, Shin DH, Kwon GS. Reformulation of Fungizone by PEG-DSPE Micelles: Deaggregation and Detoxification of Amphotericin B. Pharm Res 2016;33:2098-106. [Crossref] [PubMed]
- Sinnollareddy M, Peake SL, Roberts MS, et al. Using pharmacokinetics and pharmacodynamics to optimise dosing of antifungal agents in critically ill patients: a systematic review. Int J Antimicrob Agents 2012;39:1-10. [Crossref] [PubMed]
- Pérez-Pitarch A, Ferriols-Lisart R, Aguilar G, et al. Dosing of caspofungin based on a pharmacokinetic/pharmacodynamic index for the treatment of invasive fungal infections in critically ill patients on continuous venovenous haemodiafiltration. . Int J Antimicrob Agents 2018;51:115-21. [Crossref] [PubMed]
- Cornely OA, Bassetti M, Calandra T, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients. Clin Microbiol Infect 2012;18 Suppl 7:19-37. [Crossref] [PubMed]
- Pea F, Lewis RE. Overview of antifungal dosing in invasive candidiasis. J Antimicrob Chemother 2018;73:i33-43. [Crossref] [PubMed]
- Cousin L, Berre ML, Launay-Vacher V, et al. Dosing guidelines for fluconazole in patients with renal failure. Nephrol Dial Transplant 2003;18:2227-31. [Crossref] [PubMed]
- Trotman RL, Williamson JC, Shoemaker DM, et al. Antibiotic dosing in critically ill adult patients receiving continuous renal replacement therapy. Clin Infect Dis 2005;41:1159-66. [Crossref] [PubMed]
- Gharibian KN, Mueller BA. Fluconazole dosing predictions in critically-ill patients receiving prolonged intermittent renal replacement therapy: a Monte Carlo simulation approach. Clin Nephrol 2016;86:43-50. [Crossref] [PubMed]
- Peng LW, Lien YH. Pharmacokinetics of single, oral-dose voriconazole in peritoneal dialysis patients. Am J Kidney Dis 2005;45:162-6. [Crossref] [PubMed]
- Spriet I, Annaert P, Meersseman P, et al. Pharmacokinetics of caspofungin and voriconazole in critically ill patients during extracorporeal membrane oxygenation. J Antimicrob Chemother 2009;63:767-70. [Crossref] [PubMed]
- Kiser TH, Fish DN, Aquilante CL, et al. Evaluation of sulfobutylether-beta-cyclodextrin (SBECD) accumulation and voriconazole pharmacokinetics in critically ill patients undergoing continuous renal replacement therapy. Crit Care 2015;19:32. [Crossref] [PubMed]
- Roger C, Wallis SC, Muller L, et al. Caspofungin Population Pharmacokinetics in Critically Ill Patients Undergoing Continuous Veno-Venous Haemofiltration or Haemodiafiltration. Clin Pharmacokinet 2017;56:1057-68. [Crossref] [PubMed]
- Aguilar G, Azanza JR, Carbonell JA, et al. Anidulafungin dosing in critically ill patients with continuous venovenous haemodiafiltration. J Antimicrob Chemother 2014;69:1620-3. [Crossref] [PubMed]
- Paiva JA, Pereira JM, Tabah A, et al. Characteristics and risk factors for 28-day mortality of hospital acquired fungemias in ICUs: data from the EUROBACT study. Crit Care 2016;20:53. [Crossref] [PubMed]
- Spernovasilis N, Kofteridis DP. Pre-Existing Liver Disease and Toxicity of Antifungals. J Fungi (Basel) 2018;4. [Crossref] [PubMed]
- Theocharidou E, Agarwal B, Jeffrey G, et al. Early invasive fungal infections and colonization in patients with cirrhosis admitted to the intensive care unit. Clin Microbiol Infect 2016;22:189.e1-189.e7. [Crossref] [PubMed]
- Bajaj JS, Reddy RK, Tandon P, et al. Prediction of Fungal Infection Development and Their Impact on Survival Using the NACSELD Cohort. Am J Gastroenterol 2018;113:556-63. [Crossref] [PubMed]
- Alempijevic T, Zec S, Milosavljevic T. Drug-induced liver injury: Do we know everything? World J Hepatol 2017;9:491-502. [Crossref] [PubMed]
- Doß S, Potschka H, Doss F, et al. Hepatotoxicity of Antimycotics Used for Invasive Fungal Infections: In Vitro Results. Biomed Res Int 2017;2017:9658018. [Crossref] [PubMed]
- Kyriakidis I, Tragiannidis A, Munchen S, et al. Clinical hepatotoxicity associated with antifungal agents. Expert Opin Drug Saf 2017;16:149-65. [PubMed]
- Raschi E, Poluzzi E, Koci A, et al. Assessing liver injury associated with antimycotics: Concise literature review and clues from data mining of the FAERS database. World J Hepatol 2014;6:601-12. [Crossref] [PubMed]
- Tverdek FP, Heo ST, Aitken SL, et al. Real-Life Assessment of the Safety and Effectiveness of the New Tablet and Intravenous Formulations of Posaconazole in the Prophylaxis of Invasive Fungal Infections via Analysis of 343 Courses. Antimicrob Agents Chemother 2017;61. [Crossref] [PubMed]
- Stone NR, Bicanic T, Salim R, et al. Liposomal Amphotericin B (AmBisome((R))): A Review of the Pharmacokinetics, Pharmacodynamics, Clinical Experience and Future Directions. Drugs 2016;76:485-500. [Crossref] [PubMed]
- Chamilos G, Luna M, Lewis RE, et al. Effects of liposomal amphotericin B versus an amphotericin B lipid complex on liver histopathology in patients with hematologic malignancies and invasive fungal infections: a retrospective, nonrandomized autopsy study. Clin Ther 2007;29:1980-6. [Crossref] [PubMed]
- Yeoh SF, Lee TJ, Chew KL, et al. Echinocandins for management of invasive candidiasis in patients with liver disease and liver transplantation. Infect Drug Resist 2018;11:805-19. [Crossref] [PubMed]
- Nadim MK, Durand F, Kellum JA, et al. Management of the critically ill patient with cirrhosis: A multidisciplinary perspective. J Hepatol 2016;64:717-35. [Crossref] [PubMed]
- Bassetti M, Peghin M, Carnelutti A, et al. Clinical characteristics and predictors of mortality in cirrhotic patients with candidemia and intra-abdominal candidiasis: a multicenter study. Intensive Care Med 2017;43:509-18. [Crossref] [PubMed]
- European Medicines Agency. Mycamine. 2011. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/mycamine
- Dekkers BGJ, Veringa A, Marriott DJE, et al. Invasive Candidiasis in the Elderly: Considerations for Drug Therapy. Drugs Aging 2018;35:781-9. [Crossref] [PubMed]
- Barchiesi F, Orsetti E, Mazzanti S, et al. Candidemia in the elderly: What does it change? PLoS One 2017;12:e0176576. [Crossref] [PubMed]
- Matthaiou DK, Dimopoulos G, Taccone FS, et al. Elderly versus nonelderly patients with invasive aspergillosis in the ICU: a comparison and risk factor analysis for mortality from the AspICU cohort. Med Mycol 2018;56:668-78. [Crossref] [PubMed]
- Mangoni AA, Jackson SH. Age-related changes in pharmacokinetics and pharmacodynamics: basic principles and practical applications. Br J Clin Pharmacol 2004;57:6-14. [Crossref] [PubMed]
- Niwa T, Imagawa Y, Yamazaki H. Drug interactions between nine antifungal agents and drugs metabolized by human cytochromes P450. Curr Drug Metab 2014;15:651-79. [Crossref] [PubMed]
- Brüggemann RJ, Alffenaar JW, Blijlevens NM, et al. Clinical relevance of the pharmacokinetic interactions of azole antifungal drugs with other coadministered agents. Clin Infect Dis 2009;48:1441-58. [Crossref] [PubMed]
- Walsh TJ, Katragkou A, Chen T, et al. Invasive Candidiasis in Infants and Children: Recent Advances in Epidemiology, Diagnosis, and Treatment. J Fungi (Basel) 2019;5. [Crossref] [PubMed]
- Hope WW, Castagnola E, Groll AH, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: prevention and management of invasive infections in neonates and children caused by Candida spp. Clin Microbiol Infect 2012;18 Suppl 7:38-52. [Crossref] [PubMed]
- Pappas PG, Kauffman CA, Andes DR, et al. Executive Summary: Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis 2016;62:409-17. [Crossref] [PubMed]
- Iosifidis E, Papachristou S, Roilides E. Advances in the Treatment of Mycoses in Pediatric Patients. J Fungi (Basel) 2018;4. [Crossref] [PubMed]
- Turkova A, Roilides E, Sharland M. Amphotericin B in neonates: deoxycholate or lipid formulation as first-line therapy - is there a 'right' choice? Curr Opin Infect Dis 2011;24:163-71. [Crossref] [PubMed]
- Benedict K, Roy M, Kabbani S, et al. Neonatal and Pediatric Candidemia: Results From Population-Based Active Laboratory Surveillance in Four US Locations, 2009-2015. J Pediatric Infect Dis Soc 2018;7:e78-85. [Crossref] [PubMed]
- Warris A, Pana ZD, Oletto ARL, et al., editors. Antifungal drug susceptibility of candida spp. in neonatal and paediatric candidaemia: A european multi-centre retrospective study (EUROCANDY). Proceedings of 36th Annual Meeting of The European Society For Pediatric Infectious Diseases ESPID; 2018; Malmö, Sweden.
- Manzoni P, Benjamin DK Jr, Franco C, et al. Echinocandins for the nursery: an update. Curr Drug Metab 2013;14:203-7. [PubMed]
- Manzoni P, Wu C, Tweddle L, et al. Micafungin in premature and non-premature infants: a systematic review of 9 clinical trials. Pediatr Infect Dis J 2014;33:e291-8. [Crossref] [PubMed]
- Wasmann RE, Muilwijk EW, Burger DM, et al. Clinical Pharmacokinetics and Pharmacodynamics of Micafungin. Clin Pharmacokinet 2018;57:267-86. [Crossref] [PubMed]
- Kovanda LL, Walsh TJ, Benjamin DK Jr, et al. Exposure-Response Analysis of Micafungin in Neonatal Candidiasis: Pooled Analysis of Two Clinical Trials. Pediatr Infect Dis J 2018;37:580-5. [Crossref] [PubMed]
- Somer A, Torun SH, Salman N. Caspofungin therapy in immunocompromised children and neonates. Expert Rev Anti Infect Ther 2011;9:347-55. [Crossref] [PubMed]
- Sáez-Llorens X, Macias M, Maiya P, et al. Pharmacokinetics and safety of caspofungin in neonates and infants less than 3 months of age. Antimicrob Agents Chemother 2009;53:869-75. [Crossref] [PubMed]
- Tsekoura M, Ioannidou M, Pana ZD, et al. Efficacy and Safety of Echinocandins for the Treatment of Invasive Candidiasis in Children: A Meta-analysis. Pediatr Infect Dis J 2019;38:42-9. [Crossref] [PubMed]
- Kuyucu N. Amphotericin B use in children: conventional and lipid-based formulations. Expert Rev Anti Infect Ther 2011;9:357-67. [Crossref] [PubMed]
- Ullmann AJ, Aguado JM, Arikan-Akdagli S, et al. Diagnosis and management of Aspergillus diseases: executive summary of the 2017 ESCMID-ECMM-ERS guideline. Clin Microbiol Infect 2018;24 Suppl 1:e1-38. [Crossref] [PubMed]
- McCarthy MW, Katragkou A, Iosifidis E, et al. Recent Advances in the Treatment of Scedosporiosis and Fusariosis. J Fungi (Basel) 2018;4. [Crossref] [PubMed]
- Tortorano AM, Richardson M, Roilides E, et al. ESCMID and ECMM joint guidelines on diagnosis and management of hyalohyphomycosis: Fusarium spp., Scedosporium spp. and others. Clin Microbiol Infect 2014;20 Suppl 3:27-46. [Crossref] [PubMed]
- Friberg LE, Ravva P, Karlsson MO, et al. Integrated population pharmacokinetic analysis of voriconazole in children, adolescents, and adults. Antimicrob Agents Chemother 2012;56:3032-42. [Crossref] [PubMed]
- Doby EH, Benjamin DK Jr, Blaschke AJ, et al. Therapeutic monitoring of voriconazole in children less than three years of age: a case report and summary of voriconazole concentrations for ten children. Pediatr Infect Dis J 2012;31:632-5. [Crossref] [PubMed]
- Yang Q, Wang T, Xie J, et al. Pharmacokinetic/pharmacodynamic adequacy of echinocandins against Candida spp. in intensive care unit patients and general patient populations. Int J Antimicrob Agents 2016;47:397-402. [Crossref] [PubMed]
- van der Elst KC, Veringa A, Zijlstra JG, et al. Low Caspofungin Exposure in Patients in Intensive Care Units. Antimicrob Agents Chemother 2017;61. [Crossref] [PubMed]
- Brüggemann RJ, Middel-Baars V, de Lange DW, et al. Pharmacokinetics of Anidulafungin in Critically Ill Intensive Care Unit Patients with Suspected or Proven Invasive Fungal Infections. Antimicrob Agents Chemother 2017;61. [Crossref] [PubMed]
- Jullien V, Azoulay E, Schwebel C, et al. Population pharmacokinetics of micafungin in ICU patients with sepsis and mechanical ventilation. J Antimicrob Chemother 2017;72:181-9. [Crossref] [PubMed]
- Martin-Loeches I, Antonelli M, Cuenca-Estrella M, et al. ESICM/ESCMID task force on practical management of invasive candidiasis in critically ill patients. Intensive Care Med 2019;45:789-805. [Crossref] [PubMed]
- Alobaid AS, Wallis SC, Jarrett P, et al. Effect of Obesity on the Population Pharmacokinetics of Fluconazole in Critically Ill Patients. Antimicrob Agents Chemother 2016;60:6550-7. [Crossref] [PubMed]
- Maseda E, Grau S, Luque S, et al. Population pharmacokinetics/pharmacodynamics of micafungin against Candida species in obese, critically ill, and morbidly obese critically ill patients. Crit Care 2018;22:94. [Crossref] [PubMed]
- Ortwine JK, Pogue JM, Faris J. Pharmacokinetics and pharmacodynamics of antibacterial and antifungal agents in adult patients with thermal injury: a review of current literature. J Burn Care Res 2015;36:e72-84. [Crossref] [PubMed]
- Cota JM. Intravenous Antibiotic and Antifungal Agent Pharmacokinetic-Pharmacodynamic Dosing in Adults with Severe Burn Injury. Clin Ther 2016;38:2016-31. [Crossref] [PubMed]
- Boucher BA, King SR, Wandschneider HL, et al. Fluconazole pharmacokinetics in burn patients. Antimicrob Agents Chemother 1998;42:930-3. [Crossref] [PubMed]
- Han S, Kim J, Yim H, et al. Population pharmacokinetic analysis of fluconazole to predict therapeutic outcome in burn patients with Candida infection. Antimicrob Agents Chemother 2013;57:1006-11. [Crossref] [PubMed]
- Sasaki J, Yamanouchi S, Kudo D, et al. Micafungin concentrations in the plasma and burn eschar of severely burned patients. Antimicrob Agents Chemother 2012;56:1113-5. [Crossref] [PubMed]
- Sasaki J, Yamanouchi S, Sato Y, et al. Penetration of micafungin into the burn eschar in patients with severe burns. Eur J Drug Metab Pharmacokinet 2014;39:93-7. [Crossref] [PubMed]
- Jullien V, Blanchet B, Benyamina M, et al. Pharmacokinetics of caspofungin in two patients with burn injuries. Antimicrob Agents Chemother 2012;56:4550-1. [Crossref] [PubMed]
- Tukacs M. Pharmacokinetics and Extracorporeal Membrane Oxygenation in Adults: A Literature Review. AACN Adv Crit Care 2018;29:246-58. [Crossref] [PubMed]
- Watt KM, Gonzalez D, Benjamin DK Jr, et al. Fluconazole population pharmacokinetics and dosing for prevention and treatment of invasive Candidiasis in children supported with extracorporeal membrane oxygenation. Antimicrob Agents Chemother 2015;59:3935-43. [Crossref] [PubMed]
- Autmizguine J, Hornik CP, Benjamin DK Jr, et al. Pharmacokinetics and Safety of Micafungin in Infants Supported With Extracorporeal Membrane Oxygenation. Pediatr Infect Dis J 2016;35:1204-10. [Crossref] [PubMed]
- Watt KM, Cohen-Wolkowiez M, Williams DC, et al. Antifungal Extraction by the Extracorporeal Membrane Oxygenation Circuit. J Extra Corpor Technol 2017;49:150-9. [PubMed]
- Koch BC, Wildschut ED, Goede AL, et al. Insufficient serum caspofungin levels in a paediatric patient on ECMO. Med Mycol Case Rep 2012;2:23-4. [Crossref] [PubMed]
- Ruiz S, Papy E, Da Silva D, et al. Potential voriconazole and caspofungin sequestration during extracorporeal membrane oxygenation. Intensive Care Med 2009;35:183-4. [Crossref] [PubMed]
- Brüggemann RJ, Antonius T, Heijst A, et al. Therapeutic drug monitoring of voriconazole in a child with invasive aspergillosis requiring extracorporeal membrane oxygenation. Ther Drug Monit 2008;30:643-6. [Crossref] [PubMed]
- Aguilar G, Ferriols R, Carbonell JA, et al. Pharmacokinetics of anidulafungin during venovenous extracorporeal membrane oxygenation. Crit Care 2016;20:325. [Crossref] [PubMed]
- Parcell BJ. Invasive pulmonary aspergillosis post extracorporeal membrane oxygenation support and literature review. Med Mycol Case Rep 2014;4:12-5. [Crossref] [PubMed]
Cite this article as: Pyrpasopoulou A, Iosifidis E, Antachopoulos C, Roilides E. Antifungal drug dosing adjustment in critical patients with invasive fungal infections. J Emerg Crit Care Med 2019;3:37.


