
Hemodynamic monitoring of ARDS by critical care echocardiography
Introduction
Critical care echocardiography (CCE) is more and more frequently used and even more than the pulmonary artery catheter in patients with sepsis (1). In acute respiratory distress syndrome (ARDS) patients, same result was reported in a French database (2). CCE is very suitable for the management of patients with respiratory and circulatory failure (3), as it may directly modify diagnostic and therapeutic procedures. In 2,508 transesophageal echocardiography (TEE) studies, the main indication was hemodynamic instability and findings were left ventricular (LV) dysfunction in 27%, right ventricular (RV) dysfunction in 11% and hypovolemia in 16% of cases. TEE had also a significant on diagnostic and treatment, either surgical or changes in medical therapy (4).
A consensus of experts in the field of hemodynamic monitoring recognized CCE as a true hemodynamic monitoring device, albeit discontinuous (5). An echo study has to be done as soon as possible in the case of hemodynamic instability, if the patient is not obviously fluid-responsive with low central venous pressure (5). TEE is probably less operator-dependent than transthoracic echocardiography (TTE), the reason why TEE is better than TTE for hemodynamic monitoring when performed in intubated patients; it then allows reproducible and sequential hemodynamic assessments (6). For an accurate hemodynamic evaluation, it has been reported that intensivists may use TEE in a qualitative approach based on 4 main parameters: respiratory variation of the superior vena cava (SVC), LV systolic function, RV size, and paradoxical septal motion (7).
CCE is therefore key in patients developing ARDS and this review illustrates and discusses the main reasons why, which are defining the cause of circulatory failure, optimizing hemodynamic and respiratory management by looking at the effect of mechanical ventilation on RV function which requires good knowledge of heart-lung interactions, and managing veno-venous extracorporeal membrane oxygenation (VV ECMO), if required.
ARDS and circulatory failure
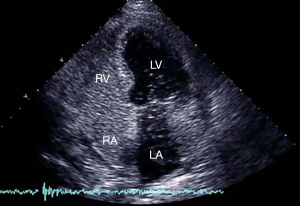
Fifty years after its first description by Ashbaugh et al. (8), the management of ARDS remains a challenge for intensivists. Despite advances in supportive care during the last ten years, ARDS is still associated with high mortality and morbidity. In a recent large multicenter prospective cohort study conducted in 50 countries in different continents, the prevalence of ARDS was 10.4% among admissions to the ICU, with an in-hospital mortality of 40% (9). ARDS is frequently associated with hemodynamic instability. In one study more than 60% of patients experience shock (10) and in other studies around 65% require infusion of catecholamines (11,12). Circulatory failure appears as the main factor associated with mortality (13). The definition of ARDS recently revisited by an expert panel (14) is still debated because neither the stratification by severity nor the PaO2/FiO2 ratio was independently associated with mortality (15). In the case of shock, the PaO2/FiO2 ratio as a marker of severity remains questionable because some hemodynamic effects can interact. For example, the low PvO2 effect or the reopening of a patent foramen ovale (Figure 1) tends to overestimate the severity of ARDS. Conversely, a low cardiac index may decrease lung shunt, leading to underestimation of severity. In this way, refractory hypoxemia is rarely the cause of death and most patients die from shock and multiorgan failure (9).
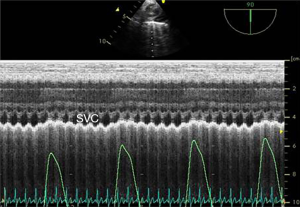
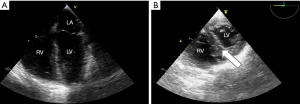
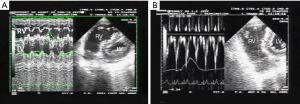
In ARDS, shock is schematically driven by two main mechanisms. On the one hand, there is an obstructive mechanism due to the effect of pulmonary hypertension on RV function, resulting from alteration of the pulmonary capillary circulation (16) and the deleterious effects of mechanical ventilation (MV) (17). On the other hand, sepsis-related circulatory failure occurs in half of patients (18). In this case, it may be associated with relative or absolute hypovolemia, vasoplegia, and myocardial depression (19), and the role of CCE is in part discussed by De Backer and colleagues in another paper of this issue dedicated to critical care ultrasonography. Briefly, the complexity and heterogeneity of patients with septic shock implies individualized approaches to hemodynamic management and CCE is very suitable as it may detect combined mechanisms of circulatory failure. A recent study using a clustering approach based on clinical and CCE parameters in a large population of septic shock patients characterized different cardiovascular phenotypes. Five different clusters were defined: well resuscitated, LV systolic dysfunction, LV hyperkinesia, hypovolemia, and right ventricular failure. This new approach to the characterization of shock is especially interesting as it could help physicians to optimize hemodynamic support quickly at the bedside (20). The only specificity in ARDS is the application of a low tidal volume which may limit the accuracy of the parameters of fluid responsiveness, while the largest study in the field included 22% of ARDS and most patients were ventilated with a tidal volume below 8 mL/kg (21). This study reported respiratory variations of the SVC as the most specific parameter of fluid responsiveness (21) (Figure 2). ARDS-related RV failure is the main mechanism of shock for the remaining unstable ARDS patients. It is related to an abrupt increase in pulmonary artery pressure. Most studies report the incidence of acute cor pulmonale (ACP), echocardiographically defined by RV dilatation with paradoxical septal motion. However, based on the physiology of the right ventricle, experts have rather defined RV failure as a “significant” RV dilatation leading to systemic venous congestion (22,23), which may be highly suspected by CCE (Figure 3A). Paradoxical septal motion, when present, reflects the fact that this RV failure is mediated by systolic overload (Figure 3B). The prevalence of ACP during ARDS reaches 20–25% in studies where patients are ventilated with a lung protective approach; its occurrence has been reported to be associated with higher mortality (24,25). Nevertheless, both mechanisms of circulatory failure in ARDS, sepsis and pulmonary hypertension, are often interlinked since pneumonia is the first cause of ARDS and has also been identified as an independent risk factor for ACP (10).
Hemodynamic monitoring in ARDS by CCE
CCE has a growing role in the management of ARDS patients, mainly because of the huge interactions between the lung (and then ventilation) and the right ventricle. The choice of the modality of echocardiography (transthoracic, TTE versus TEE) strictly depends on the clinical condition of individual patients, on the skills of the operator, as well as on the clinical inquiry. If possible, TEE should be preferred since it overcomes technical problems associated with acoustic views and measurements can usually be performed easily (26). Since patients with severe ARDS often require prolonged periods of prone positioning, TEE has also be proven to be performed safely and efficiently in this situation (27). Finally, it has been reported that TEE is more accurate than TTE in reporting ACP (24) and that factors limiting the feasibility of TTE are high PEEP or significant weight gain (28), both of which are frequently observed in ARDS.
During ARDS, assessment of RV size is a key objective. It may be easily reached by comparing the RV end-diastolic area (RVEDA) with the LV end-diastolic area (LVEDA) (26). An RVEDA/LVEDA ratio between 0.6 and 1 indicates moderate RV dilatation, while an RVEDA/LVEDA ratio >1 indicates severe RV dilatation. ACP has been defined as the combination of an RVEDA/LVEDA ratio >0.6 and the presence of paradoxical septal motion during end-systole (24) (Figure 3). LV ejection fraction should also be assessed and clinically interpreted taking into account the dosage of inotropic and vasoactive drugs, if administered. Valvular diseases (especially mitral and aortic regurgitation) should also be investigated as they could influence the course and severity of the lung disease and influence treatments, as for fluid requirement. Since a patent foramen ovale may worsen hypoxemia in ARDS patients, echocardiographic assessment should establish whether or not it is present, and, if so, its severity (Figure 1). Patent foramen ovale has recently been reported to concern between 16 and 19% of ARDS patients (29).
Because of positive pressure ventilation on the one hand, and the dramatic decrease in lung compliance on the other hand, ARDS patients are especially subject to wide variations of airway pressure, especially alveolar and transpulmonary pressures. Some experts have recently emphasized the pivotal role of evaluating such interactions for hemodynamic assessment of patients suffering from ARDS (30). They proposed combining continuous invasive blood pressure monitoring with CCE and the use of pulse pressure variation (PPV) to dichotomize ARDS patients (30). As CCE only allows discontinuous hemodynamic monitoring, it has to be associated with a more continuous modality to have real-time monitoring of blood pressure which can be used as a “warning” signal in the case of significant PPV. From a practical point of view, the absence of any PPV indicates that the circulatory system (both LV and RV) is not altered by the effect of mechanical ventilation. Conversely, significant PPV mainly illustrates the effects of tidal inflation on the right ventricle and forces physicians to explore the underlying mechanisms further by performing CCE. Thereafter, two different situations well characterized by CCE may explain PPV: a decrease in RV preload or an increase in RV afterload. In Figure 4, we propose an algorithm of hemodynamic management based on the association of blood pressure monitoring and CCE.
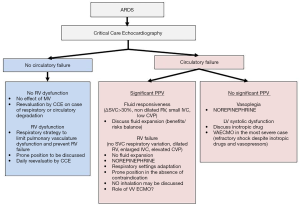
Decrease in RV preload during tidal ventilation is the consequence of the decrease in systemic venous return because of the increase in intrathoracic pressure, suggesting that the patient is fluid-responsive. Management of fluids is still the subject of intense debate in the intensive care community. Besides the fact that hypovolemia when not corrected may promote shock and organ hypoperfusion, and is a confounder for severity evaluation as previously discussed, the main issue is identification of those fluid-responsive patients who would really benefit from more fluid, bearing in mind that positive fluid balance is associated with a worse outcome in ARDS (31,32). Although it does not decrease mortality, fluid restriction improves oxygenation and lung injury score (33,34). For such tricky management, respiratory variation of the SVC (ΔSVC) evaluated by TEE is especially interesting as it could reflect the conflict between the amount of blood in the thorax and the respiratory settings. Furthermore, ΔSVC, when adequately used, is much more specific than sensitive (21) (Figure 2). In cases of a non-dilated right ventricle, significant ΔSVC, and low central venous pressure (or small inferior vena cava by a subcostal view), PPV can help physicians to gauge the benefits and risks of fluid expansion (30).
PPV can also illustrate the increase in RV afterload driven by high transpulmonary pressure. Decreased lung compliance leads to a significant increase in transpulmonary pressure, i.e., plateau pressure minus intrathoracic pressure (35,36). Increase in transpulmonary pressure and RV afterload are well correlated (37,38) and ARDS patients are then exposed to the risk of RV failure (10). In this situation, PPV, when observed, is classically described by CCE as the association of a decrease in RV stroke volume with RV dilatation (either cyclic during tidal ventilation or constant along the respiratory cycle), dilated inferior vena cava, high central venous pressure (CVP), and no or minimal ∆SVC (30) (Figure 5). In these cases, PPV, when significant, becomes a false positive for fluid responsiveness (39) and must lead to adaptation of the respiratory settings, the control of hypercapnia, or discussion of prone positioning of the patient, while more fluid expansion is not indicated or even contraindicated as it can worsen RV failure (40).
Furthermore, CCE is not performed blindly but rather because of abnormal vital signs suggesting poor organ perfusion (skin mottling, elevated lactate, oliguria, and so on). Moreover, CVP, when combined with CCE, may be informative, low CVP suggesting a potentially underfilled right ventricle and high CVP an overloaded right ventricle. CCE allows a paradigm shift from invasive, quantitative, and continuous hemodynamic monitoring to less invasive, qualitative, discontinuous and functional monitoring (41,42). This is why a single measurement of cardiac output is probably less informative than serial evaluations, before/after fluids, before/after dobutamine, before/after application of a high PEEP, or before/after prone positioning. For this latter indication, CCE visualizes the improvement of RV function following prone positioning (43). In a systematic review, Wetterslev et al. reported that cardiac output measurements using either echocardiography or thermodilution were not interchangeable, while trends were (42). According to our experience, we suggest that CCE be performed on ICU admission in ARDS patients to provide valuable information, not only to evaluate the clinical status of that moment but also to highlight clinical elements indicating previous diseases (valvular disease, pre-existing chronic heart disease…). Thereafter, CCE should be periodically repeated according to the clinical course, but certainly should be done at least once a day. Besides, patients should be re-evaluated with CCE whenever higher ventilatory support is required or any new undifferentiated state of shock develops or a significant PPV “warning” signal is present. Although not yet validated in a randomized controlled study, an RV protective approach in part based on the evaluation of RV function by CCE has recently been formalized, putting the pulmonary circulation and the right ventricle at the center of the decision-making process, by correcting the risk factors for developing RV failure, a PaO2/FiO2 ratio <150 mmHg, a driving pressure ≥18 cmH2O, and a PaCO2 ≥48 mmHg (10).
Potential role of CCE in managing extracorporeal membrane oxygenation
ECMO has been known since the 1970’s, but its use has exploded in the last 10 years. Although its value in ARDS is still questionable, some experts suggest that VV ECMO could represent an effective bridge to recovery in patients with very severe ARDS and to standard medical management including prone positioning in whom either hypoxemia is refractory or lung protective ventilation is no longer feasible (44). VV ECMO could then facilitate the use of more protective ventilation by removing carbon dioxide from the blood and increasing blood oxygenation (45). CCE may be used for different objectives: (I) to evaluate RV function before ECMO, (II) to evaluate RV function during ECMO so as to see whether the previously failing right ventricle is fully unloaded, (III) to check that the cannula is properly positioned, (IV) to monitor cardiac output as during ECMO most devices based on thermodilution cannot be accurately used.
To date, there is a paucity of data on cardiac function assessed by CCE in patients with ARDS unresponsive to conventional therapy and then submitted to VV ECMO. In a recent prospective study of echocardiographic evaluation before ECMO initiation, 34% of the patients showed normal findings, 43% exhibited isolated pulmonary hypertension, and 23% showed RV dilation and pulmonary hypertension (46). VV ECMO has been proven to reduce RV afterload (47). In 13 consecutive patients with severe respiratory failure requiring VV ECMO, Miranda et al. reported that pulmonary artery pressure dropped significantly immediately after starting VV ECMO before ventilator settings were altered, followed by a slight drop in CVP and an increase in cardiac index, with stable doses of vasopressors (48). Both oxygenation and decarboxylation play a role here in unloading the right ventricle. Therefore, the presence of pulmonary hypertension in patients with severe ARDS may not be a contraindication for VV ECMO, as this may even alleviate RV failure related to the pulmonary hypertension. Those results, combined with the negative experience of venoarterial (VA) ECMO for ARDS during the 1980’s, explain the fact that most refractory ARDS patients are now initiated on VV ECMO, when required. This is illustrated by a study based on the ELSO registry, in which only 18% of ARDS patients with shock (defined by the need for one or more inotropes/vasopressors) were started on VA ECMO, and this percentage seemed to drop over the years (49). The need for a conversion from VV to VA ECMO in this registry is reported as only 4.1% (49). The same results were observed in the recently published Eolia trial, in which only 6% of the patients receiving VV ECMO were switched to VA or conversely (44). However, VA ECMO could certainly be considered when ARDS is associated with severe RV failure leading to “obstructive” shock (30) (or when combined with severe septic cardiomyopathy leading to very low cardiac output and severely reduced LV ejection fraction despite adequate use of inotropic support and/or norepinephrine (50).
Conclusions
CCE is an incredible technique for hemodynamic monitoring in ARDS. It may allow the independent determination of the need for more fluid, for norepinephrine or dobutamine infusion, but also for adjustment of the mechanical ventilation settings and respiratory strategy. In the most severe cases, CCE affords support to the setting up and tailoring of ECMO, if used. Since CCE is never performed blindly, intensivists should remember that it helps improve patient management and is not a goal in itself. It means that intensivists should not treat an “abnormal” echo picture, but rather interpret echo studies in light of the clinical situation. CCE should always be associated with clinical and laboratory data, continuous invasive monitoring of blood pressure, and a central venous catheter in severe cases.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Papolos A, Narula J, Bavishi C, et al. U.S. Hospital use of echocardiography. Insights from the nationwide inpatient sample. J Am Coll Cardiol 2016;67:502-11. [Crossref] [PubMed]
- Dres M, Austin P, Pham T, et al. CUB-REA Group. Acute Respiratory Distress Syndrome cases volume and Intensive Care Unit mortality in Medical Patients. Crit Care Med 2018;46:e33-40. [Crossref] [PubMed]
- De Backer D, Cholley B, Slama M, et al. Hemodynamic monitoring using echocardiography in the critically ill. Springer-Verlag Berlin Heidelberg 2011.
- Hüttemann E, Schelenz C, Kara F, et al. The use and safety of transesophageal echocardiography in the general ICU – a minireview. Acta Anaesthesiol Scand 2004;48:827-36. [Crossref] [PubMed]
- Vincent JL, Rhodes A, Perel A, et al. Clinical review: update on hemodynamic monitoring – a consensus of 16. Crit Care 2011;15:229. [Crossref] [PubMed]
- Vignon P, Merz TM, Vieillard-Baron A. Ten reasons for performing hemodynamic monitoring using transesophageal echocardiography. Intensive Care Med 2017;43:1048-51. [Crossref] [PubMed]
- Vieillard-Baron A, Charron C, Chergui K, et al. Bedside echocardiographic evaluation of hemodynamics in sepsis: is a qualitative evaluation sufficient? Intensive Care Med 2006;32:1547-52. [Crossref] [PubMed]
- Ashbaugh DG, Bigelow DB, Petty TL, et al. Acute respiratory distress in adults. Lancet 1967;2:319-23. [Crossref] [PubMed]
- Bellani G, Laffey JG, Pham T, et al. Epidemiology, Patterns of Care, and Mortality for Patients With Acute Respiratory Distress Syndrome in Intensive Care Units in 50 Countries. JAMA 2016;315:788-800. [Crossref] [PubMed]
- Mekontso Dessap A, Boissier F, Charron C, et al. Acute cor pulmonale during protective ventilation for acute respiratory distress syndrome: prevalence, predictors, and clinical impact. Intensive Care Med 2016;42:862-70. [Crossref] [PubMed]
- Briel M, Meade M, Mercat A, et al. Higher vs lower positive end-expiratory pressure in patients with acute lung injury and acute respiratory distress syndrome: systematic review and meta-analysis. JAMA 2010;303:865-73. [Crossref] [PubMed]
- McAuley DF, Laffey JG, O’Kane CM, et al. HARP-2 Page 748 of 749 Investigators, Irish Critical Care Trials Group. Simvastatin in the acute respiratory distress syndrome. N Engl J Med 2014;371:1695-703. [Crossref] [PubMed]
- Vieillard-Baron A, Girou E, Valente E, et al. Predictors of mortality in acute respiratory distress syndrome. Focus on the role of right heart catheterization. Am J Respir Crit Care Med 2000;161:1597-601. [PubMed]
- ARDS Definition Task Force, Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA 2012;307:2526-33. [PubMed]
- Hernu R, Wallet F, Thiollière F, et al. An attempt to validate the modification of the American-European consensus definition of acute lung injury/acute respiratory distress syndrome by the Berlin definition in a university hospital. Intensive Care Med 2013;39:2161-70. [Crossref] [PubMed]
- Zapol WM, Snider MT. Pulmonary hypertension in severe acute respiratory failure. N Engl J Med 1977;296:476-80. [Crossref] [PubMed]
- Vieillard-Baron A, Dreyfuss D. Ventilator-induced Lung Injury: Follow the Right Direction! Another Piece of the Puzzle in the Ventilator-induced Lung Injury Epic. Am J Respir Crit Care Med 2017;196:1366-8. [Crossref] [PubMed]
- Brun-Buisson C, Minelli C, Bertolini G, et al. ALIVE Study Group. Epidemiology and outcome of acute lung injury in European intensive care units. Results from the ALIVE study. Intensive Care Med 2004;30:51-61. [Crossref] [PubMed]
- Vieillard-Baron A, Prin S, Chergui K, et al. Hemodynamic instability in sepsis: bedside assessment by Doppler echocardiography. Am J Respir Crit Care Med 2003;168:1270-6. [Crossref] [PubMed]
- Geri G, Vignon P, Aubry A, et al. Cardiovascular clusters in septic shock combining clinical and echocardiographic parameters: a post hoc analysis. Intensive Care Med 2019;45:657-67. [Crossref] [PubMed]
- Vignon P, Repessé X, Bégot E, et al. Comparison of Echocardiographic Indices Used to Predict Fluid Responsiveness in Ventilated Patients. Am J Respir Crit Care Med 2017;195:1022-32. [Crossref] [PubMed]
- Vieillard-Baron A, Naeije R, Haddad F, et al. Diagnostic workup, etiologies and management of acute right ventricle failure: A state-of-the-art paper. Intensive Care Med 2018;44:774-90. [Crossref] [PubMed]
- Harjola VP, Mebazaa A, Čelutkienė J, et al. Contemporary management of acute right ventricular failure: a statement from the Heart Failure Association and the Working Group on Pulmonary Circulation and Right Ventricular Function of the European Society of Cardiology. Eur J Heart Fail 2016;18:226-41. [Crossref] [PubMed]
- Lhéritier G, Legras A, Caille A, et al. Prevalence and prognostic value of acute cor pulmonale and patent foramen ovale in ventilated patients with early acute respiratory distress syndrome: a multicenter study. Intensive Care Med 2013;39:1734-42. [Crossref] [PubMed]
- Boissier F, Katsahian S, Razazi K, et al. Prevalence and prognosis of cor pulmonale during protective ventilation for acute respiratory distress syndrome. Intensive Care Med 2013;39:1725-33. [Crossref] [PubMed]
- Vieillard-Baron A, Prin S, Chergui K, et al. Echo-Doppler demonstration of acute cor pulmonale at the bedside in the medical intensive care unit. Am J Respir Crit Care Med 2002;166:1310-9. [Crossref] [PubMed]
- Mekontso Dessap A, Proost O, Boissier F, et al. Transesophageal echocardiography in prone position during severe acute respiratory distress syndrome. Intensive Care Med 2011;37:430-4. [Crossref] [PubMed]
- Cook CH, Praba AC, Beery PR, et al. Transthoracic echocardiography is not cost-effective in critically ill surgical patients. J Trauma 2002;52:280-4. [Crossref] [PubMed]
- Legras A, Caille A, Begot E, et al. ARCO and CRICS network. Acute respiratory distress syndrome (ARDS)-associated acute cor pulmonale and patent foramen ovale: a multicenter noninvasive hemodynamic study. Crit Care. 2015;19:174. [Crossref] [PubMed]
- Vieillard-Baron A, Matthay M, Teboul JL, et al. Experts' opinion on management of hemodynamics in ARDS patients: focus on the effects of mechanical ventilation. Intensive Care Med 2016;42:739-49. [Crossref] [PubMed]
- Sakr Y, Vincent JL, Reinhart K, et al. High tidal volume and positive fluid balance are associated with worse outcome in acute lung injury. Chest 2005;128:3098-108. [Crossref] [PubMed]
- Jozwiak M, Silva S, Persichini R, et al. Extravascular lung water is an independent prognostic factor in patients with acute respiratory distress syndrome. Crit Care Med 2013;41:472-80. [Crossref] [PubMed]
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network, Wiedemann HP, Wheeler AP, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006;354:2564-75.
- Grissom CK, Hirshberg EL, Dickerson JB, et al. Fluid management with a simplified conservative protocol for the acute respiratory distress syndrome. Crit Care Med 2015;43:288-95. [Crossref] [PubMed]
- Gattinoni L, Chiumello D, Carlesso E, et al. Bench-to-bedside review: chest wall elastance in acute lung injury/acute respiratory distress syndrome patients. Crit Care 2004;8:350-5. [Crossref] [PubMed]
- Mauri T, Yoshida T, Bellani G, et al. Esophageal and transpulmonary pressure in the clinical setting: meaning, usefulness and perspectives. Intensive Care Med 2016;42:1360-73. [Crossref] [PubMed]
- Whittenberger JL, McGregor M, Berglund E, et al. Influence of state of inflation of the lung on pulmonary vascular resistance. J Appl Physiol 1960;15:878-82. [Crossref] [PubMed]
- Jardin F, Brun-Ney D, Cazaux P, et al. Relation between transpulmonary pressure and right ventricular isovolumetric pressure change during respiratory support. Cathet Cardiovasc Diagn 1989;16:215-20. [Crossref] [PubMed]
- Mahjoub Y, Pila C, Friggeri A, et al. Assessing fluid responsiveness in critically ill patients: False-positive pulse pressure variation is detected by Doppler echocardiographic evaluation of the right ventricle. Crit Care Med 2009;37:2570-5. [Crossref] [PubMed]
- Mercat A, Diehl JL, Meyer G, et al. Hemodynamic effects of fluid loading in acute massive pulmonary embolism. Crit Care Med 1999;27:540-4. [Crossref] [PubMed]
- Pinsky MR, Payen D. Functional hemodynamic monitoring. Crit Care 2005;9:566-72. [Crossref] [PubMed]
- Wetterslev M, Moller-Sorensen H, Johansen RR, et al. Systematic review of cardiac output measurements by echocardiography vs. thermodilution: the techniques are not interchangeable. Intensive Care Med 2016;42:1223-33. [Crossref] [PubMed]
- Vieillard-Baron A, Charron C, Caille V, et al. Prone positioning unloads the right ventricle in severe ARDS. Chest 2007;132:1440-6. [Crossref] [PubMed]
- Combes A, Hajage D, Capellier G, et al. Extracorporeal Membrane Oxygenation for Severe Acute Respiratory Distress Syndrome. N Engl J Med 2018;378:1965-75. [Crossref] [PubMed]
- Serpa Neto A, Schmidt M, Azevedo LC, et al. Associations between ventilator settings during extracorporeal membrane oxygenation for refractory hypoxemia and outcome in patients with acute respiratory distress syndrome: a pooled individual patient data analysis: Mechanical ventilation during ECMO. Intensive Care Med 2016;42:1672-84. [Crossref] [PubMed]
- Lazzeri C, Cianchi G, Bonizzoli M, et al. Right ventricle dilation as a prognostic factor in refractory ARDS requiring VV–ECMO. Minerva Anestesiol 2016;82:1043-9. [PubMed]
- Bunge JJH, Caliskan K, Gommers D, et al. Right ventricular dysfunction during acute respiratory distress syndrome and veno-venous extracorporeal membrane oxygenation. J Thorac Dis 2018;10:S674-82. [Crossref] [PubMed]
- Reis Miranda D, van Thiel R, Brodie D, et al. Right ventricular unloading after initiation of venovenous extracorporeal membrane oxygenation. Am J Respir Crit Care Med 2015;191:346-8. [Crossref] [PubMed]
- Kon ZN, Bittle GJ, Pasrija C, et al. Venovenous Versus Venoarterial Extracorporeal Membrane Oxygenation for Adult Patients With Acute Respiratory Distress Syndrome Requiring Precannulation Hemodynamic Support:A Review of the ELSO Registry. Ann Thorac Surg 2017;104:645-9. [Crossref] [PubMed]
- Bréchot N, Luyt CE, Schmidt M, et al. Venoarterial extracorporeal membrane oxygenation support for refractory cardiovascular dysfunction during severe bacterial septic shock. Crit Care Med 2013;41:1616-26. [Crossref] [PubMed]
Cite this article as: Godement M, Vieillard-Baron A. Hemodynamic monitoring of ARDS by critical care echocardiography. J Emerg Crit Care Med 2019;3:36.


