
Echocardiographic approach to shock
Introduction
Circulatory shock is a life-threatening condition in which the circulation is unable to deliver sufficient oxygen to the tissues in order to meet their basal metabolic requirements, resulting in organ dysfunction (1,2). While clinical signs of shock are very important to detect that the patient is in shock, these signs do not indicate what is the nature of shock nor whether cardiac output is increased, normal or decreased (3). Accordingly, the hemodynamic evaluation is urgently required to identify the type of shock and hence the best therapies to apply (1,2,4,5).
Four different pathophysiological mechanisms can result in shock (2,6). Hypovolemia, either absolute (due to fluid losses) or relative (due to redistribution of fluids from central to peripheral compartments, related to an increase in intrathoracic pressures or dilation of large capacitance veins) results in a decrease in cardiac preload and hence in cardiac output. Cardiogenic, or failure of pump function, resulting from an impaired contractility, severe valvular dysfunction or severe arrythmias. Obstructive shock results from compression of heart cavities from cardiac tamponade or increase in right ventricular afterload (often from pulmonary embolism). Finally, distributive shock results from the impairment in vascular tone, associated with severe hypotension, venous dilation (and hence relative hypovolemia), altered distribution of perfusion between the different organs but also inside each organ (microvascular impairment).
Echocardiography allows to obtain the full hemodynamic evaluation (7) and seems to be associated with improved outcomes in observational cohorts (8). For many reasons, echocardiography is the perfect hemodynamic tool for the initial evaluation of the patient in shock. First, it can be rapidly be available at bedside. Several studies have shown that characterization of the pathophysiological mechanism of shock can be obtained within less than 5 min. Second, the echocardiographic evaluation allows to identify several patterns that characterize the different types of shock (Table 1). Third, echocardiography allows to rapidly indicate the potential hemodynamic strategies (fluid responsiveness/impairment of left or right ventricular function/increased right ventricular afterload……) that can be applied. Fourth, it allows to rapidly evaluate the response to the applied therapies. Last, but not least, the information obtained by echocardiography is achieved within a timeframe which not long enough to insert and derive information from many invasive hemodynamic devices.

Full table
Typical echocardiographic characteristics of hypovolemic shock
Hypovolemic shock is characterized by a decrease in stressed volume. Accordingly, the cardiac volumes and pressures are usually low. This is reflected by small size of cardiac cavities, sometimes associated with virtual left end systolic volume or “kissing ventricles”. The inferior and superior vena cava sizes are usually small and disclose significant respiratory variations. Left heart filling pressures are low: low mitral E wave, low E/E’ ratio. Aortic velocity time integral (VTI) and cardiac output are typically low.
Typical echocardiographic characteristics of obstructive shock due to cardiac tamponade
Cardiac tamponade is induced by the increase in pericardial pressure. Importantly, the relation between pericardial pressure and volume is curvilinear. Also, chronic accumulation of pericardial fluid initially minimally increases pericardial pressure due to the progressive increase in pericardial compliance. Accordingly, diagnosis of cardiac tamponade relies on the detection of hemodynamic signs of cardiac tamponade. These include systolic collapse of right atrium, diastolic collapse of right ventricle (RV) together with dilated superior and inferior vena cava. Doppler patterns are also indicative, with respiratory fluctuations in artic VTI, mitral and tricuspid flows. Finally, Aortic VTI and cardiac output are typically low.
Typical echocardiographic characteristics of obstructive shock due to elevated right ventricular afterload
Increases in RV afterload may lead obstructive shock. The RV is sensitive to pressure and volume overload. Importantly, the RV prolongs its systolic time in face of an increased RV afterload so that the pressure in the RV exceeds the pressure of the left ventricle (LV) at the end of systole leading to paradoxical septal movement. Dilation of RV is the cornerstone and its association with paradoxical septal movement defines acute cor pulmonale. Acute cor pulmonale is usually evaluated by assessing the ratio between RV and LV end diastolic volumes in long axis, coupled with detection of paradoxical septal movement in short axis (9,10).
RV systolic function can be evaluated by tricuspid displacement assessed by tricuspid annular plane excursion (TAPSE) by M-Mode or tricuspid S wave by tissue Doppler. RV strain measurement is also promising. Of note, assessment of RV systolic function is relevant to evaluate the evolution of the patient and response to therapy but not for the diagnosis of acute cor pulmonale and obstructive shock
Measurements of pulmonary artery pressure either directly by tricuspid regurgitant jet by continuous wave Doppler or indirectly by analysis of pulmonary artery flow (by pulsed wave Doppler) may be useful for confirming the increased RV afterload but mostly for assessing the effects of therapeutic interventions. Analysis of pulmonary artery flow consists in the measurement of pulmonary acceleration time or morphologic analysis (presence of notch or biphasic flow). Respiratory fluctuations in pulmonary artery flow are often present.
Detection of clots in right heart cavities or pulmonary artery is of course confirming the diagnosis of pulmonary embolism as the cause of obstructive shock.
As obstructive shock is a low flow shock, aortic VTI and cardiac output are typically low.
Typical echocardiographic characteristics of cardiogenic shock
Cardiogenic shock is characterized by pump failure related to severe alteration in contractility (in most cases), severe valvular dysfunction or shunts. It can also result from malignant arrhythmias. It can be due to LV, RV or global failure. Due to the multiplicity of causes and pathophysiological mechanisms leading to cardiogenic shock, recognition of cardiogenic shock is based on the combination of low cardiac output and elevated filling pressures. The typical pattern includes a low stroke volume associated with elevated left atrial pressure (elevated mitral E wave and increased E/E’ ratio). Dilation of LV is observed in patients with chronic or subacute impairment in LV function (chronic heart failure, myocarditis) but may be absent when an abrupt decrease in LV function is leading to cardiogenic shock (such as acute myocardial infarction or malignant arrythmias).
Cardiogenic shock associated with isolated right heart failure is characterized by a low cardiac output associated with elevated right atrial pressure (dilated right atrium and dilated inferior and superior vena cava) without significant pulmonary hypertension.
The sonographer should evaluate valvular function in order to detect severe valvular dysfunction. In most cases, this should trigger the advice of a cardiologist consultant.
When a typical pattern of cardiogenic shock is observed but its cause is not obvious, it is also safe to request cardiologic consultation.
Typical echocardiographic characteristics of distributive shock
The key characteristic of distributive shock is the high cardiac output. Due to the impaired endothelial function, distributive shock is associated with profound vasoplegia. Myocardial depression may be present, especially in septic shock. The decrease in LV afterload may mask LV dysfunction which may become obvious only after correction of hypotension (11,12). In addition, hypovolemia is often associated, as a consequence of the decreased in stressed volume related to venous dilation. Five different patterns have recently been recognized (13): well resuscitated, persistent fluid responsiveness, predominant LV dysfunction, severe RV failure and hyperkinetic.
Due to these pleiotropic presentations, the diagnosis of distributive shock is often made based on the presence of a high cardiac output (normal or elevated aortic VTI).
The echocardiographic evaluation should nevertheless also focus on evaluating factors that may trigger therapeutic interventions such as preload responsiveness and cardiac function. Preload responsiveness can be evaluated using many echocardiographic indices. Evaluation of cardiac function is more complex due to the diversity of indices. In most cases ejection fraction is a good marker of the impairment of cardiac function, even though more subtle alterations of myocardial contractility can be detected using more advanced technologies (12,14,15). Many patients with myocardial depression in sepsis do not require the use of inotropic agents, as cardiac output is often adequate even in the presence of impaired cardiac function, due to the associated decrease in LV afterload and slight LV dilation. Therapies should then be indicated based on the indication of the inadequacy of cardiac output as reflected by signs of tissue hypoperfusion: skin mottling, increased capillary refill time, oliguria, low venous oxygen saturation, increased veno-arerial PCO2 gradients, and elevated lactate levels (16).
It is also relevant to evaluate right ventricular function in sepsis. Right ventricular function is also frequently impaired in sepsis due to the combined effects of sepsis associated impairment in RV contractility and elevation in RV afterload (ARDS and mechanical ventilation), In one fifth of the patients, RV dysfunction is the predominant feature (13).
Finally, it is also important to carefully check for the presence of dynamic left ventricular outflow tract or midventricular obstructions (17).
Diagnostic algorithm
Importantly, the diagnostic workup should be based on the pathophysiologic mechanisms of shock (Figure 1). As some of these mechanisms may co-exist, the hemodynamic evaluation should always try to identify the most severe one(s).
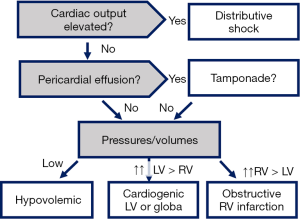
We suggest to begin by determination of cardiac output: an elevated cardiac output indicated distributive shock and excludes all other causers of shock as the dominant one. If cardiac output is low, tamponade should be ruled out easily. Then pressure and volumes should be evaluated: low pressures/volumes indicate hypovolemic shock, elevated left side pressures/volumes indicate cardiogenic shock due to LV failure, elevated right-side pressures/volumes indicate RV failure.
Therapeutic approach
Therapeutic interventions should aim at correcting the cause of shock as soon as possible. Meanwhile, supportive therapy should be also initiated. Echocardiography allows to orient therapeutic interventions but also to guide supportive therapies. Supportive therapies should be based non-only on the prominent hemodynamic alteration but also using the therapies associated with the higher benefit/risk ratio (18). The advantage of echocardiography is that it may help not only to indicate the administration of fluids and vasoactive agents, but also to fine tune ventilatory settings as it provides information on the RV (19). Also, for guiding fluid therapy, several indices can be used (20) so that is particularly helpful when some indices are inconclusive as hampered by technical limitations or providing results in the grey zone (21).
Interest for repeated assessment
As a non-invasive technique, echocardiography can easily be repeated not only for evaluation of changing hemodynamic patterns but also for finetuning hemodynamic support. Implementation of serial echocardiography to guide hemodynamic management of patients with shock was associated with less fluid but more dobutamine administration (22). In this before versus after study, this approach resulted in less acute kidney injury and improved 28 days survival.
Five key points
- Echocardiography can be used to rapidly identify the type of shock;
- Echocardiography can be used to guide administration of fluids and vasoactive agents;
- Echocardiography can be used to fine tune ventilatory settings in mechanically ventilated patients;
- In patients with heart failure, echocardiography allows to identify the precise mechanism responsible for cardiac dysfunction;
- Serial echocardiographic evaluations provide additional information so that echocardiography should not be limited to the initial assessment.
Conclusions
Echocardiography has now become the preferred approach for initial management of patients with shock. It provides very rapidly an integrative view of the hemodynamic alterations, allowing to identify the type of shock and to select the optimal therapy. Importantly, due to its non-invasive nature, echocardiography can be easily repeated providing important information on the response to therapy and allowing serial evaluation.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Cecconi M, De Backer D, Antonelli M, et al. Consensus on Circulatory Shock and Hemodynamic Monitoring. Task Force of the European Society of Intensive Care Medicine. Intensive Care Med 2014;40:1795-815. [Crossref] [PubMed]
- Vincent JL, De Backer D. Circulatory shock. N Engl J Med 2013;369:1726-34. [Crossref] [PubMed]
- Hiemstra B, Koster G, Wiersema R, et al. The diagnostic accuracy of clinical examination for estimating cardiac index in critically ill patients: the Simple Intensive Care Studies-I. Intensive Care Med 2019;45:190-200. [Crossref] [PubMed]
- De Backer D, Cecconi M, Lipman J, et al. Challenges in the management of septic shock: a narrative review. Intensive Care Med 2019;45:420-33. [Crossref] [PubMed]
- De Backer D, Vieillard-Baron A. Clinical examination: a trigger but not a substitute for hemodynamic evaluation. Intensive Care Med 2019;45:269-71. [Crossref] [PubMed]
- Weil MH, Shubin H. Proposed reclassification of shock states with special reference to distributive defects. Adv Exp Med Biol 1971;23:13-23. [Crossref] [PubMed]
- Hemodynamic monitoring using echocardiography in the critically ill. New York: Springer, 2011.
- Feng M, McSparron JI, Kien DT, et al. Transthoracic echocardiography and mortality in sepsis: analysis of the MIMIC-III database. Intensive Care Med 2018;44:884-92. [Crossref] [PubMed]
- Vieillard-Baron A, Schmitt JM, Augarde R, et al. Acute cor pulmonale in acute respiratory distress syndrome submitted to protective ventilation: incidence, clinical implications, and prognosis. Crit Care Med 2001;29:1551-5. [Crossref] [PubMed]
- Vieillard-Baron A, Millington SJ, Sanfilippo F, et al. A decade of progress in critical care echocardiography: a narrative review. Intensive Care Med 2019;45:770-88. [Crossref] [PubMed]
- Vieillard-Baron A, Caille V, Charron C, et al. Actual incidence of global left ventricular hypokinesia in adult septic shock. Crit Care Med 2008;36:1701-6. [Crossref] [PubMed]
- Boissier F, Razazi K, Seemann A, et al. Left ventricular systolic dysfunction during septic shock: the role of loading conditions. Intensive Care Med 2017;43:633-42. [Crossref] [PubMed]
- Geri G, Vignon P, Aubry A, et al. Cardiovascular clusters in septic shock combining clinical and echocardiographic parameters: a post hoc analysis. Intensive Care Med 2019;45:657-67. [Crossref] [PubMed]
- Orde SR, Pulido JN, Masaki M, et al. Outcome prediction in sepsis: speckle tracking echocardiography based assessment of myocardial function. Crit Care 2014;18:R149. [Crossref] [PubMed]
- Bergenzaun L, Ohlin H, Gudmundsson P, et al. Mitral annular plane systolic excursion (MAPSE) in shock: a valuable echocardiographic parameter in intensive care patients. Cardiovasc Ultrasound 2013;11:16. [Crossref] [PubMed]
- De Backer D. Detailing the cardiovascular profile in shock patients. Crit Care 2017;21:311. [Crossref] [PubMed]
- Chauvet JL, El-Dash S, Delastre O, et al. Early dynamic left intraventricular obstruction is associated with hypovolemia and high mortality in septic shock patients. Crit Care 2015;19:262. [Crossref] [PubMed]
- De Backer D, Foulon P. Minimizing catecholamines and optimizing perfusion. Crit Care 2019;23:149. [Crossref] [PubMed]
- De Backer D, Bakker J, Cecconi M, et al. Alternatives to the Swan-Ganz catheter. Intensive Care Med 2018;44:730-41. [Crossref] [PubMed]
- Vignon P, Repesse X, Begot E, et al. Comparison of Echocardiographic Indices Used to Predict Fluid Responsiveness in Ventilated Patients. Am J Respir Crit Care Med 2017;195:1022-32. [Crossref] [PubMed]
- Biais M, Ehrmann S, Mari A, et al. Clinical relevance of pulse pressure variations for predicting fluid responsiveness in mechanically ventilated intensive care unit patients: the grey zone approach. Crit Care 2014;18:587. [Crossref] [PubMed]
- Kanji HD, McCallum J, Sirounis D, et al. Limited echocardiography-guided therapy in subacute shock is associated with change in management and improved outcomes. J Crit Care 2014;29:700-5. [Crossref] [PubMed]
Cite this article as: De Backer D, Giglioli S. Echocardiographic approach to shock. J Emerg Crit Care Med 2019;3:35.


