Buprenorphine analgesia following major abdominal surgery: a systematic review and meta-analysis
Introduction
Optimizing perioperative analgesia is imperative to a smooth recovery following major abdominal surgery (1). Multiple guidelines, including enhanced recovery after surgery (ERAS) recommendations, encourage multimodal analgesia and avoidance of opioids (2). However, postoperative pain management can be challenging and as such opioids are frequently needed (3-5). Opioid medications come with a range of adverse effects, and resultantly the choice of opioid to administer should be made responsibly.
A recent meta-analysis has found buprenorphine to be an effective analgesic for managing acute pain (6). Being a partial mu-opioid receptor agonist and kappa-opioid receptor antagonist, it is largely accepted that buprenorphine has favorable adverse effect profile relative to morphine (7,8). Furthermore, buprenorphine has the significant benefit of a low, if not absent, risk of abuse and is employed to treat opiate addiction (9,10). Buprenorphine can be administered via multiple means including the convenient sublingual route. It therefore presents the opportunity to avoid intravenous opiate analgesia in the nil-per-oral patient and possible facilitation of earlier cessation of patient-controlled opioid analgesia (PCA). The preclusion of the need for intravenous lines and PCA equipment may allow earlier mobilization and a reduced infection risk (11). Due to its high affinity for mu-opioid receptors, buprenorphine has a longer half-life than morphine and requires less frequent dosing (12-14).
Given the many potential advantages of buprenorphine, we sought to investigate its effectiveness in managing pain following abdominal surgery compared to morphine. We also examined buprenorphine’s opioid-related side effects relative to morphine. This was achieved by means of a systematic review and meta-analysis of randomized controlled trials.
Methods
Search strategy
Five databases (Cochrane trials registry, SCOPUS, Medline, CINAHL and Web of Science) were systematically searched from the inception of the databases until May 2019. This search was conducted by two independent reviewers (L White & A Hodge) searching the terms (I) “buprenorphine” AND “acute pain”; (II) “buprenorphine” AND “post-operative pain”; (III) “buprenorphine” AND “surgical pain”. A manual reference check and citation check of included papers was performed to identify any additional studies.
Study eligibility
For a study to be included, the authors were required to report on the use of buprenorphine versus morphine in the management of acute pain after “major abdominal surgery” in the inpatient hospital setting. Given the lack of consensus in the literature, we defined major abdominal surgery as surgery involving the abdominal cavity, abdominal wall or abdominal organs PLUS an expected duration greater than 60 minutes or involving large abdominal wall incisions for example a laparotomy.
Only randomized controlled trials (RCTs) were eligible for inclusion and there were no language criteria for exclusion. Two reviewers (L White & A Hodge) independently assessed each study for inclusion in this systematic review.
Morphine is a well-studied and understood opiate and was chosen as a “treatment-as-usual” control group. Studies investigating the use of buprenorphine for chronic pain or opioid addiction were excluded.
Data extraction
Two reviewers (LW and AH) independently extracted data from each article that met the inclusion criteria. The data extracted from each study included the study design, patient characteristics and clinical outcome results. The data collected by each reviewer was then compared for homogeneity.
Clinical outcome measures
Our a priori primary outcomes of interest were analgesic effect [as measured by visual analogue scale (VAS)] and rescue analgesia requirement. The secondary outcomes were incidence of respiratory depression, incidence of sedation, nausea, vomiting, dizziness and hypotension.
Level of evidence, risk of bias & outcome level of evidence ranking
Each article was evaluated using the Centre for Evidence Based Medicine (CEBM): Levels of Evidence Introduction Document (15). These studies were then assessed for risk of bias and methodological quality using the Cochrane Collaboration’s tool for assessing the risk of bias (16). The results from each study were then grouped by outcome. Where quantitative analysis was unfeasible, qualitative analysis was performed.
Statistical analyses
The combined data was analyzed using RevMan 5.3 software (The Nordic Cochrane Centre, Copenhagen, Denmark). Odds ratios (OR) with 95% confidence intervals (CIs) were calculated for dichotomous outcomes using the Mantel-Haenszel model with random effects. Weighted mean difference (WMD) with 95% CI was calculated for continuous outcomes. Heterogeneity was assess using the I2 statistic, with an I2>50% indicating significant heterogeneity. P value of <0.05 provided evidence of significant OR and WMD. A P value of <0.10 was used to demonstrate heterogeneity of intervention effects. A sensitivity analysis was performed on each outcome by separating the adult and pediatric studies.
Reporting
This study was reported in line with PRISMA guidelines (17).
Results
Literature search results
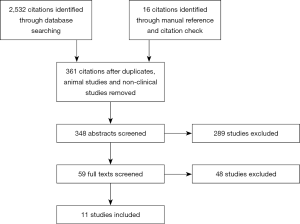
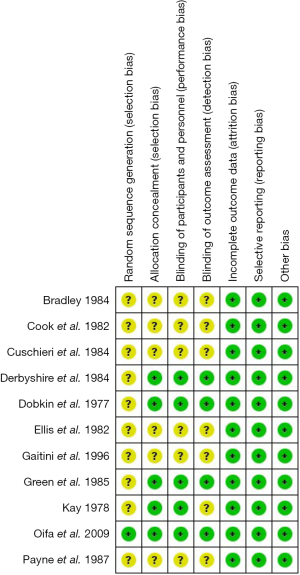
The initial systematic literature search yielded 2,532 citations and a further 16 citations were identified through a manual citation and reference search of relevant articles (Figure 1). Following the removal of duplicates, animal studies and non-clinical studies, 361 citations remained. Of these, 348 abstracts were screened and 59 full texts were retrieved for review. Eleven articles met the inclusion criteria (Figure 1). These 11 studies included 764 patients (12-14,18-25). All studies investigated the management of post major abdominal surgery pain, comparing buprenorphine to morphine (Table 1). Five studies met the criteria for high quality RCTs, leaving six low quality RCTs (Figure 2).
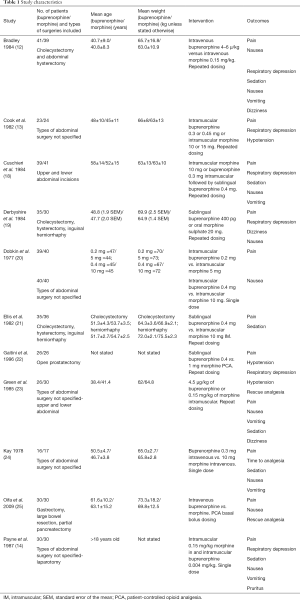
Full table
Primary outcomes
Post-operative pain was measured using a variety of endpoints including various pain scores and drop-out rate as a consequence of pain. Pain scores from 1 to 6 hours were measured by six studies. The outcomes varied between these papers, with 3 demonstrating no difference and 3 showing lower pain scores with buprenorphine (12,13,20,22,24,25). A quantitative analysis was unable to be performed on these studies. Three studies investigated pain at 6 to 12 hours and revealed a significant improvement in analgesia with buprenorphine (VAS WMD =−0.75; 95% CI: −1.16 to −0.34; I2=49%; P=0.0003). Four studies assessed pain at 24 hours, at which time there was no difference in experienced pain (VAS WMD =−0.41; 95% CI: −1.04 to 0.22; I2=97%; P=0.21) (13,18,19,21). The same four studies showed no difference at 48 hours (VAS WMD =0.03; 95% CI: −0.84 to 0.90; I2=97%; P=0.94) (13,18,19,21).
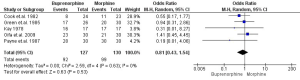
Nine studies investigated the use of rescue analgesia (13,14,18,19,21-25). Five used incidence of rescue analgesia use and four used the cumulative totals of various analgesics. Qualitatively, six studies found no difference between buprenorphine and morphine and three studies showed significantly less use of rescue analgesia in the buprenorphine group. Quantitatively, there was no difference in incidence of breakthrough analgesia requirement between buprenorphine and morphine (OR 0.81; 95% CI: 0.43 to 1.54; I2=0%; P=0.53; n=191; Figure 3).
Secondary outcomes
Eight studies (12-14,18,20-22,25) investigated respiratory depression via a variety of endpoints including incidence of respiratory depression [respiratory rate (RR) <10 breaths/min], PaCO2 and mean RR. Six studies showed no difference and two showed an increased incidence of respiratory depression. Three studies with a total 220 patients had sufficient data for analysis (12,14,18). There was no difference in respiratory depression between groups (OR 4.07; 95% CI: 0.44 to 37.62; I2=0%; P=0.22).
Seven studies with a pool of investigated the incidence of sedation (12,18-21,23,24). There was no significant difference of sedation between buprenorphine and morphine (OR 1.77; 95% CI: 0.38 to 8.34; I2=59%; P=0.47; n=250).
Eight studies with a combined investigated the incidence of post-operative nausea (12-14,18,21-25), in which there was no difference between buprenorphine and morphine (OR 1.05; 95% CI: 0.62 to 1.76; I2=0%; P=0.86; n=442). Similarly, no difference was found in postoperative vomiting in an analysis of six studies (OR 0.99; 95% CI: 0.47 to 2.09; I2=18%; P=0.98; n=330) (12,14,18,22-24). Three studies examined and found no difference in post-operative dizziness (12,19,23). Six out of 7 studies showed no difference in the incidence of postoperative hypotension (12-14,20,22,23,25).
Discussion
This is the first systematic review and meta-analysis comparing buprenorphine with other opioids in the period following abdominal surgery. We found level one evidence of the non-inferiority of buprenorphine relative to morphine in this setting. Conversely, there was no difference in the incidence of opioid-related side effects such as respiratory depression and sedation.
Buprenorphine is a mixed agonist-antagonist opioid (6,8,24). Buprenorphine is approximately 33 times more potent as an analgesic than morphine (2,26). A recent meta-analysis demonstrated the utility of buprenorphine in the management of acute pain (6). Our study suggests that postoperative analgesia with buprenorphine and morphine is equivalent for up to 48 hours and may be enhanced with buprenorphine between six and 12 hours postoperatively.
Buprenorphine has a slower onset of action than morphine, reaching peak serum concentration from 30 to 60 minutes post-administration sublingually (27), which could potentially limit its utility given the severe pain developed following major abdominal surgery. However, our finding of equivalency of both early postoperative pain and breakthrough analgesia requirements appear to refute this. This effect appears to be irrespective of route of administration, with studies investigating sublingual, intramuscular (IM) or intravenous (IV) buprenorphine showing equivalence with IV, IM or oral morphine (12,13,18-20,23).
Of note is our finding that respiratory depression and sedation is equivalent between buprenorphine and morphine in patients having undergone abdominal surgery. As such, our findings are in contradiction to the widely held belief that buprenorphine offers a ceiling effect regarding respiratory depression (8). Being a partial agonist, buprenorphine is relatively resistant to the antagonistic effects of naloxone. Without a reliable antidote, buprenorphine overdose may require ICU admission for respiratory monitoring (28). As such it may pose increased risk to the postoperative patient, particularly in those with reduced respiratory reserve. Further study is required to investigate if this plays out in the postoperative setting.
This study has the potentially major public health implications. Opioid drug abuse has reached global epidemic status. Medical prescription has proven a significant gateway to drug abuse and a critical source of opioids for the black market (29-31). Buprenorphine is a commonly used treatment for opioid addiction (32,33). Being a partial agonist at the mu opioid receptor and an antagonist at the kappa opioid receptor, postoperative patients are much less likely to experience euphoria and withdrawal with its use (6,33,34). Subsequently, risk of addiction is reduced with buprenorphine and patient drug seeking is significantly limited (9,10). It follows that buprenorphine’s use in the postoperative setting could play a vital preventative role in efforts to contain opioid abuse.
It should also be remembered that with buprenorphine’s antagonism of kappa receptors, patient dysphoria—a troublesome side effect of opioids—is unlikely with its use (26).
This study was limited by the lack of evidence for the potential secondary benefits of buprenorphine in the immediate postoperative period. Firstly, abdominal surgery frequently induces a delay in gastric emptying and the urge remain recumbent. This impairs opioid absorption and may render pharmacokinetics unpredictable (35,36). Furthermore, postoperative patients are not uncommonly on complete bowel rest for a short period as a result of intolerance to per oral fluids and solids. This often necessitates pro re nata IM, IV or subcutaneous opioid administration, regularly in the form of PCA. Such equipment is bulky and inconvenient for patient mobilization. More flexible routes of administration and an extended half-life mean buprenorphine has the potential to limit the duration of or completely circumvent the need for PCA. The hypothetical improvements to patient mobility could shorten ileus duration as well as prevent many pulmonary, frailty and pressure area complications (37-40). Given sublingually, buprenorphine may also expedite the removal of intravenous lines and their infection risks (11). Such potential benefits need further study if the true capabilities of the drug are to be elicited.
Our paper is hindered by a limited patient cohort, most of which was studied over 30 years ago. Necessarily, there is a lack of evidence in laparoscopic or robotic surgery. The term abdominal surgery encompasses a large array of operations and our study examines a varied population as a consequence. The intervention and control groups of the included studies differed in their route of administration and this must be considered when examining our outcomes. Each of the papers had incomplete outcome data. Multiple studies did not specify whether patients or assessors were blinded to the allocated intervention.
Conclusions
Buprenorphine is equivalent to morphine in managing pain after abdominal surgery. Buprenorphine has many possible but as to yet unproven benefits in this setting, including eliminating the risk of opioid addiction and improving mobility. Respiratory depression appears to be unaltered when using buprenorphine or morphine. Without an effective antidote, buprenorphine may pose increased risk to patients with limited respiratory reserve.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Perkins FM, Kehlet H. Chronic pain as an outcome of surgery. A review of predictive factors. Anesthesiology 2000;93:1123-33. [Crossref] [PubMed]
- Schug SA, Palmer GM, Scott DA, et al. Acute pain management: scientific evidence, fourth edition, 2015. Med J Aust 2016;204:315-7.
- Christensen KP, Møller AM, Nielsen JK, et al. The Effects of Anesthetic Technique on Postoperative Opioid Consumption in Ankle Fracture Surgery. Clin J Pain 2016;32:870-4. [Crossref] [PubMed]
- Arumugam S, Lau CS, Chamberlain RS. Use of preoperative gabapentin significantly reduces postoperative opioid consumption: a meta-analysis. J Pain Res 2016;9:631-40. [Crossref] [PubMed]
- Doleman B, Read D, Lund JN, et al. Preventive Acetaminophen Reduces Postoperative Opioid Consumption, Vomiting, and Pain Scores After Surgery: Systematic Review and Meta-Analysis. Reg Anesth Pain Med 2015;40:706-12. [Crossref] [PubMed]
- White L, Hodge A, Vlok R, et al. Efficacy and adverse effects of buprenorphine in acute pain management: systematic review and meta-analysis of randomised controlled trials. Br J Anaesth 2018;120:668-78. [Crossref] [PubMed]
- Dahan A, Yassen A, Bijl H, et al. Comparison of the respiratory effects of intravenous buprenorphine and fentanyl in humans and rats. Br J Anaesth 2005;94:825-34. [Crossref] [PubMed]
- Dahan A, Yassen A, Romberg R, et al. Buprenorphine induces ceiling in respiratory depression but not in analgesia. Br J Anaesth 2006;96:627-32. [Crossref] [PubMed]
- Jasinski DR, Pevnick JS, Griffith JD. Human pharmacology and abuse potential of the analgesic buprenorphine: a potential agent for treating narcotic addiction. Arch Gen Psychiatry 1978;35:501-16. [Crossref] [PubMed]
- O'Connor JJ, Moloney E, Travers R, et al. Buprenorphine abuse among opiate addicts. Br J Addict 1988;83:1085-7. [Crossref] [PubMed]
- Webster J, Osborne S, Rickard CM, et al. Clinically-indicated replacement versus routine replacement of peripheral venous catheters. Cochrane Database Syst Rev 2019;1:CD007798. [PubMed]
- Bradley JP. A comparison of morphine and buprenorphine for analgesia after abdominal surgery. Anaesth Intensive Care 1984;12:303-10. [Crossref] [PubMed]
- Cook PJ, James IM, Hobbs KE, et al. Controlled comparison of I.M. morphine and buprenorphine for analgesia after abdominal surgery. Br J Anaesth 1982;54:285-90. [Crossref] [PubMed]
- Payne KA, Murray WB, Barrett H. Intramuscular buprenorphine compared with morphine for postoperative analgesia. S Afr Med J 1987;71:359-61. [PubMed]
- Howick J, Chalmers I, Glasziou P, et al. The 2011 Oxford CEBM evidence levels of evidence (introductory document). Oxford Center for Evidence Based Medicine 2011. Available online: https://www.cebm.net/2011/06/2011-oxford-cebm-levels-evidence-introductory-document/
- Higgins JP, Altman DG, Sterne JA. Chapter 8: assessing risk of bias in included studies. Cochrane Database, 2015.
- Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 2009;6:e1000100. [Crossref] [PubMed]
- Cuschieri RJ, Morran CG, McArdle CS. Comparison of morphine and sublingual buprenorphine following abdominal surgery. Br J Anaesth 1984;56:855-9. [Crossref] [PubMed]
- Derbyshire DR, Vater M, Maile CI, et al. Non-parenteral postoperative analgesia. A comparison of sublingual buprenorphine and morphine sulphate (slow release) tablets. Anaesthesia 1984;39:324-8. [Crossref] [PubMed]
- Dobkin AB, Esposito B, Philbin C. Double-blind evaluation of buprenorphine hydrochloride for post-operative pain. Can Anaesth Soc J 1977;24:195-202. [Crossref] [PubMed]
- Ellis R, Haines D, Shah R, et al. Pain relief after abdominal surgery--a comparison of i.m. morphine, sublingual buprenorphine and self-administered i.v. pethidine. Br J Anaesth 1982;54:421-8. [Crossref] [PubMed]
- Gaitini L, Moskovitz B, Katz E, et al. Sublingual buprenorphine compared to morphine delivered by a patient-controlled analgesia system as postoperative analgesia after prostatectomy. Urol Int 1996;57:227-9. [Crossref] [PubMed]
- Green DW, Sinclair JR, Mikhael MS. Buprenorphine versus morphine. A comparison of intra-operative and postoperative analgesia. Anaesthesia 1985;40:371-5. [Crossref] [PubMed]
- Kay B. A double-blind comparison of morphine and buprenorphine in the prevention of pain after operation. Br J Anaesth 1978;50:605-9. [Crossref] [PubMed]
- Oifa S, Sydoruk T, White I, et al. Effects of intravenous patient-controlled analgesia with buprenorphine and morphine alone and in combination during the first 12 postoperative hours: a randomized, double-blind, four-arm trial in adults undergoing abdominal surgery. Clin Ther 2009;31:527-41. [Crossref] [PubMed]
- Flood P, Rathmell JP, Shafer S. Stoelting's pharmacology and physiology in anesthetic practice. Lippincott Williams & Wilkins, 2015.
- Kuhlman JJ Jr, Lalani S, Magluilo J Jr, et al. Human pharmacokinetics of intravenous, sublingual, and buccal buprenorphine. J Anal Toxicol 1996;20:369-78. [Crossref] [PubMed]
- Richards S, Torre L, Lawther B. Buprenorphine-related complications in elderly hospitalised patients: a case series. Anaesth Intensive Care 2017;45:256-61. [Crossref] [PubMed]
- Manchikanti L, Helm S 2nd, Fellows B, et al. Opioid epidemic in the United States. Pain Physician 2012;15:ES9-38. [PubMed]
- Nelson LS, Juurlink DN, Perrone J. Addressing the Opioid Epidemic. JAMA 2015;314:1453-4. [Crossref] [PubMed]
- Morris BJ, Mir HR. The opioid epidemic: impact on orthopaedic surgery. J Am Acad Orthop Surg 2015;23:267-71. [Crossref] [PubMed]
- Molfenter T, Sherbeck C, Zehner M, et al. Implementing buprenorphine in addiction treatment: payer and provider perspectives in Ohio. Subst Abuse Treat Prev Policy 2015;10:13. [Crossref] [PubMed]
- Hvittfeldt E, Charlotte G, Fridolf I, et al. Triple dosing with high doses of buprenorphine: Withdrawal and plasma concentrations. J Opioid Manag 2015;11:319-24. [Crossref] [PubMed]
- Kress HG. Clinical update on the pharmacology, efficacy and safety of transdermal buprenorphine. Eur J Pain 2009;13:219-30. [Crossref] [PubMed]
- Kennedy JM, van Rij AM. Drug absorption from the small intestine in immediate postoperative patients. Br J Anaesth 2006;97:171-80. [Crossref] [PubMed]
- Sjövall S, Kokki M, Kokki H. Laparoscopic surgery: a narrative review of pharmacotherapy in pain management. Drugs 2015;75:1867-89. [Crossref] [PubMed]
- Smetana GW. Postoperative pulmonary complications: an update on risk assessment and reduction. Cleve Clin J Med 2009;76 Suppl 4:S60-5. [Crossref] [PubMed]
- Lawrence VA, Cornell JE, Smetana GW, et al. Strategies to reduce postoperative pulmonary complications after noncardiothoracic surgery: systematic review for the American College of Physicians. Ann Intern Med 2006;144:596-608. [Crossref] [PubMed]
- Siu AL, Penrod JD, Boockvar KS, et al. Early ambulation after hip fracture: effects on function and mortality. Arch Intern Med 2006;166:766-71. [Crossref] [PubMed]
- Havey R, Herriman E. OʼBrien D. Guarding the gut: early mobility after abdominal surgery. Crit Care Nurs Q 2013;36:63-72. [Crossref] [PubMed]
Cite this article as: Vlok R, White L, Binks M, Hodge A, Ryan T, Baran R, Melhuish T. Buprenorphine analgesia following major abdominal surgery: a systematic review and meta-analysis. J Emerg Crit Care Med 2019;3:34.