
Role of echocardiography in the management of veno-arterial extra-corporeal membrane oxygenation patients
Introduction
In patients with cardiogenic shock (CS), veno-arterial extracorporeal membrane oxygenation (VA ECMO) may be used when the medical therapies fail (1-4). The main indication for VA ECMO remains a medical CS complicating acute coronary syndrome, fulminant myocarditis, acute exacerbation of end-stage heart failure, drug intoxication, hypothermia, refractory arrhythmia and cardiac arrest (1-4). The other main indication of VA ECMO is CS occurring after cardiac or lung surgery including cardiac or pulmonary transplantation (1,4,5). At last, VA ECMO may be discussed in some specific situations for patients with severe pulmonary embolism, stress cardiomyopathy and sepsis cardiomyopathy (1-4).
When a VA ECMO is implanted, physicians should anticipate the mild-term medical project of the patient. VA ECMO may be used as a bridge to myocardial recovery and cardiac transplantation. It may also be used as “a bridge to bridge”, before the implantation of a ventricular assist device. No randomized controlled trials have compared VA ECMO to other mechanical support systems in patients with CS. However, several non-randomized studies report that the early use of VA ECMO is associated with better survival rates in some CS patients (1-4,6).
Nowadays, every intensive care unit (ICU) owns a bedside ultrasound (US) unit available 24/7 impacting greatly the ICU daily practice (5,7-9). Echocardiography is the main key to evaluate a CS both for the diagnosis and the treatment (9). A bedside ultrasound unit should be available at any time for patients with VA ECMO, and must be performed at least once a day, even more if the patient becomes unstable. No other exam can replace US in the daily, bedside monitoring of CS patients both for the key information it brings but also for its availability (5-9).
In this review, the role of echocardiography in the indications, the implantation, and the follow-up of VA ECMO in ICU will be described.
Assessment before ECMO implantation
Optimal timing of VA ECMO implantation is not standardized. The number of etiologies of CS, patients’ comorbidities and unpredictable tolerance of inotrope therapies are many variables making hard to determine the best timing implantation (1-4,6). Correlated to the clinical presentation, echocardiography confirms the diagnosis of CS and determines its severity (Figure 1). In order to evaluate left ventricular (LV) failure, to identify etiology and to visualize concomitant pathologies and complications, an echocardiography must be run before any VA ECMO implantation (9-12).
In front of a cardiovascular collapse urging the urgent institution of ECMO, a complete echocardiographic study is not compulsory: a quick exam may be run completed later by a full cardiac US examination. Indeed, a readily reversible etiology (i.e., tamponade) may be rapidly diagnosed by echocardiography (12).
Echocardiography is also mandatory to eliminate a contra-indication to VA ECMO such as severe aortic insufficiency and aortic dissection (12).
Cannula position
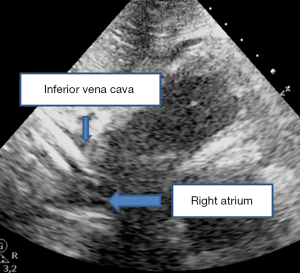
Two approaches exist for the insertion of VA ECMO: an open surgical access via the common femoral or via the axillary artery or a percutaneous approach using the Seldinger technique (1-4,13-19). Percutaneous cannulation for peripheral VA ECMO may lead to fewer local infections and similar rate of vascular complications, compared to the surgical approach (13). In most adult patients, the first line approach is the peripheral cannulation of the femoral vein and the femoral artery (1-4,6,11). The venous line drains deoxygenated blood and returns it oxygenated into the descending aorta through the arterial line to perfuse the lower limbs but also cerebral and upper limb vessels via retrograde flow. In order to localize vessels that should be cannulated and to prevent local complications, a Doppler should be performed before any punction (12,13). However, no recommendation on the use of a fluroscopic or echocardiographic guidance has been made (18,19). Thus, echocardiography remains a bedside invaluable tool both to guide punction and to help cannula positioning due to its availability and its few complications (13,19). The venous cannula should be positioned at the inferior vena cava and the right atrium junction in order to prevent structural damages or pressure against the inferior vena cava wall (Figure 2) (7,12,18,19). Both transthoracic echocardiography (TTE) and transesophageal echocardiography (TTE) can visualize the draining cannula (7,8). However, echocardiography does not appear as an alternative to X-ray but as an adjunct to another imaging methods.
Daily monitoring
Once the VA ECMO implanted, the daily role of echocardiography is to monitor the cardiac function and to early diagnose myocardial recovery or the occurrence of complications (7,12).
A compromise between the degree of ECMO flow according to circulatory requirements and the LV discharge must be found, helped by the daily US monitoring. It is one of the main clinical challenges in the daily management of VA ECMO and can change every day (7,12).
In peripheral VA ECMO, it now commonly admitted that the LV afterload is correlated to the flow injected through the arterial cannula: higher the flow is, higher is the LV afterload (1-4).
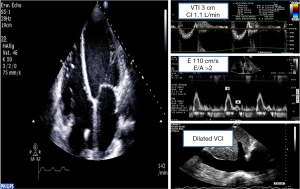
A close monitoring of the cardiac chambers is crucial after an ECMO implantation and must be done daily. The physicians need to verify whether the cardiac chambers are correctly drained and the lack of intracardiac thrombus. In addition, aortic valve opening is paramount (12,19) because as written previously, there is a competition between the retrograde aortic blood flow and the stroke volume ejected by the LV in peripheral VA ECMO. Without a correct aortic valve opening, the LV will in the end suffer from distention and we will assist to a thrombus formation (Figure 3). Repeated cardiac ultrasounds are the key exam to diagnose intra-cavity and intra-aorta thrombi (7,8). Blood stasis, seen as the presence of spontaneous echo contrast in the LV, signs a higher risk of thrombus formation (Figure 3).
Each time a patient becomes unstable (on hemodynamic or on ECMO parameters), a quick bedside echocardiography must be performed in order to diagnose a complication that would require a specific therapy such tamponade, LV thrombus and to assess the right ventricle preload.
In the end, an early detection by repeated echocardiographies of recovery is crucial by assessing both ventricular functions in order to get rid of the ECMO as soon as possible. Adaptation of ventricular function is determined by modulating the ECMO flows (Figure 4) (5).
Weaning
In some indications, a partial or a full recovery of the condition that required the use of ECMO is expected. Then, an ECMO removal can be discussed. A successful weaning from ECMO in patients with refractory CS is defined by an ECMO removal and the absence of further mechanical support because of recurring CS over the following 30 days (14,17).
In patients recovering from severe CS, comprehensive and dynamic echocardiographic measurements are mandatory to predict subsequent ECMO-weaning success (18,20-22). The parameters assessing LV systolic function, such as aortic velocity-time integral, LV ejection fraction (LVEF) and lateral mitral annulus peak systolic velocity allow to identify patients who will be weaned from VA ECMO (14,17). The Doppler parameters reflecting LV filling pressures are not predictive of weaning outcome (14,17). Regarding the right ventricular (RV) function, successfully weaned patients had decreased RV dimensions and increased values of parameters assessing RV systolic function such as ejection fraction, fraction area change and magnitudes of RV strain (19,23). Tricuspid annular plane systolic excursion and severity of tricuspid regurgitation were not useful to differentiate weaned and nonweaned patients (12,19,23).
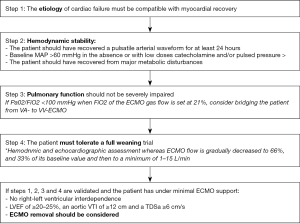
Weaning trials are essential to assess the behavior of ventricles during increases in preload (14,24,25). When ECMO flow decreases, LV preload increases and afterload decreases (24). During the decrease of the ECMO flow, the behaviour of the LV and the Frank-Starling reserve can be evaluated (24,25). Thus, weaning trials are essential to determine whether the ECMO can be safely removed (14,24,25).
The weaning trials are also very important to evaluate the RV function. It is difficult to determine the functioning of the RV in maximal ECMO flow because the ECMO circuit creates negative pressure by draining venous blood from the right atrium (13,14,25). As ECMO flow decreases, preload of the RV increases allowing a better look on RV function (5,14,25). The occurrence of a right-LV interdependence defined as the dysfunction of one ventricle secondary to the other’s disorder during a weaning trial had a strong negative prognostic value in VA ECMO (15,25).
Which strategies for carrying out ECMO weaning trials?
In hemodynamic stable patients, two strategies of ECMO weaning trials directed by echocardiography have been reported: the first one using TTE (14) and the second one hemodynamic TEE (15). The choice should be guided by clinical parameters such as patient’s echogenicity and TEE contraindications.
When the TTE strategy is chosen, ECMO removal should be considered if a partially or fully recovery from the initial cardiac dysfunction has been observed, the full weaning trial has been tolerated and if a LVEF of >20–25%, a lateral mitral annulus peak systolic velocity ≥6 cm/s and an aortic VTI of >10 cm under minimal ECMO support have been described by a trained echocardiographist.
In the TEE study, four stages for the ECMO weaning should be completed (15). First, on full-flow ECMO support, baseline characteristics should be assessed: LV and RV volumes and ejection fraction. Secondly, as ECMO flow is gradually decreased in increments of 0.5 L/min to half of the original flow rate, hemodynamic tolerance (i.e., heart rate and blood pressure), RV and LV volumes and ejection fraction are evaluated step by step. The occurrence of a significant hypotension or a LV or a RV distension should lead to the end of the weaning trial and the ECMO support should be returned to full flow. During stage 3, the ECMO flow is reduced to a minimum rate of 1.2 to 1.5 L/min and hemodynamic responses are closely monitored during a volume challenge. At the final stage, stage 4, an infusion of inodilators—dobutamine and/or milrinone—on 4–6 hour-periods should be initiated in order to assess LV and RV function. Before any LVAD placement, a close look to RV function should be performed in patient with LV dysfunction. These drugs remain the best way to assess RV function in patients on ECMO. Final ECMO removal may be considered if both LV and RV functions recover (20-22). Otherwise, when a LV dysfunction persisted without RV failure, implantation of a left ventricular assist device may be discussed regarding contraindications.
Conclusions
Echocardiography is the key exam to lead on patients suffering from refractory CS. It guides the strategy by identifying CS etiology and helps in the indication of ECMO. In a daily use, it detects early recovery and complications such as LV thrombus. In the end, it leads weaning strategy.
Acknowledgments
None.
Footnote
Conflicts of Interest: N Aissaoui—Astra-Zeneca, Medtronic, Thoratec. The other author has no conflicts of interest to declare.
References
- Abrams D, Combes A, Brodie D. Extracorporeal Membrane Oxygenation in Cardiopulmonary Disease in Adults. J Am Coll Cardiol 2014;63:2769-78. [Crossref] [PubMed]
- Combes A, Brodie D, Chen YS, et al. The ICM research agenda on extracorporeal life support. Intensive Care Med 2017;43:1306-18. [Crossref] [PubMed]
- Abrams D, Garan AR, Abdelbary A, et al. Position paper for the organization of ECMO programs for cardiac failure in adults. Intensive Care Med 2018;44:717-29. [Crossref] [PubMed]
- Le Gall A, Follin A, Cholley B, et al. Veno-arterial-ECMO in the intensive care unit: From technical aspects to clinical practice. Anaesth Crit Care Pain Med 2018;37:259-68. [Crossref] [PubMed]
- Ortuno S, Delmas C, Diehl JL, et al. Weaning from veno-arterial extra-corporeal membrane oxygenation: which strategy to use? Ann Cardiothorac Surg 2019;8:E1-8. [Crossref] [PubMed]
- Bonello L, Delmas C, Schurtz G, et al. Mechanical circulatory support in patients with cardiogenic shock in intensive care units: A position paper of the "Unité de Soins Intensifs de Cardiologie" group of the French Society of Cardiology, endorsed by the "Groupe Athérome et Cardiologie Interventionnelle" of the French Society of Cardiology. Arch Cardiovasc Dis 2018;111:601-12. [Crossref] [PubMed]
- Vieillard-Baron A, Millington SJ, Sanfillipo F, et al. A decade of progress in critical care echocardiography: a narrative review. Intensive Care Med 2019. [Epub ahead of print].
- Levitov A, Frankel HL, Blaivas M, et al. Guidelines for the Appropriate Use of Bedside General and Cardiac Ultrasonography in the Evaluation of Critically Ill Patients-Part II: Cardiac Ultrasonography. Crit Care Med 2016;44:1206-27. [Crossref] [PubMed]
- Mebazaa A, Combes A, van Diepen S, et al. Management of cardiogenic shock complicating myocardial infarction. Intensive Care Med 2018. [Epub ahead of print]. [Crossref] [PubMed]
- Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J 2016;37:2129-200. [Crossref] [PubMed]
- Levy B, Bastien O, Karim B, et al. Experts' recommendations for the management of adult patients with cardiogenic shock. Ann Intensive Care 2015;5:52. [PubMed]
- Firstenberg MS, Orsinelli DA. ECMO and ECHO: the evolving role of quantitative echocardiography in the management of patients requiring extracorporeal membrane oxygenation. J Am Soc Echocardiogr 2012;25:641-3. [Crossref] [PubMed]
- Danial P, Hajage D, Nguyen LS, et al. Percutaneous versus surgical femoro-femoral veno-arterial ECMO: a propensity score matched study. Intensive Care Med 2018;44:2153-61. [Crossref] [PubMed]
- Aissaoui N, Luyt CE, Leprince P, et al. Predictors of successful extracorporeal membrane oxygenation (ECMO) weaning after assistance for refractory cardiogenic shock. Intensive Care Med 2011;37:1738-45. [Crossref] [PubMed]
- Cavarocchi NC, Pitcher HT, Yang Q, et al. Weaning of extracorporeal membrane oxygenation using continuous hemodynamic transesophageal echocardiography. J Thorac Cardiovasc Surg 2013;146:1474-9. [Crossref] [PubMed]
- Pappalardo F, Pieri M, Arnaez Corada B, et al. Timing and Strategy for Weaning From Venoarterial ECMO are Complex Issues. J Cardiothorac Vasc Anesth 2015;29:906-11. [Crossref] [PubMed]
- Aissaoui N, El-Banayosy A, Combes A. How to wean a patient from veno-arterial extracorporeal membrane oxygenation. Intensive Care Med 2015;41:902-5. [Crossref] [PubMed]
- Platts DG, Sedgwick JF, Burstow DJ, et al. The role of echocardiography in the management of patients supported by extracorporeal membrane oxygenation. J Am Soc Echocardiogr 2012;25:131-41. [Crossref] [PubMed]
- Donker DW, Meuwese CL, Braithwaite SA, et al. Echocardiography in extracorporeal life support: A key player in procedural guidance, tailoring and monitoring. Perfusion 2018;33:31-41. [Crossref] [PubMed]
- Douflé G, Roscoe A, Fan E, et al. Echocardiograohy for adult patients supported with extracorporeal membrane oxygentation. Crit Care 2015;19:326. [Crossref] [PubMed]
- ELSO Guidelines for Cardiopulmonary Extracorporeal Life Support Extracorporeal Life Support Organization, Version 1.3 November 2013 Ann Arbor, MI, USA. Available online: http://www.elsonet.org
- Durinka JB, Bogar LJ, Hirose H, et al. End-organ recovery is key to success for extracorporeal membrane oxygenation as a bridge to implantable left ventricular assist device. ASAIO J 2014;60:189-92. [Crossref] [PubMed]
- Huang KC, Lin LY, Chen YS, et al. Three-Dimensional Echocardiography-Derived Right Ventricular Ejection Fraction Correlates with Success of Decannulation and Prognosis in Patients Stabilized by Venoarterial Extracorporeal Life Support. J Am Soc Echocardiogr 2018;31:169-79. [Crossref] [PubMed]
- Aissaoui N, Guerot E, Combes A, et al. Two-dimensional strain rate and Doppler tissue myocardial velocities: analysis by echocardiography of hemodynamic and functional changes of the failed left ventricle during different degrees of extracorporeal life support. J Am Soc Echocardiogr 2012;25:632-40. [Crossref] [PubMed]
- Aissaoui N, Caudron J, Leprince P, et al. Right-left ventricular interdependence: a promising predictor of successful extracorporeal membrane oxygenation (ECMO) weaning after assistance for refractory cardiogenic shock. Intensive Care Med 2017;43:592-4. [Crossref] [PubMed]
Cite this article as: Bailleul C, Aissaoui N. Role of echocardiography in the management of veno-arterial extra-corporeal membrane oxygenation patients. J Emerg Crit Care Med 2019;3:25.


