
Management of pneumonia in intensive care
Introduction
Pneumonia is an inflammation of the alveolar airspace, most commonly triggered by bacteria but also arising from other classes of pathogen and less frequently by autoimmune processes. The infiltration of the alveolar space by leucocytes and a fibrinous exudate impairs lung function, and in its more severe forms can require invasive ventilation and admission to an intensive care unit (ICU). This review will focus on severe infectious pneumonia and its management in the ICU.
There are limited data on the incidence of severe pneumonia, although reports from the United States suggest nearly 20% of adults hospitalized with pneumonia will be admitted to ICU (1), with a clear seasonal pattern being evident. However the burden of pneumonia is considerably greater in low and middle income countries where the critical care resources are considerably less (2). Pneumonia is not only a precipitating factor leading to admission to ICU, but is also the commonest secondary infection acquired by critically ill patients (3). ICU-acquired pneumonia is largely ventilator-associated and presents specific problems of diagnosis and management (4).
The causative organisms in pneumonia are heavily influenced by where patients acquire the disease and the condition of their immune system (5), with differences in the flora causing community-acquired and hospital-acquired infections (6). Patients with impaired immune function are at risk of opportunistic pathogens such as fungi and otherwise ‘low pathogenicity’ bacteria (7), in addition to the more classically pathogenic organisms which infect the immunologically intact as well. This review will consider four broad categories of pneumonia, namely community-acquired, ventilator-acquired pneumonia, hospital-acquired and pneumonia in the immunocompromised host. It will also consider the overlap between pneumonia and acute respiratory distress syndrome (ARDS) (8).
The greatest challenges in the Intensive Care management of pneumonia are in the diagnosis and identification of the causative organisms, the selection of appropriate antibiotics and determining the duration of therapy, and in the prevention of secondary pneumonia. This review will cover the epidemiology of severe pneumonia as well as the aetiology and how our understanding of microbial ecology is changing the concept of infection. It will then turn to diagnosis, considering the existing diagnostic strategies and future directions in diagnostic technologies, before turning to management of both antibiotic and non-antibiotic therapies. Beyond antimicrobial therapy, management focuses on supportive therapy, protective ventilation and the avoidance of complications of ICU admission.
Epidemiology of severe pneumonia
Severe pneumonia lacks a unifying definition, however for community-acquired infection the Infectious Diseases Society of America/American Thoracic Society (IDSA/ATS) definition being pneumonia which requires admission to an ICU (9,10) is widely used. Although this definition concerns community-acquired pneumonia (CAP), it can also be extended to hospital-acquired pneumonia and pneumonia in the immunocompromised host. Ventilator-associated pneumonia (VAP), with its attributable mortality of 8–10% (11) and its restriction to critically ill patients, can be similarly considered ‘severe’.
ARDS, being defined as respiratory compromise with bilateral radiographic infiltrates not fully explained by cardiac failure (12), clearly has considerable overlap with the diagnosis of severe pneumonia. It is therefore unsurprising that pneumonia constitutes around 60% of patients in studies of ARDS (8). The evidence which underpins practice in ARDS applies to those patients with severe pneumonia who meet the criteria for ARDS, and has been reviewed recently elsewhere (13) and will be summarised below.
Amongst patients hospitalized with CAP, up to 20% may require admission to ICU (1), although this proportion may vary widely by country (14) and by availability of ICU beds (15), and shows marked seasonal variability (16). Pneumonia is the commonest cause of death in low income countries, whilst in upper-middle income and higher income countries it is the 6th commonest cause of death and the leading infectious cause of death (17). Estimates of the mortality rates from European countries show a wide range of reported values from <1% to 48% although differences in reporting may be responsible for some of this variation. From the perspective of an ICU clinician, in England at least, pneumonia remains the commonest infectious cause of admission, and carries a mortality rate of 35% (18) and in international surveys is the commonest infection found in ICU patients (19).
Pneumonia is also a frequent complication of hospital stay, with hospital-acquired pneumonias (HAP) developing in between 1% and 5% of all hospitalized patients (20,21). HAP is defined as pneumonia developing at least 48 hours after hospital admission, and is generally considered to be ‘non-ventilator associated’ as VAP is its own diagnostic entity (22). Giuliano and colleagues recently assessed the mortality associated with HAP in a large US database (21), the crude mortality of 13.1% exceeded that of a population matched for illness severity without pneumonia (mortality 11.2%) and those admitted with CAP (mortality 3.5%), with only patients with VAP having a higher mortality (17.5%). There is limited data on the rates of admission to ICU for HAP. However what data there is suggests that, despite the reported mortality, ICU admission may occur in only 3–5% of patients developing HAP (23). One of the significant difficulties in determining the epidemiology of HAP is the frequency with which it is over-diagnosed (20,23), an issue we will return to when considering VAP.
To further complicate matters, the entity of ‘healthcare associated pneumonia’ or HCAP had been proposed by the Infectious Diseases Society of America/American Thoracic Society (IDSA/ATS) (22), as a an infection developing in someone with close healthcare contact such as recent hospital admission, nursing home residence or in receipt of intravenous antibiotics within the past 30 days (22). It was felt that such a definition was required as patients with HCAP are thought to be at increased risk of infections with multi-drug resistant (MDR) organisms. However more recent studies examining the differences in MDR rates between CAP and HCAP have indicated that MDR organisms are not restricted to HCAP patients and can be predicted by individual patient characteristics rather than needing to be considered as a separate entity (5,24) and HCAP was removed from the most recent IDSA/ATS guidelines (25).
Patients with immunocompromise, be that through immunosuppressive drugs, infection with HIV, haematological malignancy or primary and secondary immune deficiency disorders are at risk of infections in general and pneumonia specifically. Whilst specific rates of infection will vary depending on the cause and severity of the immune deficiency, this group are worth highlighting because of their susceptibility to opportunistic infections such as fungi and parasites, alongside the more conventional pneumonia pathogens. This will be discussed further in the next section.
The final category of pneumonia which will be considered in this review is VAP, which by its very definition is solely confined to the ICU. VAP remains the commonest secondary infection to occur in ventilated ICU patients (26), despite considerable efforts to reduce and control this disease (27,28). VAP affects around 16% of ICU patients (29), although as incidence is significantly altered as a function of exposure (30) it is usually expressed as incident density (cases per 1,000 ventilator days) (31), with quoted rates of 13.6 cases per 1,000 ventilator days globally (29). The mortality associated with VAP is difficult to disentangle from the severity of the underlying illness, however the most rigorous estimates indicate that VAP incurs an additional mortality burden of 6–10% (30,32).
Microbiology of severe pneumonia
The microbial pathogens which cause pneumonia are influenced by the environmental exposure of the patient prior to them developing infection, as well as their underlying immune state. The generally accepted pathogenesis of bacterial pneumonia involves colonisation of the upper respiratory tract by the organism, followed by migration to the lower respiratory tract and proliferation leading to infection. It remains unclear why some people become colonized with pathogenic organisms but do not progress to pneumonia, whilst others do, although being resistant to colonisation with a specific organism does appear to protect against infection from that same organism (33). The acquisition of viral pneumonia is likely to differ from that of bacterial, in that exposure leads to viral infection and proliferation, although again there is a wide range of responses to viral infection ranging from resistance, through asymptomatic shedding to severe pneumonitis (34).
Across all types of pneumonia, culture-negative infection remains the commonest reported state, occurring in around 65% of CAP patients (35), and up to 70% of patients with suspected VAP (36) with similar rates reported for patients with HAP (37). Whilst some culture-negative results may reflect misdiagnosis, especially in the case of VAP where so many conditions may mimic it (38), a significant proportion are likely to be due to the imperfect nature of conventional microbiology (39). The limitations of current diagnostics will be discussed below, but any appreciation of the microbial ecology of pneumonia needs to be considered in this light.
When an organism is detected in CAP, the dominant organism remains Streptococcus pneumoniae, with gram negative organisms such as Haemophilus influenzae and Moraxella catarrhalis as well as the atypical or intracellular organisms (Legionella pneumophilia, Mycoplasma pneumoniae, Coxiella burnetii, Chlamydia psittaci). Although there is considerable anxiety about community-acquired staphylococcal pneumonia, and particularly methicillin resistant Staph. aureus (MRSA), surveillance reports suggest that it is rare (40,41).
With the advent of routine use of viral polymerase chain reaction (PCR) testing of respiratory samples, viral coinfection is increasingly recognised and may comprise up to a third of cases presenting to hospital (39). It is also now more widely accepted that several respiratory viruses, and not just influenza, can cause a widespread pneumonitis (42).
The microbiological aetiology of ventilator and hospital-acquired pneumonia differs somewhat from that seen in CAP, with gram-negative organisms being far more prevalent alongside Staphylococcus aureus which forms the predominant gram-positive organism. Table 1 below illustrates the difference.
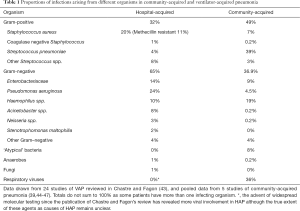
Full table
The microbial aetiology of HAP is similar to VAP (48,49), although there is a growing recognition of nosocomial viral infections (49), which have mostly been reported in HAP rather than VAP (43).
Fungal pneumonia is often considered in patients with immunocompromise, such as those taking immunosuppressive drugs for autoimmune disease or solid organ transplantation (50,51), haematology-oncology patients (52) and those with HIV (53), although broad-spectrum antibiotic use, mechanical ventilation and major surgery are also recognised risk factors (50,51). Critically ill patients frequently develop immunoparesis during the course of their illness (26,54), and fungal pneumonia is reported although remains rare in the absence of additional causes of immunosuppression such as neutropaenia or use of immunosuppressant drugs (43,55).
The reasons for the different microbial aetiology of community and hospital-acquired pneumonias may reflect different environmental exposure, as the hospital environment is much more likely to contain the multi-resistant gram-negative organisms which typify HAP/VAP (56). In addition, HAP/VAP tends to occur after patients have been exposed to antibiotics, which will exert selective pressure on microorganisms and lead to a predominantly resistant flora emerging (57). However it is also apparent that severe illness itself can induce a change in the pulmonary microbiota, with a shift towards enteric-type gram negative organisms and a reduction in diversity (58) which may be independent of inter-current antibiotic therapy (59). Changes in bacterial ingress, elimination and growth during acute illness alter the balance of lung microbiota (60), and it has been suggested that VAP (and possibly HAP) represent more ‘overgrowth’ of pathogenic bacteria rather than de-novo acquisition and infection (61). A high prevalence of oral commensals such as Mycoplasma salivarium has been reported in VAP, and these organisms may also drive immune suppression and facilitate further infection (62).
Diagnosis of pneumonia in ICU
Pneumonia is defined as inflammation of the airspaces (63), and as such the gold standard for diagnosis is histopathological examination. However, in the vast majority of cases, lung biopsy is neither practical nor desirable in a severely ill patient, whilst post-mortem examination is clearly too late to alter management. Clinicians are, therefore, required to rely on surrogate markers of alveolar inflammation and infection which present a degree of uncertainty.
Clinical criteria
The clinical criteria for pneumonia comprise evidence of systemic inflammation, such as pyrexia, tachycardia and leukocytosis/neutrophilia, combined with localizing chest signs such as rales, crepitations and bronchial breathing (64,65). These will frequently be accompanied by productive cough and breathlessness. However, clinical examination has limited sensitivity and specificity in the relatively uncomplicated primary care setting (66). It loses further diagnostic performance in Intensive Care where examination is restricted, mechanical ventilation induces auscultatory artifact (67) and where many disease processes may mimic the non-specific findings in pneumonia (38).
Given the poor performance of clinical criteria alone, these are often combined with radiographic techniques, including plain chest X-rays, ultrasound and computed tomography.
Radiographic criteria
Radiological investigations in suspected pneumonia aim to demonstrate the presence of alveolar inflammation, either in the form of lobar/sub-lobar consolidation or diffuse alveolitis. The gold standard for radiological examination of the lungs is computed tomography (CT), given its ability to localize and characterise pulmonary lesions to a much greater resolution than plain radiographs (67). However CT of the chest requires the patient to be moved off the ICU, which increases the risk of adverse events in both ventilated and non-ventilated patients (68) and is therefore not a routine investigation for ICU patients with suspected pneumonia.
Plain chest radiography (CXR) can be performed at the bedside, and is the recommended modality for imaging in CAP (9,64). Interestingly, neither the most recent European (69) nor American (25) guidelines on Hospital- and Ventilator-acquired pneumonia make recommendations on radiological investigation of pneumonia, despite the fact that nearly all clinical studies of HAP/VAP require presence of radiographic infiltrates as an inclusion criteria and both European Centre for Disease Control (70) and US Centres for Disease Control (71) definitions require radiographic demonstration of infiltrates. Amongst ICU clinicians, in the UK at least, up to 1/3rd do not think radiographic evidence is required to diagnose pneumonia (72).
Bedside ultrasound may outperform plain radiography (67,73) in the detection of consolidation, although lesions must be relatively superficial within the lung to be detected. To date no guidelines have adopted chest ultrasound as a recommended diagnostic modality, and no study has demonstrated its use changing outcomes for patients.
Microbiological culture
As noted at the start of this article, pneumonia requires the presence of an infectious organism and therefore detection of the responsible organism should guide treatment. Microbiological cultures are an imperfect method of detection, and frequently give rise of false negative results (39). In CAP the ATS/IDSA (9) and British Thoracic Society (BTS) guidelines (64) recommend routine microbiological sampling of sputum and blood in severe pneumonia, which will cover all cases admitted to Intensive Care, alongside specific antigen testing for Legionella pneumophilia and Streptococcus pneumoniae. The use of invasive sampling, by bronchoscopy or fine needle aspiration are discussed but not specifically recommended, in part due to a lack of systematic examination of the role of invasive sampling in CAP, and the generally good performance of sputum relative to bronchoalveolar lavage (BAL) when theses samples have been compared head to head (74). For the intubated patient, endotracheal aspirate (ETA) is a form of sputum sample which is less prone to contamination from the oropharynx and is the sample most likely to be available in ventilated ICU patients. Where BAL may have a role is in patients who do not have productive sputum or where there is a failure to respond to conventional treatment (74). The pre-test probability of pneumonia is higher in patients presenting with apparent CAP than VAP, and therefore culture or detection of a pathogenic organism is generally considered diagnostic without the need for quantitative cultures.
In VAP the need for deep respiratory cultures is equally strong, and both US and European guidelines recommend obtaining samples by ETA and undertaking quantitative or semi-quantitative cultures. Debate continues within the critical care and infectious disease community regarding the role of BAL and protected specimen brush (PSB), being more invasive than ETA, in the diagnosis of pneumonia. The issues revolve around the rapid and near universal colonisation of the trachea and upper bronchial tree within a few days of mechanical ventilation (75), and whether the benefits of targeting antimicrobial therapy outweigh the risks of invasive sampling. Given the high prevalence of upper bronchial colonisation and the lack of specificity of clinical features of pneumonia (38), ETA culture is likely to significantly overestimate the rate of true pneumonia (76), whilst applying high quantitative cut-offs may lead to false negative results (76). BAL and quantitative culture has therefore become the standard in biological research in VAP, where a well-defined clinical phenotype is required (77,78). What remains less certain is the benefit in clinical practice of invasive sampling, although intuitively directing antibiotics only at those patients with proven pneumonia seems to be rational approach.
There have been three randomised trials (79-81) investigating the role of invasive sampling on outcomes in patients with suspected pneumonia, with divergent results. Whilst Solé Violán and colleagues (79), and the Canadian Critical Care Trials Group (81) did not show any change in mortality, Fagon and colleagues (80) demonstrated a significant reduction in mortality. Whilst there may be many reasons for these discrepancies, it is notable that only Fagon and colleagues demonstrated a significant difference in antibiotic use, with the invasive strategy driving a substantial increase in antibiotic free days. Observational studies do suggest that implementation of invasive strategies in ICU can drive reduction in the use of antibiotics (76,82), and that stopping antibiotics in culture negative patients leads to fewer MDR infections without an increase in mortality (83).
In hospital-acquired pneumonia optimal diagnostic strategy remains uncertain, but obtaining good-quality respiratory secretions for culture by sputum, ETA or BAL is recommended (25). Specific recommendations for immunocompromised patients are lacking.
Molecular diagnostics
PCR
Although culture-free microbiological techniques, such as antigen testing, have existed for a long time, they are limited to single organisms and indeed often specific strains or types of those organisms. By contrast there have been significant recent developments in the field of PCR based diagnostics, which rely on the amplification and detection of specific genetic sequences. Selecting species or genus specific genetic sequences can allow the rapid and sensitive detection of relatively low numbers of gene-copy numbers (84), indicating the presence of an organism’s genetic material. The use of real-time (RT) PCR allows for quantitation of gene-copies, which provides an indication of the number of organisms present. However, the existence of multiple copies of a gene within some organisms means that there may not be a direct correlation between the PCR quantitation and organism number (85). Although clinical PCR for respiratory infections was pioneered in the diagnostics of viral infections, there are an increasing number of bacterial PCR-based diagnostics on the market and this number is likely to grow rapidly over the next years.
Multiple single PCR reactions are time consuming and expensive, and therefore groups have looked to combine them into multiplex reactions (86), although the optimisation required for these can be considerable and they may be increasingly susceptible to sample-based inhibitors and internal control failure (87).
The advantage of molecular techniques is that they do not rely on the organism being alive, or capable of division and growth. They are therefore less susceptible to the risks of false negative results arising from inter-current antibiotic use and have a much higher yield than culture-based techniques (39). However, their increased sensitivity runs the risk of detecting irrelevant or colonising organisms, and DNA from infecting pathogens may persist long after active infection has ended, thus these tests may increase rather than decrease the burden of antibiotics. Conversely the selectivity of these tests may also be a disadvantage, as organisms which are not covered by the included sequences will not be detected. To date, no study has been reported using bacterial or fungal PCR to guide clinical management of severe pneumonia.
Sequencing
An alternative molecular approach to detection of micro-organisms is to sequence the nucleic acids present and then match these against known sequences from databases of microbial DNA. The ubiquity of the 16s ribosomal RNA gene in every bacteria, with pan-bacterial conserved sequences and genus/species specific sequences (88) has been exploited to develop unbiased tests for bacteria. To date the diagnostic performance of these approaches has been disappointing (89), although total burden of 16s DNA may act as a useful marker of bacterial load in VAP (90) and blood culture (91). Until recently the alternative approach of sequencing all base-pairs present in a sample, so called metagenomics, has been prohibitively expensive and too slow to be of use clinically although it forms the basis of many studies of the lung microbiome (58). However the development of devices such as the Nanopore device (92) may bring this approach to clinical utility soon. As with targeted PCR, these highly sensitive approaches risk the detection of clinically irrelevant organisms, and their use requires careful evaluation in well conducted trials before they can be recommended for routine clinical use.
Novel optical techniques
Several novel techniques have been developed for the detection of bacteria, including the use of fluorescent probes for in-vivo imaging via devices such as the alveolar fibroscope developed by Dhaliwal et al. (93). These probes may be combined with those for activated neutrophils (93) so allowing combined confirmation of alveolar bacterial presence and inflammation, the hallmark of pneumonia. These devices remain experimental, and they are currently someway from clinical use.
Ex-vivo automated microscopy of samples, allowing detection of growth of bacteria at much lower numbers than required for conventional microbial culture also holds promise, allowing for the rapid detection of viable bacteria. This approach may provide rapid, sensitive testing whilst reducing the risks of detecting irrelevant organisms (94).
Host markers
The second component of pneumonia is the inflammatory host response, and this has been the focus of considerable research interest over the past couple of decades. Biomarkers can be measured in both pulmonary secretions, ETA or lavage, or blood.
Although the list of lavage based biomarkers of ventilator-acquired pneumonia which have been advanced is considerable (95), only one test (based on alveolar concentrations of IL-1 and IL-8) has been successfully validated (77,78). This test is undergoing evaluation as a measure to improve antibiotic stewardship, with the trial having been completed and results expected to be published soon (96) (NCT01972425).
There has been much less work on pulmonary biomarkers in community-acquired and non-VAP hospital-acquired pneumonia, and whilst it is likely that the intense pulmonary inflammation which accompanies VAP (97) is mirrored in these diseases, this remains to be conclusively demonstrated.
Circulating host biomarkers such as C-reactive protein (CRP) and procalcitonin (PCT) are in routine use as monitors for infection in many ICUs, however evidence of their diagnostic utility in hospital-acquired pneumonia is limited (98,99) and their use for this purpose is not recommended in guidelines (25,69). By contrast, there is some evidence of utility of CRP in community acquired pneumonia (100) and BTS guidelines suggest it can be used to discriminate between pneumonia and non-pneumonic processes (64), whilst the IDSA/ATS guidelines remain silent on the issue (9). This divergence between HAP/VAP and CAP will likely reflect the differing pre-test probability of pneumonia seen in patients presenting with a de-novo infection and those developing secondary complications in hospital. Where PCT may have a role is in the guidance of de-escalation and early discontinuation of antibiotics, with this being studied in critically ill patients with pneumonia being the predominant diagnosis (101). However, patients in the standard management arms of PCT studies tend to have prolonged courses of antibiotics, in the PRORATA study median duration of antibiotic therapy was 13 days whilst in the PCT group it was 10 days (102), both of which may be longer than is required to treat most pneumonias (see below). By contrast in De Jong’s recent study, median antibiotic duration was reduced from 7 to 5 days, with a significant and persistent mortality benefit in the PCT group (101).
Severity scoring and determining which patients require admission to ICU
There are a number of pneumonia severity scores, which use a range of clinical, demographic and laboratory parameters to score patients and assign a severity band to them. This work has come almost entirely from the CAP literature, which is unsurprising as these patients will present to a range of settings from primary care to emergency departments and there is a need to identify those requiring hospital admissions. Patients with CAP who are ventilated immediately have a mortality of 24%, but those who deteriorate at a later time point have a mortality of 49% (14). Although it is unclear to what extent this mortality difference was due to delays in care and to what extent it was due to different disease trajectories, early admission to ICU for patients with markers of severe pneumonia appears prudent.
The two most commonly used pneumonia severity scores, the Pneumonia Severity Index (PSI) (103) and CURB-65 scores (104) are both well validated predictors of mortality at 30 days. However their ability to predict ICU admission or requirement for mechanical ventilation is moderate at best (105), as are the severe pneumonia criteria set out by in the ATS/IDSA guidelines (9). A significant proportion of patients with pneumonia die without being admitted to ICU, due to treatment limitation decisions and comorbidities, and therefore it is perhaps unsurprising that predictors of 30 day mortality may not correspond well with need for ICU admission (10). Kolditz and colleagues identified a number of readily determinable factors which predicted ICU admission, including focal chest signs at presentation, multilobar involvement on CXR, comorbid conditions especially home oxygen requirement and physiological instability (14). However, these criteria have not yet been validated in an external cohort, and neither has a specific score nor criteria been developed.
Management of pneumonia in ICU—antimicrobials
The corner stone of pneumonia management is the administration of appropriate antibiotics in a timely fashion. Delay, and especially administration of antibiotics which do not cover the infecting organism are associated with adverse outcomes including increased mortality and prolonged length of stay (106). However, given the problems outlined in the microbiological diagnosis of pneumonia above, the infecting organism is almost never known at the time of illness onset and clinicians must use empiric therapy targeted at likely organisms. This approach leads to early use of broad-spectrum antibiotics, especially in patients who are high risk of MDR infection such as those with VAP, HAP, and immunocompromised patients. Once the results of microbiological cultures are known, antibiotics should be adjusted and where appropriate deescalated (9,25,64,69), however this occurs variably in practice (107).
Antibiotic selection should be informed by the local antibiogram, combining data on the prevalent organisms and their likely resistance patterns. Updating the local antibiogram is an important wider societal reason for obtaining microbiological cultures in patients with pneumonia. Given the empiric nature of therapy, it is important to recognise the differences in organism and resistance patterns depending on location of acquisition and risk factors for MDR bacteria (see epidemiology section above). In CAP, empiric therapy is mostly aimed at gram-positive organisms, specifically Streptococcus pneumoniae, and generally low-resistance gram-negative organisms such as Haemophilus influenzae alongside ‘atypical’ bacteria. Although specific recommended antibiotics will vary by national availability and local antibiogram, a beta-lactam antibiotic with beta-lactamase resistance or combined with a beta-lactamase inhibitor such as co-amoxiclav or a 2nd generation cephalosporin are advised (6,64). The addition of a macrolide to empiric therapy for CAP is strongly recommended (9,64) and is associated with a reduction in mortality (108). It is unclear if this benefit is solely due to coverage of hard-to-detect atypical organisms, or if the immunomodulatory effects of macrolides are also important (109).
In patients who are at greater risk of MDR bacteria empiric therapy needs to cover these organisms, often including Pseudomonas and methicillin resistant Staphylococcus aureus (MRSA). These two organisms in particular pose a problem as they are frequently resistant to most front-line antibiotics, and their treatment can involve antibiotics such as aminoglycosides and glycopeptides, which have an unfavourable toxicity profile (110). Empiric therapy runs the risk of over-treatment and thus increases the risk of toxic side effects. Therefore, patients need to be assessed for their risk of harbouring these particularly difficult to treat organisms prior to initiating treatment. Risk factors for Pseudomonas and MRSA include prior intravenous antibiotic use, previous culture of these organisms from a colonising or infecting site and recent hospitalisation of >5 days. The problem is that these risk factors describe many of the patients who develop VAP and HAP. The use of dual antibiotic therapy vs monotherapy in HAP and VAP has been examined in several trials (111-113) and a meta-analysis (114). These suggest that, in populations with a moderate rate of pseudomonas infection (14%), no benefit to dual therapy (114). Observational studies, which examine patients with high disease severity, who are often excluded from clinical trials, suggest that patients with septic shock may benefit from dual therapy (115). As a result the European guidelines for HAP and VAP advocate dual therapy for patients with high disease severity (69) whilst IDSA/ATS guidelines advocate dual therapy for those with risk factors for resistant Pseudomonas or MRSA (25). However, the findings of Kett and colleagues sound a note of caution over this advice, they observed that patients with HAP treated with IDSA/ATS compliant dual antibiotic therapy had a higher mortality than those treated with monotherapy (116). For patients with, or at risk of, extended spectrum beta-lactamase secreting organisms, carbapenems appear to be superior to piperacillin-tazobactam (117). Although this data comes from blood stream infections mostly arising from urinary and intraabdominal sources, it seems reasonable to apply it to pneumonias at present. In both CAP and HAP/VAP it is likely that the development and widespread adoption of rapid diagnostics will lead to more targeted therapy and will allow more precise trialling of antibiotic strategies.
Nebulised antibiotics are used in the management of MDR bacteria in several contexts, and is supported by the American guidelines on HAP and VAP (25), however this is based on weak evidence and many units do not use nebulised antibiotics at all (118). Where they are used, practice is variable and seldom in accordance with the advised best practice (118). Before this approach achieves widespread adoption, well conducted trials are needed.
The optimal duration of antibiotic therapy in pneumonia remains uncertain. There have been several trials which have examined fixed duration treatments (119-121), which have been analysed in systematic reviews (122). These trials compared ‘short’ (7–8 days) with long (10–15 days) of therapy, and in meta-analysis there was no benefit to prolonged treatment in terms of mortality, cure rate or recurrence, whilst prolonged therapy was associated with increased risk of subsequent MDR infection. As these studies excluded patients with lung abscesses and collections, empyema, necrotising pneumonia and bronchiectasis/cystic fibrosis, this approach cannot be extrapolated to these patients. Concern about possible recurrence of Pseudomonal, and other non-fermenting gram-negative organism, infection with short course antibiotics (119) has led to a recommendation that patients with these infections should have 14 days of therapy (25,69). Fixed duration antibiotic therapy applies fairly arbitrary time-scales to patients and fails to account for individual variation. There is now reasonable evidence that ‘low risk’ patients, as determined by falling CPIS scores or falling PCT levels can have their antibiotics stopped at shorter time points, without harm and with some evidence of benefit (101,123), so allowing more individualised therapy. Conversely, if a patient is failing to improve a reassessment of needs to occur, looking for incorrect diagnosis (for instance missing a non-infective inflammatory process), resistant organisms, inappropriate therapy, super-infection or the development of collections, empyema and lung abscesses.
Management of pneumonia
Adjunctive therapy
Beyond antibiotic therapy, a number of adjuvant therapies have been trialled in pneumonia although none have a proven role in critically ill patients in ICU (124).
There is some evidence that corticosteroids may enhance recovery in CAP and reduce mortality (125,126), however these trials enrolled very few patients who were in ICU or required mechanical ventilation. Concerns have been raised about the use of steroids to treat severe CAP in ICU (127), with the few observational studies which focus on this group suggesting steroids are associated with a prolonged length of stay (128) and increased mortality (129). In patients with A/H1N1 pandemic influenza, use of steroids was associated with increased mortality and an increase rate of subsequent HAP (130). Hopefully the forthcoming CAPE-COD study of corticosteroids in critically ill patients with CAP will shed more light on this area (NCT02517489). With the exception of those developing vasopressor resistant septic shock (131), there appears to be no place for the use of steroids in the management of HAP and VAP. In patients with HIV who develop Pneumocystis jirovecii pneumonia steroids have been demonstrated to improve outcomes (132), however this is not proven in patients with PCP without HIV and their use in other immunocompromised patients without proven PCP is controversial (133).
Other investigators have adopted an alternative approach of immuno-stimulation in pneumonia, acknowledging the evidence of immune cell failure in both early and late sepsis (26,134). To date immuno-stimulation has been attempted without determining patients’ functional immune status, the failure to appropriately target therapy may explain the negative results from trials of G-CSF in pneumonia (135). Alternative strategies such as GM-CSF (136) and interferon gamma (137) are under investigation but no large randomised trials have yet been published (124). Augmented passive immunity is another strategy which shows promise is early work, although here human trials are awaited (138).
Ventilation
The need for respiratory support is the commonest reason for patients with pneumonia to be admitted to ICU (14). There are a growing range of respiratory support options, from simple oxygen therapy, through high flow humidified oxygen (HFO), non-invasive ventilation (NIV) and continuous positive airway pressure (CPAP) via various interface devices to full invasive ventilation. HFO has gained considerable popularity in recent years and has in certain circumstances demonstrable benefits relative to NIV/CPAP. Frat and colleagues demonstrated a reduction in intubation rates in patients with more severe respiratory failure and a decreased 90-day mortality compared to simple oxygen therapy and NIV (139), the majority of patients in this study presented with pneumonia. More recently however, in patients with immunocompromise and respiratory failure, HFO did not reduce intubation rates or mortality relative to simple oxygen therapy (140). Whilst NIV has a proven role in the management of exacerbations of chronic obstructive pulmonary disease (COPD) (141), its role in pneumonia including pneumonic exacerbations of COPD is largely restricted to rescue therapy pending mechanical ventilation (64) and intubation should not be delayed by the use of NIV (142).
In patients requiring invasive ventilation, the optimal mode of ventilation remains to be determined. As noted above, pneumonia is the commonest cause of ARDS (8), and as ARDS is frequently missed (8,13), care should be taken to look for this syndrome in patients admitted with or developing severe pneumonia. Patients who meet the criteria for moderate to severe ARDS should be managed with low volume (6 mL/kg predicted body weight) tidal ventilation and limited plateau pressures (143). Adjunctive therapy such as muscle relaxation (144) and early proning (145) are also indicated for those meeting severe ARDS criteria. Care should be taken to avoid fluid overload in patients with severe pneumonia (146), and consideration given to de-resuscitation and trying to minimise extra-vascular lung water (147). For patients who don’t meet moderate to severe ARDS criteria the evidence is less clear, a recent trial from Schultz and colleagues demonstrated no benefit of low tidal volumes relative to moderate (8–10 mL/kg PBW) in patients without ARDS (148), however, this study included less than 20% patients with pneumonia. It remains unclear whether patients who are at higher risk of developing ARDS will benefit from low rather than moderate tidal ventilation (149).
Management—prevention of secondary complications
Patients with severe pneumonia in ICU are at risk of the complications of critical care, including deep venous thrombosis (150), stress ulceration (151), decubitus ulcers (152), delirium (153) and secondary infections (19). Careful attention to the risk factor management, appropriate prophylactic therapies and alertness to the development of these complications is key to good clinical management. Patients who present with a primary pneumonia, whether it is community or hospital-acquired, are at high risk of subsequent nosocomial infections, especially if ventilated (30). The various preventative measures for VAP have been reviewed extensively elsewhere (154), their implementation can lead to a reduction in VAP incidence, reduction in antibiotic use and mortality in longer staying patients (27). The reduction in antibiotic use is likely to drive a virtuous circle, reducing colonisation with MDR bacteria and subsequent infections with these hard to treat organisms.
Conclusions
Pneumonia remains both a common reason for intensive care admission and the commonest secondary infection acquired within intensive care. Its effective management relies on the selection of appropriate antimicrobial therapy, which at present is reliant on good epidemiological surveillance to inform empirical antibiotic choice. Identifying patients with risk factors for MDR organisms is critical to ensuring early, appropriate therapy. Diagnostic uncertainty, especially with regards to VAP, likely leads to over-use of antibiotics and brings with it the risk of antibiotic-associated harm. The advent of rapid diagnostic strategies brings the promise of targeted, appropriate therapy and reduction in unnecessary drugs. For this promise to be realised we will need to change our culture of prescribing and understanding of microbiological tests results in a clinical context.
Acknowledgements
Dr. Conway Morris is supported by grants from the Wellcome Trust (WT 2055214/Z/16/Z), Academy of Medical Sciences and European Intensive Care Society.
Footnote
Conflicts of Interest: The author has no conflicts of interest to declare.
References
- Storms AD, Chen J, Jackson LA, et al. Rates and risk factors associated with hospitalization for pneumonia with ICU admission among adults. BMC Pulm Med 2017;17:208. [Crossref] [PubMed]
- Rudan I, O'Brien KL, Nair H, et al. Epidemiology and etiology of childhood pneumonia in 2010: estimates of incidence, severe morbidity, mortality, underlying risk factors and causative pathogens for 192 countries. J Glob Health 2013;3:010401. [PubMed]
- Healthcare-associated infections acquired in intensive care units - Annual Epidemiological Report 2016 [2014 data]. European Centre for Disease Prevention and Control. January 2017:1-9.
- Hunter JD. Ventilator associated pneumonia. BMJ 2012;344:e3325. [Crossref] [PubMed]
- Shindo Y, Ito R, Kobayashi D, et al. Risk Factors for Drug-Resistant Pathogens in Community-acquired and Healthcare-associated Pneumonia. Am J Respir Crit Care Med 2013;188:985-95. [Crossref] [PubMed]
- Cilloniz C, Martin-Loeches I, Garcia-Vidal C, et al. Microbial Etiology of Pneumonia: Epidemiology, Diagnosis and Resistance Patterns. Int J Mol Sci 2016;17:E2120. [Crossref] [PubMed]
- Letourneau AR, Issa NC, Baden LR. Pneumonia in the immunocompromised host. Curr Opin Pulm Med 2014;20:272-9. [Crossref] [PubMed]
- Bellani G, Laffey JG, Pham T, et al. Epidemiology, Patterns of Care, and Mortality for Patients With Acute Respiratory Distress Syndrome in Intensive Care Units in 50 Countries. JAMA 2016;315:788-800. [Crossref] [PubMed]
- Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society Consensus Guidelines on the Management of Community-Acquired Pneumonia in Adults. Clin Infect Dis 2007;44:S27-72. [Crossref] [PubMed]
- Chalmers JD. Identifying severe community-acquired pneumonia: moving beyond mortality. Thorax 2015;70:515-6. [Crossref] [PubMed]
- Nguile-Makao M, Zahar JR, Français A, et al. Attributable mortality of ventilator-associated pneumonia: respective impact of main characteristics at ICU admission and VAP onset using conditional logistic regression and multi-state models. Intensive Care Med 2010;36:781-9. [Crossref] [PubMed]
- ARDS Definition Task Force, Ranieri VM, Rubenfeld GD, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA 2012;307:2526-33. [PubMed]
- Laffey JG, Misak C, Kavanagh BP. Acute respiratory distress syndrome. BMJ 2017;359:j5055. [Crossref] [PubMed]
- Kolditz M, Ewig S, Klapdor B, et al. Community-acquired pneumonia as medical emergency: predictors of early deterioration. Thorax 2015;70:551-8. [Crossref] [PubMed]
- Hua M, Halpern SD, Gabler NB, et al. Effect of ICU strain on timing of limitations in life-sustaining therapy and on death. Intensive Care Med 2016;42:987-94. [Crossref] [PubMed]
- Burrell A, Huckson S, Pilcher DV. ICU Admissions for Sepsis or Pneumonia in Australia and New Zealand in 2017. N Engl J Med 2018;378:2138-9. [Crossref] [PubMed]
- World Health Organisation. Global health observatory data. Available online: http://www.who.int/gho/mortality_burden_disease/en/, accessed 31/10/18.
- Intensive Care National Audit and Research Centre (ICNARC). Key statistics from the case mix program 2015-2016. Available online: www.icnarc.org/, accessed 31/10/18.
- Vincent JL, Rello J, Marshall J, et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA 2009;302:2323-9. [Crossref] [PubMed]
- Burton LA, Price R, Barr KE, et al. Hospital-acquired pneumonia incidence and diagnosis in older patients. Age Ageing 2016;45:171-4. [Crossref] [PubMed]
- Giuliano KK, Baker D, Quinn B. The epidemiology of nonventilator hospital-acquired pneumonia in the United States. Am J Infect Control 2018;46:322-7. [Crossref] [PubMed]
- American Thoracic Society, Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 2005;171:388-416. [Crossref] [PubMed]
- Russell CD, Koch O, Laurenson IF, et al. Diagnosis and features of hospital-acquired pneumonia: a retrospective cohort study. J Hosp Infect 2016;92:273-9. [Crossref] [PubMed]
- Yap V, Datta D, Metersky ML. Is the Present Definition of Health Care–Associated Pneumonia the Best Way to Define Risk of Infection with Antibiotic-Resistant Pathogens? Infect Dis Clin North Am 2013;27:1-18. [Crossref] [PubMed]
- Kalil AC, Metersky ML, Klompas M, et al. Management of Adults With Hospital-acquired and Ventilator-associated Pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis 2016;63:e61-111. [Crossref] [PubMed]
- Conway Morris A, Datta D, Shankar-Hari M, et al. Cell-surface signatures of immune dysfunction risk-stratify critically ill patients: INFECT study. Intensive Care Med 2018;44:627-35. [Crossref] [PubMed]
- Morris AC, Hay AW, Swann DG, et al. Reducing ventilator-associated pneumonia in intensive care: Impact of implementing a care bundle. Crit Care Med 2011;39:2218-24. [Crossref] [PubMed]
- Álvarez-Lerma F, Palomar-Martínez M, Sánchez-García M, et al. Prevention of Ventilator-Associated Pneumonia. Crit Care Med 2018;46:181-8. [Crossref] [PubMed]
- Kollef MH, Chastre J, Fagon JY, et al. Global Prospective Epidemiologic and Surveillance Study of Ventilator-Associated Pneumonia due to Pseudomonas aeruginosa. Crit Care Med 2014;42:2178-87. [Crossref] [PubMed]
- van Vught LA, Klein Klouwenberg PM, Spitoni C, et al. Incidence, Risk Factors, and Attributable Mortality of Secondary Infections in the Intensive Care Unit After Admission for Sepsis. JAMA 2016;315:1469-79. [Crossref] [PubMed]
- Walsh TS, Morris AC, Simpson AJ. Ventilator associated pneumonia: can we ensure that a quality indicator does not become a game of chance? Br J Anaesth 2013;111:333-7. [Crossref] [PubMed]
- Timsit JF, Zahar JR, Chevret S. Attributable mortality of ventilator-associated pneumonia. Curr Opin Crit Care 2011;17:464-71. [Crossref] [PubMed]
- Bogaert D, de Groot R, Hermans PW. Streptococcus pneumoniae colonisation: the key to pneumococcal disease. Lancet Infect Dis 2004;4:144-54. [Crossref] [PubMed]
- Riley S, Kwok KO, Wu KM, et al. Epidemiological Characteristics of 2009 (H1N1) Pandemic Influenza Based on Paired Sera from a Longitudinal Community Cohort Study. PLoS Med 2011;8:e1000442. [Crossref] [PubMed]
- Cillóniz C, Ewig S, Polverino E, et al. Microbial aetiology of community-acquired pneumonia and its relation to severity. Thorax 2011;66:340-6. [Crossref] [PubMed]
- Hellyer TP, Morris AC, McAuley DF, et al. Diagnostic accuracy of pulmonary host inflammatory mediators in the exclusion of ventilator-acquired pneumonia. Thorax 2015;70:41-7. [Crossref] [PubMed]
- Schlueter M, James C, Dominguez A, et al. Practice patterns for antibiotic de-escalation in culture-negative healthcare-associated pneumonia. Infection 2010;38:357-62. [Crossref] [PubMed]
- Meduri GU, Mauldin GL, Wunderink RG, et al. Causes of fever and pulmonary densities in patients with clinical manifestations of ventilator-associated pneumonia. Chest 1994;106:221-35. [Crossref] [PubMed]
- Gadsby NJ, Russell CD, McHugh MP, et al. Comprehensive Molecular Testing for Respiratory Pathogens in Community-Acquired Pneumonia. Clin Infect Dis 2016;62:817-23. [Crossref] [PubMed]
- Nakou A, Woodhead M, Torres A. MRSA as a cause of community-acquired pneumonia. Eur Respir J 2009;34:1013-4. [Crossref] [PubMed]
- Self WH, Wunderink RG, Williams DJ, et al. Staphylococcus aureus Community-acquired Pneumonia: Prevalence, Clinical Characteristics, and Outcomes. Clin Infect Dis 2016;63:300-9. [Crossref] [PubMed]
- Gerna G, Piralla A, Rovida F, et al. Correlation of rhinovirus load in the respiratory tract and clinical symptoms in hospitalized immunocompetent and immunocompromised patients. J Med Virol 2009;81:1498-507. [Crossref] [PubMed]
- Chastre J, Fagon JY. Ventilator-associated pneumonia. Am J Respir Crit Care Med 2002;165:867-903. [Crossref] [PubMed]
- Rello J. Microbiological Testing and Outcome of Patients With Severe Community-Acquired Pneumonia. Chest 2003;123:174-80. [Crossref] [PubMed]
- Ruiz M, Ewig S, Torres A, et al. Severe community-acquired pneumonia. Risk factors and follow-up epidemiology. Am J Respir Crit Care Med 1999;160:923-9. [Crossref] [PubMed]
- Wilson PA, Ferguson J. Severe community-acquired pneumonia: an Australian perspective. Intern Med J 2005;35:699-705. [Crossref] [PubMed]
- Moine P, Vercken JB, Chevret S, et al. Severe community-acquired pneumonia. Etiology, epidemiology, and prognosis factors. French Study Group for Community-Acquired Pneumonia in the Intensive Care Unit. Chest 1994;105:1487-95. [Crossref] [PubMed]
- Kumar S. Healthcare-Associated Pneumonia and Hospital-Acquired Pneumonia: Bacterial Aetiology, Antibiotic Resistance and Treatment Outcomes: A Study From North India. Lung 2018;196:469-79. [Crossref] [PubMed]
- Micek ST, Chew B, Hampton N, et al. A Case-Control Study Assessing the Impact of Nonventilated Hospital-Acquired Pneumonia on Patient Outcomes. Chest 2016;150:1008-14. [Crossref] [PubMed]
- Limper AH. The changing spectrum of fungal infections in pulmonary and critical care practice: clinical approach to diagnosis. Proc Am Thorac Soc 2010;7:163-168. [Crossref] [PubMed]
- Singh N. Trends in the epidemiology of opportunistic fungal infections: predisposing factors and the impact of antimicrobial use practices. Clin Infect Dis 2001;33:1692-6. [Crossref] [PubMed]
- Marr KA, Carter RA, Crippa F, et al. Epidemiology and outcome of mould infections in hematopoietic stem cell transplant recipients. Clin Infect Dis 2002;34:909-17. [Crossref] [PubMed]
- Thomas CF, Limper AH. Current insights into the biology and pathogenesis of Pneumocystis pneumonia. Nat Rev Microbiol 2007;5:298-308. [Crossref] [PubMed]
- Conway Morris A, Kefala K, Wilkinson TS, et al. C5a mediates peripheral blood neutrophil dysfunction in critically ill patients. Am J Respir Crit Care Med 2009;180:19-28. [Crossref] [PubMed]
- Park DR. The microbiology of ventilator-associated pneumonia. Respir Care 2005;50:742-63. [PubMed]
- Dancer SJ. Controlling hospital-acquired infection: focus on the role of the environment and new technologies for decontamination. Clin Microbiol Rev 2014;27:665-90. [Crossref] [PubMed]
- White AC, Atmar RL, Wilson J, et al. Effects of requiring prior authorization for selected antimicrobials: expenditures, susceptibilities, and clinical outcomes. Clin Infect Dis 1997;25:230-9. [Crossref] [PubMed]
- Dickson RP, Singer BH, Newstead MW, et al. Enrichment of the lung microbiome with gut bacteria in sepsis and the acute respiratory distress syndrome. Nat Microbiol 2016;1:16113. [Crossref] [PubMed]
- Zakharkina T, Martin-Loeches I, Matamoros S, et al. The dynamics of the pulmonary microbiome during mechanical ventilation in the intensive care unit and the association with occurrence of pneumonia. Thorax 2017;72:803-10. [Crossref] [PubMed]
- Dickson RP, Huffnagle GB. The Lung Microbiome: New Principles for Respiratory Bacteriology in Health and Disease. PLoS Pathogens 2015;11:e1004923. [Crossref] [PubMed]
- Dickson RP, Erb-Downward JR, Huffnagle GB. The role of the bacterial microbiome in lung disease. Expert Rev Respir Med 2013;7:245-57. [Crossref] [PubMed]
- Nolan TJ, Gadsby NJ, Hellyer TP, et al. Low-pathogenicity Mycoplasma spp. alter human monocyte and macrophage function and are highly prevalent among patients with ventilator-acquired pneumonia. Thorax 2016;71:594-600. [Crossref] [PubMed]
- Rouby JJ, Martin De Lassale E, Poete P, et al. Nosocomial bronchopneumonia in the critically ill. Histologic and bacteriologic aspects. Am Rev Respir Dis 1992;146:1059-66. [Crossref] [PubMed]
- Lim WS, Baudouin SV, George RC, et al. BTS guidelines for the management of community acquired pneumonia in adults: update 2009. Thorax 2009;64 Suppl 3:iii1-iii55. [Crossref] [PubMed]
- Suetens C, Morales I, Savey A, et al. European surveillance of ICU-acquired infections (HELICS-ICU): methods and main results. J Hosp Infect 2007;65 Suppl 2:171-3. [Crossref] [PubMed]
- Wipf JE, Lipsky BA, Hirschmann JV, et al. Diagnosing pneumonia by physical examination: relevant or relic? Arch Intern Med 1999;159:1082-7. [Crossref] [PubMed]
- Lichtenstein D, Goldstein I, Mourgeon E, et al. Comparative diagnostic performances of auscultation, chest radiography, and lung ultrasonography in acute respiratory distress syndrome. Anesthesiology 2004;100:9-15. [Crossref] [PubMed]
- Jia L, Wang H, Gao Y, et al. High incidence of adverse events during intra-hospital transport of critically ill patients and new related risk factors: a prospective, multicenter study in China. Crit Care 2016;20:12. [Crossref] [PubMed]
- Torres A, Niederman MS, Chastre J, et al. International ERS/ESICM/ESCMID/ALAT guidelines for the management of hospital-acquired pneumonia and ventilator-associated pneumonia: Guidelines for the management of hospital-acquired pneumonia (HAP)/ventilator-associated pneumonia (VAP) of the European Respiratory Society (ERS), European Society of Intensive Care Medicine (ESICM), European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and Asociación Latinoamericana del Tórax (ALAT). Eur Respir J 2017;50:1700582. [Crossref] [PubMed]
- Plachouras D, Lepape A, Suetens C. ECDC definitions and methods for the surveillance of healthcare-associated infections in intensive care units. Intensive Care Med. [Epub ahead of print].
- NHSN Surveillance Definitions for Specific Types of Infections. Available online: http://admin.inicc.org/media/2015-CDCNHSN-ALLDA-HAI-Definitions.pdfCDC/
- Browne E, Hellyer TP, Baudouin SV, et al. A national survey of the diagnosis and management of suspected ventilator-associated pneumonia. BMJ Open Resp Res 2014;1:e000066. [Crossref] [PubMed]
- Long L, Zhao HT, Zhang ZY, et al. Lung ultrasound for the diagnosis of pneumonia in adults. Medicine (Baltimore) 2017;96:e5713. [Crossref] [PubMed]
- van der Eerden MM, Vlaspolder F, de Graaff CS, et al. Value of intensive diagnostic microbiological investigation in low- and high-risk patients with community-acquired pneumonia. Eur J Clin Microbiol Infect Dis 2005;24:241-9. [Crossref] [PubMed]
- Cavalcanti M, Valencia M, Torres A. Respiratory nosocomial infections in the medical intensive care unit. Microbes Infect 2005;7:292-301. [Crossref] [PubMed]
- Morris AC, Kefala K, Simpson A, et al. Evaluation of diagnostic methodology on the reported incidence of ventilator-associated pneumonia. Thorax 2009;64:516-22. [Crossref] [PubMed]
- Conway Morris A, Kefala K, Wilkinson TS, et al. Diagnostic importance of pulmonary interleukin-1 beta and interleukin-8 in ventilator-associated pneumonia. Thorax 2010;65:201-7. [Crossref] [PubMed]
- Grover V, Pantelidis P, Soni N, et al. A Biomarker Panel (Bioscore) Incorporating Monocytic Surface and Soluble TREM-1 Has High Discriminative Value for Ventilator-Associated Pneumonia: A Prospective Observational Study. PLoS One 2014;9:e109686. [Crossref] [PubMed]
- Solé Violán J, Fernández JA, Benítez AB, et al. Impact of quantitative invasive diagnostic techniques in the management and outcome of mechanically ventilated patients with suspected pneumonia. Crit Care Med 2000;28:2737-41. [Crossref] [PubMed]
- Fagon JY, Chastre J, Wolff M, et al. Invasive and noninvasive strategies for management of suspected ventilator-associated pneumonia. A randomized trial. Ann Intern Med 2000;132:621-30. [Crossref] [PubMed]
- Canadian Critical Care Trials Group. A randomized trial of diagnostic techniques for ventilator-associated pneumonia. N Engl J Med 2006;355:2619-30. [Crossref] [PubMed]
- Bonten MJ, Bergmans DC, Stobberingh EE, et al. Implementation of bronchoscopic techniques in the diagnosis of ventilator-associated pneumonia to reduce antibiotic use. Am J Respir Crit Care Med 1997;156:1820-4. [Crossref] [PubMed]
- Raman K, Nailor MD, Nicolau DP, et al. Early Antibiotic Discontinuation in Patients With Clinically Suspected Ventilator-Associated Pneumonia and Negative Quantitative Bronchoscopy Cultures. Crit Care Med 2013;41:1656-63. [Crossref] [PubMed]
- Zemanick ET, Wagner BD, Sagel SD, et al. Reliability of Quantitative Real-Time PCR for Bacterial Detection in Cystic Fibrosis Airway Specimens. PLoS One 2010;5:e15101. [Crossref] [PubMed]
- Gross L. The More the Merrier: Multiple Genomes in a Parasitic Prokaryote. PLoS Biol 2006;4:e212. [Crossref] [PubMed]
- Gadsby NJ, McHugh MP, Russell CD, et al. Development of two real-time multiplex PCR assays for the detection and quantification of eight key bacterial pathogens in lower respiratory tract infections. Clin Microbiol Infect 2015;21:788.e1-13. [Crossref] [PubMed]
- Jeffrey M. Perkel Overcoming the challenges of multiplex PCR. Available online: https://www.biocompare.com/Editorial-Articles/117895-Multiplex-PCR/, Accessed 31/10/18.
- Renvoisé A, Brossier F, Sougakoff W, et al. Broad-range PCR: Past, present, or future of bacteriology? Med Mal Infect 2013;43:322-30. [Crossref] [PubMed]
- Tkadlec J, Peckova M, Sramkova L, et al. The use of broad-range bacterial PCR in the diagnosis of infectious diseases: a prospective cohort study. Clin Micro Infect. [Epub ahead of print].
- Conway Morris A, Gadsby N, McKenna JP, et al. 16S pan-bacterial PCR can accurately identify patients with ventilator-associated pneumonia. Thorax 2017;72:1046-8. [Crossref] [PubMed]
- Su G, Fu Z, Hu L, et al. 16S Ribosomal Ribonucleic Acid Gene Polymerase Chain Reaction in the Diagnosis of Bloodstream Infections: A Systematic Review and Meta-Analysis. PLoS One 2015;10:e0127195. [Crossref] [PubMed]
- Moon J, Jang Y, Kim N, et al. Diagnosis of Haemophilus influenzae Pneumonia by Nanopore 16S Amplicon Sequencing of Sputum. Emerg Infect Dis 2018;24:1944-6. [Crossref] [PubMed]
- Akram AR, Chankeshwara SV, Scholefield E, et al. In situ identification of Gram-negative bacteria in human lungs using a topical fluorescent peptide targeting lipid A. Sci Transl Med 2018;10:eaal0033.
- Jung JH, Lee JE. Real-time bacterial microcolony counting using on-chip microscopy. Sci Rep 2016;6:21473. [Crossref] [PubMed]
- Salluh JI, Souza-Dantas VC, Póvoa P. The current status of biomarkers for the diagnosis of nosocomial pneumonias. Curr Opin Crit Care 2017;23:391-7. [Crossref] [PubMed]
- Hellyer TP, Anderson NH, Parker J, et al. Effectiveness of biomarker-based exclusion of ventilator-acquired pneumonia to reduce antibiotic use (VAPrapid-2): study protocol for a randomised controlled trial. Trials 2016;17:318. [Crossref] [PubMed]
- Wilkinson TS, Conway Morris A, Kefala K, et al. Ventilator-Associated Pneumonia Is Characterized by Excessive Release of Neutrophil Proteases in the Lung. Chest 2012;142:1425. [Crossref] [PubMed]
- Póvoa P, Coelho L, Almeida E, et al. C-reactive protein as a marker of ventilator-associated pneumonia resolution: a pilot study. Eur Respir J 2005;25:804-12. [Crossref] [PubMed]
- Pfister R, Kochanek M, Leygeber T, et al. Procalcitonin for diagnosis of bacterial pneumonia in critically ill patients during 2009 H1N1 influenza pandemic: a prospective cohort study, systematic review and individual patient data meta-analysis. Crit Care 2014;18:R44. [Crossref] [PubMed]
- Castro-Guardiola A, Armengou-Arxé A, Viejo-Rodríguez A, et al. Differential diagnosis between community-acquired pneumonia and non-pneumonia diseases of the chest in the emergency ward. Eur J Intern Med 2000;11:334-9. [Crossref] [PubMed]
- de Jong E, van Oers JA, Beishuizen A, et al. Efficacy and safety of procalcitonin guidance in reducing the duration of antibiotic treatment in critically ill patients: a randomised, controlled, open-label trial. Lancet Infect Dis 2016;16:819-27. [Crossref] [PubMed]
- Bouadma L, Luyt CE, Tubach F, et al. Articles Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet 2010;375:463-74. [Crossref] [PubMed]
- Fine MJ, Auble TE, Yealy DM, et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med 1997;336:243-50. [Crossref] [PubMed]
- Lim WS, van der Eerden MM, Laing R, et al. Defining community acquired pneumonia severity on presentation to hospital: an international derivation and validation study. Thorax 2003;58:377-82. [Crossref] [PubMed]
- Chalmers JD, Mandal P, Singanayagam A, et al. Severity assessment tools to guide ICU admission in community-acquired pneumonia: systematic review and meta-analysis. Intensive Care Med 2011;37:1409-20. [Crossref] [PubMed]
- Iregui M, Ward S, Sherman G, Fraser VJ, Kollef MH. Clinical Importance of Delays in the Initiation of Appropriate Antibiotic Treatment for Ventilator-Associated Pneumonia. Chest 2002;122:262-268. [Crossref] [PubMed]
- Kollef MH, Morrow LE, Niederman MS, et al. Clinical characteristics and treatment patterns among patients with ventilator-associated pneumonia. Chest 2006;129:1210-8. [Crossref] [PubMed]
- Sligl WI, Asadi L, Eurich DT, et al. Macrolides and Mortality in Critically Ill Patients With Community-Acquired Pneumonia. Crit Care Med 2014;42:420-32. [Crossref] [PubMed]
- Zimmermann P, Ziesenitz VC, Curtis N, et al. The Immunomodulatory Effects of Macrolides—A Systematic Review of the Underlying Mechanisms. Front Immunol 2018;9:302. [Crossref] [PubMed]
- Heavner MS, Claeys KC, Masich AM, et al. Pharmacokinetic and Pharmacodynamic Considerations of Antibiotics of Last Resort in Treating Gram-Negative Infections in Adult Critically Ill Patients. Curr Infect Dis Rep 2018;20:10. [Crossref] [PubMed]
- Fernández-Guerrero M, Gudiol F, Rodriguez-Torres A, et al. Nosocomial pneumonia: comparative multicentre trial between monotherapy with cefotaxime and treatment with antibiotic combinations. Infection 1991;19 Suppl 6:S320-5. [Crossref] [PubMed]
- Jaspers CA, Kieft H, Speelberg B, et al. Meropenem versus cefuroxime plus gentamicin for treatment of serious infections in elderly patients. Antimicrob Agents Chemother 1998;42:1233-8. [Crossref] [PubMed]
- Rubinstein E, Lode H, Grassi C. Ceftazidime monotherapy vs. ceftriaxone/tobramycin for serious hospital-acquired gram-negative infections. Antibiotic Study Group. Clin Infect Dis 1995;20:1217-28. [Crossref] [PubMed]
- Paul M, Lador A, Grozinsky-Glasberg S, et al. Beta lactam antibiotic monotherapy versus beta lactam-aminoglycoside antibiotic combination therapy for sepsis. Cochrane Database Syst Rev 2014.CD003344. [PubMed]
- Kumar A, Safdar N, Kethireddy S, et al. A survival benefit of combination antibiotic therapy for serious infections associated with sepsis and septic shock is contingent only on the risk of death: A meta-analytic/meta-regression study. Crit Care Med 2010;38:1651-64. [Crossref] [PubMed]
- Kett DH, Cano E, Quartin AA, et al. Implementation of guidelines for management of possible multidrug-resistant pneumonia in intensive care: an observational, multicentre cohort study. Lancet Infect Dis 2011;11:181-9. [Crossref] [PubMed]
- Harris PNA, Tambyah PA, Lye DC, et al. Effect of Piperacillin-Tazobactam vs Meropenem on 30-Day Mortality for Patients With E colior Klebsiella pneumoniae Bloodstream Infection and Ceftriaxone Resistance. JAMA 2018;320:984-94. [Crossref] [PubMed]
- Alves J, Alp E, Koulenti D, et al. Nebulization of antimicrobial agents in mechanically ventilated adults in 2017: an international cross-sectional survey. Eur J Clin Microbiol Infect Dis 2018;37:785-94. [Crossref] [PubMed]
- Chastre J, Wolff M, Fagon JY, et al. Comparison of 8 vs 15 days of antibiotic therapy for ventilator-associated pneumonia in adults: a randomized trial. JAMA 2003;290:2588-98. [Crossref] [PubMed]
- Capellier G, Mockly H, Charpentier C, et al. Early-Onset Ventilator-Associated Pneumonia in Adults Randomized Clinical Trial: Comparison of 8 versus 15 Days of Antibiotic Treatment. PLoS One 2012;7:e41290. [Crossref] [PubMed]
- Kollef MH, Chastre J, Clavel M, et al. A randomized trial of 7-day doripenem versus 10-day imipenem-cilastatin for ventilator- associated pneumonia. Crit Care 2012;16:R218. [Crossref] [PubMed]
- Pugh R, Grant C, Cooke RP, et al. Short‐course versus prolonged‐course antibiotic therapy for hospital‐acquired pneumonia in critically ill adults. Cochrane Database Syst Rev 2011.CD007577. [PubMed]
- Singh N, Rogers P, Atwood CW, et al. Short-course empiric antibiotic therapy for patients with pulmonary infiltrates in the intensive care unit. A proposed solution for indiscriminate antibiotic prescription. Am J Respir Crit Care Med 2000;162:505-11. [Crossref] [PubMed]
- Morton B, Pennington SH, Gordon SB. Immunomodulatory adjuvant therapy in severe community-acquired pneumonia. Expert Rev Respir Med 2014;8:587-96. [Crossref] [PubMed]
- Torres A, Sibila O, Ferrer M, et al. Effect of Corticosteroids on Treatment Failure Among Hospitalized Patients With Severe Community-Acquired Pneumonia and High Inflammatory Response. JAMA 2015;313:677-86. [Crossref] [PubMed]
- Blum CA, Nigro N, Briel M, et al. Adjunct prednisone therapy for patients with community- acquired pneumonia: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet 2015;385:1511-8. [Crossref] [PubMed]
- Póvoa P, Salluh JIF. What is the role of steroids in pneumonia therapy? Curr Opin Infect Dis 2012;25:199-204. [Crossref] [PubMed]
- Salluh JIF, Soares M, Coelho LM, et al. Impact of systemic corticosteroids on the clinical course and outcomes of patients with severe community-acquired pneumonia: a cohort study. J Crit Care 2011;26:193-200. [Crossref] [PubMed]
- Chon GR, Lim CM, Koh Y, et al. Analysis of systemic corticosteroid usage and survival in patients requiring mechanical ventilation for severe community-acquired pneumonia. J Infect Chemother 2011;17:449-55. [Crossref] [PubMed]
- The ESICM H1N1 Registry Contributors, Martin-Loeches I, Lisboa T, et al. Use of early corticosteroid therapy on ICU admission in patients affected by severe pandemic (H1N1)v influenza A infection. Intensive Care Med 2010;37:272-83.
- Annane D, Renault A, Brun-Buisson C, et al. Hydrocortisone plus Fludrocortisone for Adults with Septic Shock. N Engl J Med 2018;378:809-18. [Crossref] [PubMed]
- Ewald H, Raatz H, Boscacci R, et al. Adjunctive corticosteroids for Pneumocystis jiroveci pneumonia in patients with HIV infection. Cochrane Database Syst Rev 2015.CD006150. [PubMed]
- Wieruszewski PM, Barreto JN, Frazee E, et al. Early Corticosteroids for Pneumocystis Pneumonia in Adults Without HIV Are Not Associated With Better Outcome. Chest 2018;154:636-644. [Crossref] [PubMed]
- Shankar-Hari M, Datta D, Wilson J, et al. Early PREdiction of sepsis using leukocyte surface biomarkers: the ExPRES-sepsis cohort study. Intensive Care Med 2018;44:1836-48. [Crossref] [PubMed]
- Bo L, Wang F, Zhu J, et al. Granulocyte-colony stimulating factor (G-CSF) and granulocyte-macrophage colony stimulating factor (GM-CSF) for sepsis: a meta-analysis. Crit Care 2011;15:R58. [Crossref] [PubMed]
- Pinder EM, Rostron AJ, Hellyer TP, et al. Randomised controlled trial of GM-CSF in critically ill patients with impaired neutrophil phagocytosis. Thorax 2018;73:918-25. [Crossref] [PubMed]
- Nakos G, Malamou-Mitsi VD, Lachana A, et al. Immunoparalysis in patients with severe trauma and the effect of inhaled interferon-gamma. Crit Care Med 2002;30:1488-94. [Crossref] [PubMed]
- Morton B, Mitsi E, Pennington SH, et al. Augmented Passive Immunotherapy with P4 Peptide Improves Phagocyte Activity in Severe Sepsis. Shock 2016;46:635-41. [Crossref] [PubMed]
- Frat JP, Thille AW, Mercat A, et al. High-Flow Oxygen through Nasal Cannula in Acute Hypoxemic Respiratory Failure. N Engl J Med 2015;372:2185-96. [Crossref] [PubMed]
- Azoulay E, Lemiale V, Mokart D, et al. Effect of High-Flow Nasal Oxygen vs Standard Oxygen on 28-Day Mortality in Immunocompromised Patients With Acute Respiratory Failure. JAMA 2018. [Epub ahead of print]. [Crossref] [PubMed]
- Ram FSF, Picot J, Lightowler J, et al. Non-invasive positive pressure ventilation for treatment of respiratory failure due to exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2004.CD004104. [PubMed]
- Carrillo A, Gonzalez-Diaz G, Ferrer M, et al. Non-invasive ventilation in community-acquired pneumonia and severe acute respiratory failure. Intensive Care Med 2012;38:458-66. [Crossref] [PubMed]
- Acute Respiratory Distress Syndrome Network, Brower RG, Matthay MA, et al. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The Acute Respiratory Distress Syndrome Network. N Engl J Med 2000;342:1301-8. [Crossref] [PubMed]
- Papazian L, Forel JM, Gacouin A, et al. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med 2010;363:1107-16. [Crossref] [PubMed]
- Guérin C, Reignier J, Richard JC, et al. Prone Positioning in Severe Acute Respiratory Distress Syndrome. N Engl J Med 2013;368:2159-68. [Crossref] [PubMed]
- Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006;354:2564-75. [Crossref] [PubMed]
- Chao WC, Tseng CH, Chien YC, et al. Association of day 4 cumulative fluid balance with mortality in critically ill patients with influenza: A multicenter retrospective cohort study in Taiwan. PLoS One 2018;13:e0190952. [Crossref] [PubMed]
- Writing Group for the PReVENT Investigators, Simonis FD, Serpa Neto A, et al. Effect of a Low vs Intermediate Tidal Volume Strategy on Ventilator-Free Days in Intensive Care Unit Patients Without ARDS. JAMA 2018;320:1872-80. [Crossref] [PubMed]
- Lipes J, Bojmehrani A, Lellouche F. Low Tidal Volume Ventilation in Patients without Acute Respiratory Distress Syndrome: A Paradigm Shift in Mechanical Ventilation. Crit Care Res Pract 2012;2012:416862. [Crossref] [PubMed]
- Cook DJ, Crowther MA, Meade MO, et al. Prevalence, incidence, and risk factors for venous thromboembolism in medical-surgical intensive care unit patients. J Crit Care 2005;20:309-13. [Crossref] [PubMed]
- Krag M, Marker S, Perner A, et al. Pantoprazole in Patients at Risk for Gastrointestinal Bleeding in the ICU. N Engl J Med 2018. [Epub ahead of print]. [Crossref] [PubMed]
- Richardson A, Peart J, Wright SE, et al. Reducing the incidence of pressure ulcers in critical care units: a 4-year quality improvement. Int J Qual Health Care 2017;29:433-9. [Crossref] [PubMed]
- Salluh JIF, Wang H, Schneider EB, et al. Outcome of delirium in critically ill patients: systematic review and meta-analysis. BMJ 2015;350:h2538. [Crossref] [PubMed]
- Branch-Elliman W, Wright SB, Howell MD. Determining the Ideal Strategy for Ventilator-associated Pneumonia Prevention. Cost–Benefit Analysis. Am J Respir Crit Care Med 2015;192:57-63. [Crossref] [PubMed]
Cite this article as: Conway Morris A. Management of pneumonia in intensive care. J Emerg Crit Care Med 2018;2:101.


