
Euglycemic ketoacidosis in a postoperative deformity correction spine surgery patient: a case report
Introduction
SGLT2 inhibitors are used in the treatment of type 2 diabetics in the outpatient setting. This class of drug acts on the kidneys to control serum glucose (1). SGLT-2 is a protein found in the proximal convoluted tubules and is involved in glucose reabsorption (2). Induced euglycemic diabetic ketoacidosis occurs secondary to the increased renal excretion of glucose. The fall in serum glucose decreases insulin secretion by the pancreas. These changes lead to increased glucagon to insulin ratio resulting in increased gluconeogenesis. The final result is increased ketogenesis and the development of ketoacidosis.
These drugs are relatively new to the management of diabetes. The lack of ICU provider experience with these drugs can lead to inappropriate ICU management. A good history and physical, medication review and basic laboratory values can diagnose SGLT-2 inhibitor euglycemic ketoacidosis. We have discussed a case of SGLT-2 inhibitor euglycemic ketoacidosis in a post-surgical patient in the ICU.
Case presentation
A 79-year-old Caucasian male was admitted to our Neuroscience Intensive Care Unit (NICU) status post extension of spinal fusion and rod replacement due to rod fracture, disk herniation above his previous decompression, and recurrent back pain. His construct now spans T4–S1 with bilateral iliac bolts. The estimated blood loss (EBL) was 1.5 liters. He has a history of back pain, type 2 diabetes, ulcerative colitis, depression, hypertension, hyperlipidemia, gastroesophageal reflux disease, stage 3 chronic kidney disease, and arthritis. His outpatient prescriptions were obtained through the electronic medical record (Table 1).
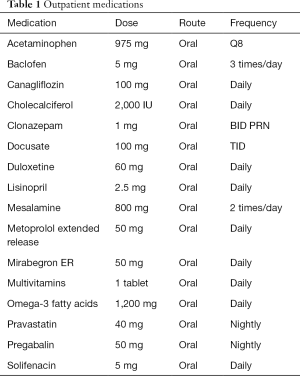
Full table
Standard blood counts, chemistries, coagulation studies, and urinalysis were obtained. He was again diagnosed with severe obstructive sleep apnea (OSA) with an apnea-hypopnea index (AHI) >30 and his CPAP settings were increased from 4 to 11 cmH2O. The pre-op labs were shown in Table 2.
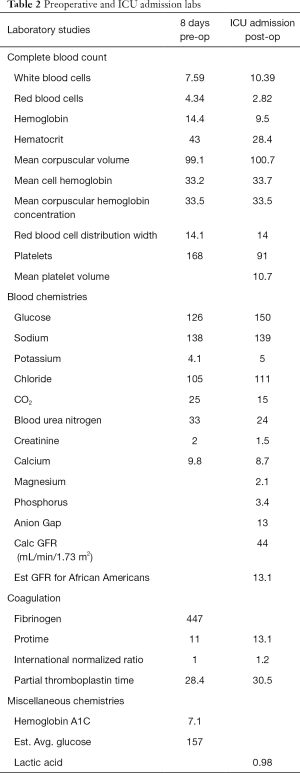
Full table
The patient’s baseline arterial blood gas in the operating room indicated a metabolic acidosis without respiratory compensation, as demonstrated by the following values: (see Table 3). The anesthesiology team aggressively resuscitated the patient with 2 liters of isotonic crystalloids during his surgery as well as with 2 liters albumin, one unit of packed red blood cells (PRBC’s), one unit of fresh frozen plasma (FFP), one unit of cryoprecipitate, and one unit pooled platelets. Despite aggressive resuscitative efforts, the patient’s blood gas on admission to the ICU was severely acidotic (Table 3). The anesthesia team extubated the patient and within minutes transported him to the neuro ICU for ongoing resuscitation and post-operative management.
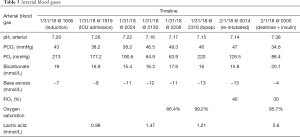
Full table
Aggressive volume resuscitation continued in the ICU with an additional 2 L of isotonic crystalloids. The total resuscitation volume from the OR and ICU was 7 L. However, looking for the source of his refractory acidemia was deemed prudent. A full set of labs (standard care in the management of this institution’s postoperative spine patients) was sent and CPAP was initiated, with suspicion that some acidemia may be due to relative hypoventilation if he was trying to compensate with respiratory drive but was unable to increase his minute ventilation appropriately. The results of the patient’s initial post-op labs are shown in Table 2.
Despite resuscitative efforts and titration of non-invasive positive pressure ventilation (NIPPV), the patient’s blood gases failed to improve (Table 3). After 5 hours in the ICU with continued support, the patient’s mental status began to decline with increasing somnolence. The decision was made to electively intubate the patient for airway protection and impending respiratory failure. Diagnostic testing continued into the cause of the patient’s acidemia. Levels of methanol and ethanol were negative. Uric acid was within normal limits. Polyethylene glycol was not tested. The patient’s lactate level was normal in the OR and on arrival to the ICU. The acetone level resulted at “moderate”. This confirmed a ketoacidosis without significant anion gap, confirming the Euglycemic Ketoacidosis diagnosis. Further confirmation was obtained with a urine ketone level of “moderate”. IV glucose infusion and insulin infusion was started and the ketoacidosis resolved. The patient was discharged home on postoperative day 9.
Discussion
Clinical diagnosis: Invokana induced euglycemic diabetic ketoacidosis
The diagnosis of acidosis in perioperative and intensive care patients is a relatively common problem to encounter. Acidosis is separated into respiratory or metabolic in origin. For most complex surgical patients that remain intubated, respiratory acidosis is corrected by changing ventilator settings to allow for better carbon dioxide removal. This is easily accomplished with increasing the minute ventilation or allowing a longer expiratory time. Metabolic acidosis is broken down to anion gap and non-anion gap acidosis. A long standing mnemonic used in medicine for anion gap acidosis is MUDPILES. This consists of methanol, uremia, diabetic ketoacidosis, propylene glycol/paracetamol, infection/isoniazid, lactic acidosis, ethylene glycol and salicylates. Non anion gap acidosis in the intensive care unit is normally related to a small number of disease processes. This is comprised of hyperalimentation, drugs such as acetazolamide or spironolactone, renal tubular acidosis, severe diarrhea and volume resuscitation with only normal saline. Our postoperative neurosurgical population rarely encounters non anion gap acidosis unless fluid resuscitation is maintained primarily with normal saline leading to hyperchloremia.
On closer examination of the patient’s laboratory values in the operating room and on admission to the intensive care unit, a metabolic acidosis was identified. On further discussion with the anesthesiologist, this acidosis was present early in the surgical case despite adequate perfusion pressures with mean arterial pressures maintained between 65–70 mmHg. The blood pH remained in the acidotic state despite volume resuscitation in the operating room and the ICU. The lactic acid remained normal through resuscitation. The patients remained hemodynamically stable through resuscitation and when the anion gap was calculated, it was normal. This was not the expected acidosis that is typically present in our spine surgery population. The differential diagnosis now had to be re-visited as the etiology did not fit our running diagnosis of non-anion gap acidosis. Our first thought was that the patient had a renal tubular acidosis that was undiagnosed. Reviewing the patient’s labs eliminated this diagnosis quickly. His serum potassium and bicarbonate were normal preoperatively. The patient also did not respond to a bicarbonate infusion as would be expected in RTA. The patient also had known chronic kidney disease and metabolic acidosis from AKI was in our differential. Unfortunately, the patients serum creatinine remained stable and below baseline. All surgical patients remain NPO the night before, therefore starvation ketoacidosis was a possibility. In addition, strong ion difference acidosis was possible with the massive resuscitation in the operating room. Starvation ketoacidosis can appear after an overnight fast but it typically requires 3 to 14 days of starvation to reach maximal severity. Even with prolonged fasting the acidosis is normally mild and the plasma pH rarely goes below 7.3. Strong Ion Difference acidosis is primarily seen with aggressive normal saline resuscitation. Our patient received balanced salt solutions and colloids making this diagnosis less likely
After further review of the patient’s medical records, it was discovered that the patient was on Invokana (canagliflozin). This is a SGLT-2 inhibitors used to treat type 2 diabetics in the outpatient setting. This medication is commonly known to cause an anion gap acidosis but in this case that was not present. This presentation even confused the consulting physicians. Ultimately, we initiated our original plan of care and the patient’s acidosis resolved. This brought to light the importance of expanding our differential diagnosis of acidosis as more medications are being made to treat common disease processes. It addresses the importance of obtaining a detailed history from the patients and thoroughly reviewing the patient’s medical records and medications.
SGLT2 inhibitor acidosis is becoming a more common pathology. It is of utter most importance that physicians in the preoperative, intraoperative and postoperative period be aware of the physiologic disturbance cause by these drugs. The mainstay of treatment of induced euglycemic diabetic ketoacidosis is prevention. Medical providers should attempt to avoid prolonged NPO periods, acute illnesses that can induce ketoacidosis and acutely decreasing insulin dosing. If euglycemic diabetic ketoacidosis is discovered, the SGLT-2 drug should be immediately stopped and glucose administered. Based on the clinical situation an insulin infusion might need to be started. Medical providers should consider the supplementation of glucose while the patient remains NPO such as the use of glucose containing solution in the operating room and ICU.
Correct identification of SGLT2 inhibitor induced euglycemic diabetic ketoacidosis is imperative in managing the patient’s acidosis. Euglycemic diabetic ketoacidosis is defined by the presence of a metabolic acidosis (pH <7.3) with a serum bicarbonate <18 mEq/L, ketosis and a blood glucose <200 mg/dL (3). Although this is a not a diagnostic standard for induced euglycemic diabetic ketoacidosis, it does help medical providers identify this endocrine disturbance once other causes have been ruled out. Basic laboratory test such as arterial blood gas, basic metabolic panel and serum ketone can identify euglycemic diabetic ketoacidosis. Of important note is that the patient may present with or without an anion gap. This can be calculated from the values obtained in the basic metabolic panel. Urinalysis can also be useful for detecting ketones and elevated urine glucose.
Given the rarity of this diagnosis, consultations were made to both Nephrology and Endocrinology/Metabolism. As nephrology is in house 24/7, they were the first to consult, and doubted the diagnosis and suggested infusion of a solution of 0.45% saline with 75 mEq of sodium bicarbonate for treatment of what they suspected was renal tubular acidosis. They also suggested an endocrinology consultation.
Endocrinology also doubted the diagnosis, as the case reports they could initially find all presented with a high anion gap of 15–20, and given this patient’s gap of 10–15 were also leaning towards RTA. They suggested a renal consult.
Ultimately the ICU decided to execute their treatment plan of dextrose infusion and insulin infusion to break his ketosis (4). Both consulting services agreed that this was a low risk intervention and deferred to our clinical judgment.
Outcome
The patient was given one amp of D50 and continued on plasmalyte-A solution at 125 mL/h. An insulin infusion was also initiated at 3 U/h and capillary blood glucoses were checked hourly. Within 4 hours the patient’s blood gases were in Table 3.
The IV solution was changed to D5 0.45% saline and the insulin drip was titrated per our hospital’s critical care insulin infusion protocol. The patient was extubated the following afternoon without difficulty and made an uneventful recovery. He was monitored in the ICU for two additional days to ensure his anion gap remained closed and no further metabolic sequelae ensued. He was discharged to rehab on postoperative day 9.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: The patient has given consent for this case report to be published.
References
- Rosenstock J, Ferrannini E. Euglycemic Diabetic Ketoacidosis: A Predictable, Detectable, and Preventable Safety Concern With SGLT2 Inhibitors. Diabetes Care 2015;38:1638-42. [Crossref] [PubMed]
- Fralick M, Schneeweiss S, Patorno E. Risk of Diabetic Ketoacidosis after Initiation of an SGLT2 Inhibitor. N Engl J Med 2017;376:2300-2. [Crossref] [PubMed]
- Modi A, Agrawal A, Morgan F. Euglycemic Diabetic Ketoacidosis: A Review. Curr Diabetes Rev 2017;13:315-21. [Crossref] [PubMed]
- Candelario N, Wykretowicz J. The DKA that wasn't: a case of euglycemic diabetic ketoacidosis due to empagliflozin. Oxf Med Case Reports 2016;2016:144-6. [Crossref] [PubMed]
Cite this article as: Theodore D, Pongracz B, Titus B. Euglycemic ketoacidosis in a postoperative deformity correction spine surgery patient: a case report. J Emerg Crit Care Med 2018;2:83.


