
Fluid assistance therapy reduces risks of hospital-acquired pulmonary fungal infection for acute exacerbation of chronic obstructive pulmonary disease patients
Introduction
Pulmonary fungal infection is one of the most important types of deep fungal infection. Infection accounts for a considerable proportion of patients in the respiratory ward, and the infection rate of patients with acute exacerbation of chronic obstructive pulmonary disease (AECOPD) is high. For patients admitted for acute exacerbation in GOLD groups C and D according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD), the treatment course is long, and pulmonary fungus infection can easily develop. After the fungal infection, the period of treatment must be extended, the economic burden increases, and there is more suffering and a greater risk of death. Invasive pulmonary aspergillosis is one kind of pulmonary fungus. In an observational multicenter study of 563 critically ill patients with proven putative invasive pulmonary aspergillosis or aspergillus colonization, researchers found that COPD was the most frequent cause, accounting for 31% (n=174) of all cases (1).
However, drugs for antifungal therapy have several shortcomings that can’t be ignored, such as a long course of treatment, no significant curative effects, obvious side effects, and high cost. Reducing probability of lung fungal infection in AECOPD patients during hospitalization is an important issue. In our many years’ clinical observations, when AECOPD patients in GOLD groups C and D are given an appropriate increase of intravenous fluid based on routine treatment, it could effectively reduce the patients’ probability of complications with pulmonary fungal infection. This research investigates such patients who were treated in our department from March 2013 to February 2016.These patients were randomly divided into two groups: one that received fluid assistance therapy and one that did not, and the probability of pulmonary fungal infection was compared between them. This study aimed to find a new method to decrease the probability of pulmonary fungal infection. If fluid assistance therapy could reduce pulmonary fungal infection, it would be easy for clinicians applying this means and get less complications comparing with using antifungal drug.
Methods
Study population
This study was a prospective cohort study. We investigated AECOPD patients in GOLD groups C and D who were hospitalized in the respiratory ward of our hospital from March 2013 to February 2016. Originally 209 patients were chosen, and 209 three-digit random numbers were read by using the random number list. Each patient was given a corresponding random number according to the order after their admission of hospitalization. Then these 209 random numbers were sorted by ascending power, the first 73 patients were treated by fluid assistance therapy, the other 136 were treated by non-fluid assistance therapy. This research adopted the double-blind design, the patients and outcomes evaluators were all blind, and implementers were open.
All of the patients’ diagnosis and treatment processes were carried out according to the guidelines for the diagnosis and treatment of AECOPD. Based on routine practice, one group received fluid assistance therapy, and the other group did not (routine treatment).
The fluid assistance therapy involved an intravenous infusion amount of 800–1,800 mL, while the non-therapy group received an intravenous infusion of 800 mL or less. Possible liquids included physiological saline, glucose saline solution, and sodium lactate Ringer’s solution, to which electrolyte may or may not have been added. The probability of pulmonary fungal infection was compared between groups. The diagnosis of pulmonary fungal infection was carried out according to the current expert consensus (2): once a diagnosis of pulmonary fungal infection had been made, a corresponding antifungal treatment was given to the patients. We did outpatient follow-up every month since patients took the antifungal drug and follow-up would last for about 3 to 6 months.
Inclusion criteria (Figure 1)
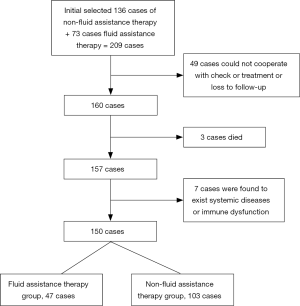
All patients included satisfied the following criteria: (I) all patients were diagnosed with AECOPD in groups C and D by established standards, and they all had type II respiratory failure; (II) they were unable to move freely and could not eat or drink by themselves; (III) the patients weighed 35–90 kg; (IV) they may or may not have used glucocorticoids by oral or atomization inhalation or intravenously (intravenous use for 7 days or more, oral use for 10 days or more, atomization inhalation use for 7 days or more); (V) their brain natriuretic peptide (BNP) levels were 5,000 pg/mL or less. (VI) They had no history of fungal infections in the last half year before the admission.
Exclusion criteria (3-9)
Patients were excluded for the following reasons: (I) immune defects or using immune inhibitors; (II) diseases of the blood system; (III) rheumatism; (IV) abnormal structure of the throat or upper respiratory tract were abnormal or having undergone related surgery; (V) unsmooth vital signs; (VI) large pulmonary bullae (with a diameter of 2 cm or more); (VII) use of endotracheal intubation and ventilator during hospitalization; and (VIII) a history of touching or feeding birds. Ultimately, we collected 150 cases for this study. A flow chart of the study is shown below. All the patients were provided informed consent, and the study was approved by the ethics committee of Zhejiang University Jinhua Hospital (ethics committee approval No. 5, 2013) and was conducted in accordance with the Helsinki Declaration of 1964 (revised 2008).
Baseline value comparison
These patients didn’t have coronary atherosclerotic heart disease, hypertension, diabetes, and some of them combined with pulmonary heart disease. They could not perform lung function test because they were very weak. All of the patients were diagnosed in COPD group C or group D. Table 1 provides general information about the selected patients, including gender, age, weight, BMI, time of using glucocorticoids and the way they were used, and BNP values. The baseline values of the two groups were analyzed by a t-test and chi-squared test, and the differences were not significant.

Full table
Research process
All patients in both groups were informed to specify the diagnosis of AECOPD groups C and D. General anti-infection, airway inflammation resistance, and other nutritional support treatments were given according to established guidelines. The fluid assistance therapy group was given appropriate amounts of intravenous fluids based on body weight, heart function, appetite, and the differential value of liquid between intake and discharge within 24 hours. The basic principle to follow was to add an appropriate amount of intravenous fluids in case of cardiac function tolerance while trying to avoid the occurrence of limb edema or heart failure.
We repeatedly sampled qualified sputum specimens during hospitalization for detection. Alveolar lavage fluid was collected for examination from some patients who could tolerate a bronchoscope. In addition, a few patients underwent pathologic examination by percutaneous lung targeted biopsy if they could tolerate puncture.
The diagnosis of pulmonary fungal infection was carried out according to the current expert consensus (2): (I) the clinical symptoms became worse despite general anti-infection treatments, and there were new lesions on imaging that could not be explained by general bacterial infections; (II) the patient had qualified sputum culture specimen results that suggested fungal infection more than 3 times; (III) percutaneous lung biopsy suggested fungal infection, or bronchoalveolar lavage fluid suggested fungal infection. In this study, the diagnosis of pulmonary fungal infection required the first item to be met, as well as either the second or the third item. Once a diagnosis of pulmonary fungal infection had been made, a corresponding antifungal treatment was given in a timely manner.
Data about fluid assistance therapy, glucocorticoid use, long-term smoking history, broad-spectrum anti-infection drug use, hypoalbuminemia were collected for logistic regression model analysis.
Statistical analysis
After removing the missing data of 59 patients, the rest of 150 cases’ data were ultimately brought into statistical analysis .We collected fungal infection cases in both groups and used the statistical software SPSS 19.0 for chi-squared analysis to compare the rates of fungal infections. The continuous variables were tested for normality before statistical analysis (10). In addition, binary classification logistic regression model analysis was carried out to determine the influence on pulmonary fungal infection by factors such as the fluid assistance therapy, glucocorticoid use, long-term smoking history, broad-spectrum anti-infection drug use, and hypoalbuminemia. And the logistic regression model was built follow the strategy of reference (11). P<0.05 was considered to indicate statistical significance.
Results
One hundred fifty patients were finally brought into statistical analysis. The probability of pulmonary fungal infection of AECOPD patients in the fluid assistance therapy group was significantly lower than in the non- therapy group (P=0.022), as shown in Table 2.

Full table
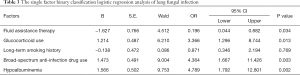
The single-factor unconditional binary classification logistic regression analysis showed that the fluid assistance therapy might be a protective factor against pulmonary fungal infection in AECOPD patients (OR 0.196, 95% CI: 0.044–0.882, P=0.034), shown in Table 3. The results also showed that except for long-term smoking history, the rest of the various factors were associated with statistically significant differences in the occurrence of pulmonary fungal infection (OR 3.366, 95% CI: 1.296–8.744, P=0.013; OR 0.871, 95% CI: 0.346–2.194, P=0.769; OR 4.364, 95% CI: 1.667–11.426, P=0.003, and OR 4.789, 95% CI: 1.792–12.801, P=0.002).
Full table
Considering that the sample size of this study was not big, all of the factors were also entered into a multi-factor two-category logistic regression analysis. The results showed that long-term smoking history was not a risk factor for pulmonary fungal infection in AECOPD patients (P=0.124). Fluid assistance therapy (P=0.001, OR 0.035, 95% CI: 0.005–0.246) was again a protective factor against lung fungal infection for patients with AECOPD. Furthermore, glucocorticoid use, broad-spectrum anti-infection drug use, and hypoalbuminemia were all risk factors (P=0.008, OR 4.992, 95% CI: 1.516–16.438; P=0.001, OR 7.693, 95% CI: 2.221–26.650; P=0.003, OR 6.393, 95% CI: 1.901–21.498). The results are shown in Table 4.

Full table
Discussion
Fungal pathogens are conditional pathogens that are widely distributed in nature. They have low toxicity and generally do not affect normal people. Pulmonary fungal infection is one of the most important types of deep fungus infection. It always occurs in patients with compromised immunity, especially in AECOPD group C and group D patients who have long-term chronic hypoxia, which results in low immunity, significantly impaired defenses, and opportunities for fungal infection. One research indicated that the percentage of patients developing aspergillosis was significantly higher among GOLD IV patients than among GOLD II–III patients (P=0.032). And it found a high percentage (25.5%) of cases progressing to aspergillosis among Aspergillus-colonized non-transplant, non-neutropenic patients, especially among patients with COPD, stressed the importance of colonization as risk factor (12).
The results of this study show that fluid assistance therapy can significantly reduce the probability of pulmonary fungus infection in AECOPD groups C and D patients during hospitalization. The analysis also confirmed various risk factors. The mechanism of how fluid assistance therapy reduces the incidence of pulmonary fungus infection is yet unclear. We hypothesize several potential reasons: (I) the fluid assistance therapy can maintain stable vital signs, particularly blood pressure, as well maintain a steady internal environment to support basic biological functions; (II) it can promote metabolism to a certain extent and improve the body's immune function; (III) it can effectively increase the circulation capacity, which can allow anti-infection drugs to reach their target locations and fulfill their proper roles.
This study was a small-sample, single-center study, but the results may lead to more favorable clinical treatment strategies for patients with AECOPD. However, a larger multicenter study is still needed to confirm the conclusions. In addition, the limitation can be easily found the diagnosis of pulmonary fungal infection in this study was based on the expert consensus, rather than the gold standard of pathological diagnosis. Fluid assistance therapy could provide help in the clinical treatment of AECOPD, reduce fungal lung infection, reduce suffering, shorten hospitalization times, and reduce social and economic burdens.
In conclusion, fluid assistance therapy can reduce risks of hospital acquired pulmonary fungal infection for AECOPD patients. This study is a small sample, single center study, although the results may bring more favorable clinical treatment strategy in patients with AECOPD. But larger, multicenter study is urgently needed. If multicenter study also confirms the conclusion, the fluid assistance therapy will provide strong help for the clinical treatment of AECOPD, it can reduce the lung fungal infection, can reduce AECOPD patients’ suffering, shorten the hospitalization of patients, and can reduce the family and the social economic burden.
Acknowledgements
We thank Dr. Song-Min Ying for his checking the manuscript and advice. We also thank all the members in the study for earnest work.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: All the patients were provided informed consent, and the study was approved by the ethics committee of Zhejiang University Jinhua Hospital (ethics committee approval No. 5, 2013) and was conducted in accordance with the Helsinki Declaration of 1964 (revised 2008).
References
- Taccone FS, Van den Abeele AM, Bulpa P, et al. Epidemiology of invasive aspergillosis in critically ill patients: clinical presentation, underlying conditions, and outcomes. Crit Care 2015;19:7. [Crossref] [PubMed]
- The Infection Committee, Respiratory Society, Chinese Medical Association. Expert consensus of diagnosis and treatment for pulmonary fungal disease. Chin J Tuberc Respir Dis 2007;30:821-34.
- Shelton BK. Opportunistic fungal infections in the critically ill. Crit Care Nurs Clin North Am 2000;12:323-40. [PubMed]
- Higa M. Clinical epidemiology of fungal infection in diabetes. Nihon Rinsho 2008;66:2239-44. [PubMed]
- Jham BC, França EC, Oliveira RR, et al. Candida oral colonization and infection in Brazilian patients undergoing head and neck radiotherapy: a pilot study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2007;103:355-8. [Crossref] [PubMed]
- Kontoyiannis DP, Marr KA, Park BJ, et al. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001-2006: overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) Database. Clin Infect Dis 2010;50:1091-100. [Crossref] [PubMed]
- Nivoix Y, Velten M, Letscher-Bru V, et al. Factors associated with overall and attributable mortality in invasive aspergillosis. Clin Infect Dis 2008;47:1176-84. [Crossref] [PubMed]
- Ostrosky-Zeichner L, Pappas PG. Invasive candidiasis in the intensive care unit. Crit Care Med 2006;34:857-63. [Crossref] [PubMed]
- Vandewoude K, Blot S, Benoit D, et al. Invasive aspergillosis in critically ill patients: analysis of risk factors for acquisition and mortality. Acta Clin Belg 2004;59:251-7. [Crossref] [PubMed]
- Zhang Z. Univariate description and bivariate statistical inference: the first step delving into data. Ann Transl Med 2016;4:91. [Crossref] [PubMed]
- Zhang Z. Model building strategy for logistic regression: purposeful selection. Ann Transl Med 2016;4:111. [Crossref] [PubMed]
- Barberán J, García-Pérez FJ, Villena V, et al. Development of Aspergillosis in a cohort of non-neutropenic, non-transplant patients colonised by Aspergillus spp. BMC Infect Dis 2017;17:34. [Crossref] [PubMed]
Cite this article as: Chen W, Zhu D, Chen H, Luo J. Fluid assistance therapy reduces risks of hospital-acquired pulmonary fungal infection for acute exacerbation of chronic obstructive pulmonary disease patients. J Emerg Crit Care Med 2018;2:52.


