Integration of bedside ultrasound into the ICU—a review of indications, techniques and interventions
Introduction
Ultrasonography is a safe and cost-effective form of imaging that has been used over the last half century to diagnose and care for patients. Recently in the past few decades the technology has improved and allowed the physician to perform point of care ultrasonography—in part due to more compact, higher quality and less expensive equipment. Ultrasound (US) is a frequency of sound higher than humans can hear at more than 20,000 Hz, diagnostic US is conducted in the millions of Hertz (MHz) range (1). Lower frequency US has better penetration, with lower resolution, while the higher frequency provides better images of more superficial structures. US penetrate fluid and solid organs (e.g., liver, spleen, kidney, heart) but have poor penetration through bone or air, limiting its usefulness in some areas of the body. The point of care US (POCUS) allows real-time dynamic images to be obtained by the physician responsible for interpretation and clinical decision making, based on presenting signs and symptoms. Techniques are easily reproducible if the patient condition changes and has a wide scope of uses divided into diagnostic and procedural categories. Critical care units and emergency departments, both frequently requiring immediate feedback, have seen the most use of POCUS. However basic skills in POCUS are now being incorporated into many teaching programs at both the undergraduate and graduate medical education level. In response to the 1999 Institute of Medicine report To Err is Human, the use of real-time US guidance during central line insertion has become standard of care (1,2). This is a review of the current literature regarding the indications and techniques used in the intensive care units (ICUs). The bulk of the evidence and data is derived from procedural guidance for central lines and diagnostics based on transthoracic echocardiograph. Newer applications include ocular scans, lung and even muscle thickness for nutritional status.
Methods
A literature review was performed using initial search key words “intensive care”, “point of care” and “ultrasound”. In order to increase sensitivity, synonyms for each keyword were harvested from MeSH and Emtree entries. To increase specificity, search strings were modified to only return studies that include particular terms in the title or abstract (Table 1). Since access to the Embase database was gained during the initial review of citations, the Embase search strategy includes updated exclusion criteria i.e., the Embase search results that mention pediatric or neonates in the title or abstract results are excluded. Database results were filtered to include only English articles published between January 2012 and July 2017. After deduplication, results from Cochrane, Embase, PubMed, and Scopus revealed 653 unique titles. Titles were then sorted based on the adjusted inclusion criteria of studies on adult USs in the intensive care setting or POCUSs reducing the number of articles for review to 559. The articles were included in a single database and reviewed by all contributing authors. The articles were divided by type of category including those used in ICU or emergency departments, excluding those that included pediatrics or neonatal units.

Full table
Diagnostic techniques
Cardiovascular/volume status
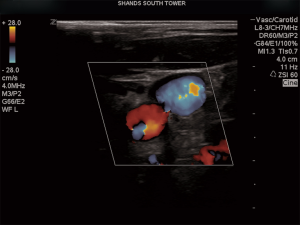
Echocardiography is a, noninvasive, inexpensive and easily accessible, ultrasonography method to obtain detailed information regarding the cardiovascular system in the ICU (3). Of the diagnoses that echocardiography can provide, its ability to identify life threatening processes such as pericardial tamponade and subtypes of aortic dissection are invaluable (4). Cardiac findings include regional wall motion abnormalities, poorly contractile myocardium, low ejection fraction, dilated cardiac chambers, valvular stenosis, regurgitations and “B-lines” (5). Use of ultrasonography in predicting fluid responsiveness can include two-dimensional, motion or Doppler modes of imaging to assess both blood flow and velocity variations throughout a respiratory cycle (Figure 1) (5).
In addition to diagnostic capabilities, echocardiography also provides qualitative and quantitative estimation of ventricular volumes and cardiac output through Doppler technology, volumetric measurements, and derived applications (6). Courses including the Intensive Care Echocardiography certification help educate clinicians to evaluate gross ventricular function, presence of pericardial effusion, inferior vena cava (IVC) size, valve thickening or unusual morphological appearance. Diagnostics are important since the treatment may differ depending on the type of pathology. For instance, the treatment options for aortic dissection differ depending on involvement of ascending or descending aorta, identification is paramount for therapy. Transesophageal echocardiogram has sensitivity, specificity, and negative predictive value of near 100% while CT scans have been shown to have a sensitivity of 90–100% and specificity of 87–100% (4). US does not have the radiation exposure or intravenous contrast that may lead to nephropathy in the critically ill patient. US is fast, provide almost no risk to the patient and can be cost effective for timely diagnosis and early treatment.
Use of basic bedside echocardiography has changed management for up to 50% of patients, in a study over 6 months, including adjustments in fluid administration, inotrope or vasopressor initiation, or identifying previously unknown, underlying disease such as pulmonary hypertension (4). Contrast echocardiography is a technique for improving echocardiographic resolution and providing real time assessment of intra-cardiac blood flow. Agitated saline contrast provides contrast in the right heart and enables detection of right to left shunts. Opacification of the left ventricular (LV) cavity by contrast agents developed to traverse the pulmonary vasculature permits improved endocardial border detection. Contrast echocardiography can also enhance delineation of Doppler signal. Additional uses of contrast echocardiography are to assess myocardial perfusion and viability. Contrast echocardiography, with improved image quality and detection of endocardial borders, has been employed to improve assessment of LV function, detection of LV thrombi, discrimination of cardiac structures, and decreased the need for additional imaging in 18% of patients (3). Contrast agents, agitated saline, increase the lesion-to-parenchyma contrast gradient allowing for increased definition of shapes and sizes and allows detection of relevant information unrecognized by baseline sonography or color Doppler imaging (7).
Intravascular volume assessment is a common entity managed by critical care physicians. It has traditionally been evaluated by and for the use of invasive procedures such as central line placement for central venous pressure (CVP) evaluation. However, invasive procedures have complications: pneumothorax, hemothorax, infection, air embolism, or inadvertent arterial catheterization (8-10). IVC diameter and collapsibility measurement via US is a non-invasive procedure that can be used to assess intravascular volume (11,12). IVC collapsibility infers fluid responsiveness and a hypovolemic state, similar to how CVP is used to estimate a patient’s response to fluid challenge. Therefore, using IVC collapsibility index is another noninvasive method to measure fluid status in real-time at the bedside.
Procedural technique
To highlight the ease and benefits of electrocardiography over more invasive procedures, we briefly describe the technique of evaluating IVC using US. IVC diameter and collapsibility, is easily reproducible at the bedside (1). With the patient in supine position, a cardiac US probe is placed below the xiphoid in a transverse, short axis position. The probe is placed in the center of the sub-xiphoid region and slowly moved to the right until the IVC is in the center of the screen. Once centered, the probe is rotated 90 degrees clockwise to place the IVC in the longitudinal view. The probe is then migrated cephalad and caudal to identify the atrial-cava junction and the entrance of the hepatic veins into the IVC, respectively (13). IVC diameter and collapsibility may be measured at the atrial-cava junction or 1–2 cm above the entrance of the hepatic veins into the IVC, with the atrial-cava junction being more influenced by diaphragm contractility (13,14). At least three measurements must be obtained with two of them being at the end of inspiration and at the end of expiration, in order to be able to calculate the IVC collapsibility index (13,15). The IVC collapsibility index is calculated by subtracting the higher diameter (D1) of the end of inspiration and at the end of expiration diameters by the lower diameter (D2), dividing it by the higher diameter (D1), and multiplying it by 100, [(D1−D2)/D1]×100 (15,16). The IVC collapsibility index is inversely related to the CVP and therefore a high IVC collapsibility index, greater than 60%, indicates hypovolemia and a low IVC collapsibility index, less than 20%, indicates the patient may be euvolemic or hypervolemic (16-19). There are a few other techniques for volume status not discussed in this review, related to transthoracic echocardiography or transesophageal echocardiography, other types of diagnostics and therapies are reviewed.
Thoracic cavity
The use of bedside US to diagnose and treat thoracic pathology is well recognized and accepted. US of the lung and pleura can be used to diagnose lung pathology including but not limited to pleural effusions, pneumothorax, pulmonary edema, pleuritis, pneumonia, and peripheral lobar infarction (20,21). A high-frequency (7–11 Mhz) linear probe will offer optimum resolution while a lower-frequency probe can be used to identify deeper structures (21). A complete examination involves twelve imaging regions divided over the anterior, lateral and posterior zones of each hemithorax divided by the anterior and posterior axillary line or eight imaging regions (21). A more rapid two-region scan can be used to rule out interstitial syndrome due to cardiogenic acute pulmonary edema in the critically ill patient, but may not be sufficient in patients who are not severely dyspneic (20). Features to consider during thoracic cavity ultrasonography are detailed below.
Lung sliding, or the sliding of the visceral pleura relative to the parietal pleura and chest wall is a normal finding. Since the US beam does not propagate through pleural air, absence of lung sliding indicates separation of the visceral and parietal pleura most consistent with pneumothorax. Absence of lung sliding, comet tail artifacts and a lung point sign indicate a pneumothorax. The lung point is defined as a transition point between the typical sonographic pattern of pneumothorax into the normal pattern of lung sliding (20).
In patients with acute dyspnea, US can identify diffuse signs of interstitial and alveolar edema. These findings follow a regular and symmetric spatial distribution and progress from lateral to the inferior and superior, anterior regions of the pulmonary surface (20). Sonographic “A lines” are visualized as hyperechoic lines perpendicular to the US beam within the lung parenchyma. This sonographic finding represents reverberation artifacts that are a normal finding and are absent in setting of pulmonary edema (21). Patients with interstitial lung disease such as pulmonary fibrosis most commonly have disease findings beginning in the posterior chest. This region can be evaluated to identify the “curtain sign,” which is described as the relative movement of the lung covering the sub-diaphragmatic organ during inspiration.
In addition, US can be used as an adjunct in detecting ventilator-associated pneumonia especially when correlated with portable chest radiographs that may be inconclusive (22). Findings suggestive of pneumonia include: subpleural consolidations, lobar/hemilobar consolidation, dynamic linear/arborescent air bronchogram with consolidations, and hyperechoic images that move with inspiration (22).
Procedural technique
Diaphragmatic sonography provides an assessment of diaphragmatic movement, including amplitude, force, and velocity of contraction. In addition, it can be helpful in providing accurate assessment and long-term evaluation of patients with diaphragmatic dysfunction as in the case of patients who required prolonged mechanical ventilation (23). This is performed using a 3.5–5 MHz phased array probe, placed below the costal margin in the mid-clavicular line or in the anterior axillary line on the area of interest. Normal inspiratory movement is caudal and expiratory movement is cranial. These movements can be measured in a mechanically ventilated patient. The average displacement of the muscle during respiration of 12±7 mm (range, 7–28 mm) and 10±8 mm (range, 5–7 mm), respectively. Additionally, pleural effusions, consolidation or atelectasis can also be detected, allowing for change of treatment or intervention (23). The use of US can aid in determination of readiness for ventilator liberation based on diaphragm motion, presence of pulmonary edema or pneumonia.
Abdomen
In the trauma and critical care setting, use of US to conduct a focused assessment with sonography for trauma (FAST) exam, as defined by the Advanced Trauma and Life Support course, to diagnose and treat pathology within the abdominal cavity is well documented in the literature. Intra-abdominal fluid, obstructive changes of the kidney and bladder, biliary disease, and intra-abdominal catastrophes using four acoustic windows (right upper quadrant, left upper quadrant, and suprapubic regions as well as a subxiphoid view of the pericardium) are easily detectable on abdominal US (24). The FAST exam consists of four views looking for dependent fluid in the peritoneal cavity (Figure 2), with additional views of the thoracic cavity to determine presence of pneumothorax; this is now known as the extended FAST (e-FAST).
Procedural technique
The FAST exam utilizes preset abdominal US settings on the US. The xiphoid process is used as an external landmark to obtain two upper quadrant views. The probe can be placed where the subxiphoid line intersects the right and left mid-axillary lines and aimed posteriorly. On the right side, this allows for a coronal view of the liver, right kidney, Morison’s pouch, and the diaphragm. The lower pole of the kidney can be visualized by scanning a rib space or two inferiorly. On the left side, the probe is placed along the subxiphoid line and the posterior axillary line with the probe facing cranially and posteriorly. This allows a coronal view of the spleen, left kidney, and diaphragm. By “fanning” anteriorly and posteriorly, one can visualize fluid in the subphrenic region and lateral to the spleen (24). The pelvic view is obtained by placing the probe in the midline superior to the pubic symphysis on both long and short axis views. The gain may need to be decreased to compensate for the bladder’s increased transmission of US. The depth should be adjusted so the bladder occupies less than half the screen in order to identify free fluid deep than the bladder and uterus in women. The subxiphoid, coronal view of the heart can be obtained using abdominal presets by placing the probe under the xiphoid process and angled cranially under the sternum. The echogenic pericardium and less echogenic myocardium contrasts with hypoechoic pericardial fluid. Imaging can be improved by having the patient take deep breaths, sliding the probe to the patient’s right, or bending the patient’s leg to relax the abdominal wall (24).
Endotracheal intubation and tracheostomy
Ultrasonography has utility in guiding clinical decision-making and intervention for airway management and for assessing pathology of the upper and lower airways. The use of US to assess successful endotracheal intubation has been performed since 1987 when Raphael and Conrad first described the procedure (25). A standard high frequency probe (6–13 MHz) is sufficient for most airway sonography, with a low-frequency probe (2–5 MHz) being best to evaluate the floor of mouth, base of tongue, and prandial status (26). Placement of the US transducer transversely over the trachea during intubation resulted in more successful guide-wire placement compared to longitudinal placement (26). US can be used preoperatively in screening and predicting difficult laryngoscopy and diagnosing pathology that can affect airway management, such as visualizing vocal cords and larynx (26,27).
In the intensive care setting, it is especially useful in identifying and quantifying stomach contents immediately prior to endotracheal intubation (26). Accurate localization of the trachea, especially in patients with obesity, thick necks, or distorted anatomy and masses; is also made possible with US visualization (26). Intubated patients in the ICU have an incidence of laryngeal edema (LE) between 3–30%, a complication that can lead to reintubation and is associated with 17.4% increased hospital mortality (27). Early detection of LE allows for effective treatment. The standard method for evaluation of LE is a cuff leak test, but its accuracy has been called into question, as it depends on many expiratory factors, including exhaled tidal volume. US can more accurately provide air column width difference (ACWD) measurement of vocal cords, measured before and after endotracheal tube deflation, and is more practical as it does not require mechanical ventilation to obtain (27). A significantly higher ACWD (above 1.6 mm) has been observed in the non-LE group compared to those with LE (27). This cutoff can be used to identify patients who can be safely extubated.
For tracheostomy and percutaneous dilatational tracheostomy (PDT), US provides real-time tracheal localization, visualization of the anterior tracheal wall and pre-tracheal tissue (26). It also allows for selection of the tracheal ring interspace for placement of the tracheostomy tube and results in a lower rate of misplacement than occurs with “blind” placement (26). The distance from surface to the tracheal lumen can be measured to help avoid penetrating the posterior tracheal wall and help select optimal length of tracheostomy cannula. Use of US has also been shown to reduce the risk of erosion to major blood vessels, especially the innominate vein and arch of the aorta. Combined with bronchoscopy, US use resulted in a change in planned puncture site in 24% of cases in a prospective series of PDTs (26).
US guided procedures and technique
The use of US is both a diagnostic tool in the hand of the clinician, and a useful adjunct to facilitate procedural performance. Overall US has become standard of care for the placement of central venous catheters, and with increased access, decreased cost and more experienced clinicians the scope of practice is increasing to procedures not routinely associated with US. The next section will review techniques used for placement of drains in the chest, peritoneal cavity and central lines. Additionally, will review techniques related to ocular and muscle US techniques.
Thoracentesis and chest tube insertion
Thoracentesis is a commonly performed procedure to physically remove and alleviate fluid accumulation in the pleural space. Complications include pneumothorax, vasovagal reactions, or rarely re-expansion pulmonary edema, hepatic or splenic puncture, and hematomas (24). The two most common indications for thoracentesis include pleural effusions causing respiratory compromise and empyema. US-guided thoracentesis is usually reserved for the complicated, mechanically vented, obese patient or those with small fluid collections.
Procedural technique
Ideally, the patient will be sitting, with arms elevated and stabilized behind the head. The chest is prepared using standard sterile technique. Alternatively, the lateral decubitus position with the affected side up can be utilized in the critical care setting.
US is used to localize the fluid collection, the liver, and the spleen; limiting the possibility of puncturing these organs. Lower frequency vector probes are used initially to survey the thorax as a whole before narrowing findings with a high frequency linear probe. The fluid collection should be marked on the body, so the area can then be locally anesthetized penetrating all layers between the skin and pleural space. A 19-gauge sheath needle is then advanced under direct visualization while aspirating the fluid. A larger 6- to 8-Fr catheter can then be advanced for drainage of fluid accumulations.
For large effusions, the Seldinger technique is preferred for placement of a 10-Fr or larger catheter using the lateral approach. More complicated fluid collections, such as empyema, may form in the setting of Para pneumonic effusions; early phase may be amenable to drainage, however, late stage effusions can become organized and often require surgical decortication (24). Since an empyema is frequently loculated, successful drainage usually requires frequent catheter manipulation facilitated by fibrinolytic instillation.
Vascular access
The use of sonographic US for central venous cannulation has become the standard of care since its introduction in 1984 (24,28,29). Recommended for placement of internal jugular vein (IJV) cannulation, based on level 1 evidence, by the American Society of Echocardiography, Society of Cardiovascular Anesthesiologists, and The National Institute for Clinical Excellence (30-32). US guided central line placement is the most common US based procedure performed in the ICU.
The procedure is performed under direct visualization using a high frequency linear array probe and it may be performed using the one or two-person technique. When performed by a single operator, the US probe is held with the non-dominant hand as the introducer needle is advanced under direct US-guided visualization (33). If an assistant is available, a second person may stabilize the US probe while the operator cannulates the venous system under direct visualization (33). The short axis on the US probe provides the safest view for IJV central line placement (33,34).
Procedural technique
The patient is placed in Trendelenburg position, and the neck is prepped from the tragus down to the ipsilateral nipple and draped in standard surgical fashion. An US probe, using a sterile covered appliance, is used to identify the IJV. The IJV is differentiated from the carotid arterial system by its compressive characteristic and lack of pulsatile flow (Figure 3). The skin and subcutaneous tissues are anesthetized and the introducer needle is advanced into the vein under direct visualization.
US guidance improves the success of central line placement, reduces the number of attempts before successful cannulation and lessens the number of first attempt failures (35-38). In addition, US guided IJV reduces the incidence of post-procedural complications such as hematomas, pneumothorax, hemothorax, inadvertent carotid cannulation, and central line infections (33,35,36,38). US guided vein cannulation has been shown to improve line placement success rate and reduce the number of complications when compared to the landmark method, especially in the hands of beginner/inexperienced operators (39-43). Based on a recent Cochrane Database systematic review that looked at US guided IJV versus anatomical landmarks, in over 700 citations over 47 years of publications, the usage of US guided IJV placement reduces the overall rate of complications by 71%, the arterial puncture rate by 72% and the overall rate of hematoma creation (44). In addition, the review displayed that the use of US increased the first stick success rate by 57% and the time to successful cannulation was decreased by 30.52 seconds (44).
The data is not as clear regarding the use of US for cannulation of the subclavian or axillary vein and thus the value of its use in clinical practice is unclear (39). Additionally, the data regarding US guided arterial catheterization is limited and currently without level 1 recommendations (45). However, evidence suggests that US guided radial arterial catheterization, when compared to blind palpation technique increases the first-attempt success rate, decreases the number of attempts, reduces the insertion time, and improves the overall success rate (46-49). Thus, overall evidence suggests that use of US guidance for catheterization of multiple veins for vascular access improves success rates, decreases insertion time and results in decreased complications.
Intra-abdominal fluid & paracentesis
Abscesses and other peritoneal fluid collections are amongst the most common use of interventional US in the critical care setting (24). Ascites is a common sequela of patients with a vast gradient of liver disease ranging from malnutrition with hypoalbuminemia to fulminant liver failure secondary to cirrhosis. Paracentesis is a widely-accepted modality to provide both diagnostic information and allow therapeutic intervention.
Paracentesis is not considered a critical procedure by most clinicians (24), however Patel et al. argue that the use of abdominal US supports a decrease in incidence of complications and overall cost (50). US provides the operator direct visualization to facilitate accuracy and precision thereby dramatically reducing the risk of injury to intra-peritoneal structures such as the bowel or vasculature. In a randomized control trial, Nazeer et al. reported that up to 25% of patients with clinical suspicion for ascites who underwent diagnostic abdominal US were spared an unnecessary procedure. Furthermore, up to 76% of patient who initially had a failed paracentesis performed by anatomical landmarks were successfully aspirated using US guided technique (24). Thus, US guidance for paracentesis can not only decrease unnecessary use of invasive procedures, but can also improve the success of the procedure and decrease risk of complications.
Procedural technique
Initial scanning of the peritoneal cavity is completed using a standard-frequency curvilinear probe with either a 3.5 or 5 MHz radiofrequency. Low-gain, high frequency transducers can be used to focus the field of view to the region of interest (24).
Before introduction of any needle, the operator must be clear that there are no obstructive vascular or other intra-peritoneal structures between their instrument and the fluid of interest. Even before fluid aspiration, US is helpful in preemptively differentiating exudative versus transudate ascites, as the latter appears anechoic. Exudative processes are often loculated may include septa or inter-loop fluid collections representing abscesses formation.
Optimal anatomical locations for paracentesis are 2 cm, midline inferior to the umbilicus. Alternative entry points include 5 cm superior and medial to the anterior superior iliac spines. Care should be taken to avoid injury to the inferior epigastric arteries as they traverse the rectus sheath longitudinally 4–8 cm lateral to the midline (24).
Paracentesis is performed with standard preparation of the skin. A needle or Seldinger technique (for catheter insertion) can then be utilized to gain access into the fluid collection. Complications are rare but may include bowel perforation, hemorrhage, or hypotension.
Nutritional evaluation & ventilator weaning
Weakness and deconditioning are adverse effects resulting from prolonged hospitalizations. Muscular wasting from prolonged ICU stays have long term catabolic effects that persist up to one year after discharge (51,52). Early nutritional support has been repeatedly demonstrated as one of the most important elements to prevent muscle wasting.
In recent years, body composition measurements have been proposed as essential tools to evaluate nutritional status in the ICU (52,53). Objective skeletal muscle measurements using US is a modern and innovative proposed method. US has been shown to be a reliable and accurate tool in assessing change in muscle mass secondary to catabolic effects of trauma, surgery, or critical illness (54). Lower extremity muscle groups are more likely to undergo atrophy in the first five days of an ICU admission when compared to their upper extremity muscle counterpart (52,55).
Procedural technique
Martin et al. describes the quadriceps femoris as the muscle group of choice. With the patient in supine position and the hip and knee in a flexed, relaxed position, a measuring tape is used to mark the origin and insertion points of the rectus femoris from the anterior inferior iliac spine to the patella, respectively.
Using a marking pen, the midpoint and the distal one third are marked between the origin and insertion of the muscle. The midpoint of the rectus femoris is the thickest portion of the muscle. The one third point is used, as the entire circumference of the rectus can be visualized at this point. A linear US probe is then used to evaluate the rectus femoris in a transverse fashion to provide a cross-sectional view of the shortest axis of the muscle. Once the rectus femoris is evaluated, additional structures including the skin, subcutaneous tissue, muscular fascia, rectus femoris, vastus intermedius, and the femur are identified (52). This technique is not routinely used, but is starting to gain more prevalence in academic research centers and may change approach to nutritional status and fragility with an objective measure.
Neurological procedures
The use of US in the field of neurology first described by Karl Dussik in 1942, the father of modern US and first physician to use US in the medical field, at the University of Vienna to diagnose brain tumors (56). US use in the field of neurology includes: B-mode sonography to monitor hydrocephalus and intracranial hemorrhage, transcranial Doppler US for diagnosing brain death, and transcranial color-coded sonography to monitor cerebral autoregulation and intracranial pressure (54,57,58).
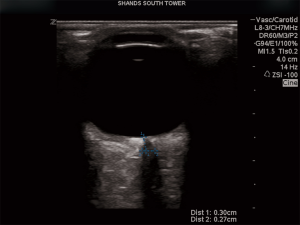
A commonly encountered issue in neurological and trauma critical care units is raised intracranial pressure (ICP). Invasive monitoring techniques are considered the gold standard for monitoring ICP (59). However, invasive procedures have risk and thus non-invasive procedures such as CT scanning and the use of US have been advocated to avoid more invasive procedures. A technique first described by Helmke and Hansen in 1997, is the measurement of the optic nerve to estimate ICP, based on the concept that the optic nerve is an extension of the central nervous system (60,61). The optic nerve is surrounded by all three layers of the meninges and cerebrospinal fluid (CSF) flows freely between the intracranial and intra-orbital subarachnoid space (62-64). Thus, the width of the optic nerve as viewed by US, can serve as a non-invasive means to assess intracranial pressure.
Procedural technique
As described by Kazdal et al., the patient should be in supine position with their eyes closed. A thick layer of US gel is placed over the upper eyelids. A 7.5 MHz US probe is then placed on the superior and lateral aspect of the orbit with the probe angled caudally and medially (60). The nerve is identified as a hypoechoic structure just posterior to the globe. Measurements should be taken 3mm behind the globe to produce optimally reproducible results with the greatest contrast (59,60). Once the nerve is identified, the optic nerve sheath diameter (ONSD) is measured (Figure 4). A “normal” ONSD on ocular US is <5 mm for adults, <4.5 mm for children aged 1–15 years old, and <4 mm for infants.67 Values above the stated ONSD are considered abnormal. The sensitivity and specificity of ocular US and abnormal ONSD based on multiple studies are 77–100% and 63–100%, respectively (59,65-68).
Conclusions
The use of US has been shown to decrease medical errors, provide more efficient real time diagnosis and allow for more widespread, less expensive interventions for defined indications. As POCUS becomes more common in the inpatient setting, including the ICU and emergency departments there is also a growing need for evidence-based guidelines, as well as a need to ensure competency, define its benefits and limit any unnecessary imaging and consequences. The technology is growing and new areas of use are constantly being developed. The increasing use of resource limited settings by patients, may allow for US to meet almost two thirds of the imaging needs in developing countries, has even been used at Mount Everest base camp to diagnose high altitude pulmonary edema (1). It may even become cost effective in a reimbursement scheme that is based on “bundling”, in some cases may obviate the need for more resource intensive imaging with a consulting radiologist.
The indications are present for the use of US as a diagnostic modality, assistance for guidance for intervention. The key is the next step, this has to focus on education and how to incorporate this modality in the daily care of patients to improve quality care and reduce the cost of healthcare. One of the new and successful models has a great method for teaching skills to the new generation of physicians, incorporates online and individual learning with hands on skills in real time settings (69). Innovative approaches that promote educational efficiency and effectiveness are needed to meet the challenges in educating the trainees, using interactive hands on experience in group settings is shown to be successful especially with the newer generations.
Acknowledgements
We would like to thank Scott Norberg for providing the ultrasound images for this publication.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Moore CL, Copel JA. Point-of-Care Ultrasonography. N Engl J Med 2011;364:749-57. [Crossref] [PubMed]
- Arntfield RT, Millington SJ. Point of care cardiac ultrasound applications in the emergency department and intensive care unit - a review. Curr Cardiol Rev 2012;8:98-108. [Crossref] [PubMed]
- Hwang HJ, Sohn IS, Kim WS, et al. The Clinical Impact of Bedside Contrast Echocardiography in Intensive Care Settings: A Korean Multicenter Study. Korean Circ J 2015;45:486-91. [Crossref] [PubMed]
- Gray R, Baldwin F, Bruemmer-Smith S. Diagnostic echocardiography in an unstable intensive care patient. Echo Res Pract 2015;2:K11-6. [Crossref] [PubMed]
- Laher AE, Watermeyer MJ, Buchanan SK, et al. A review of hemodynamic monitoring techniques, methods and devices for the emergency physician. Am J Emerg Med 2017;35:1335-47. [Crossref] [PubMed]
- Avallato C, Nicoletti I, Locatelli A. General hemodynamic assessment. In: Echocardiography for Intensivists; 2012:235-43.
- Calabrese E, Catalano O, Nunziata A, et al. Bedside contrast-enhanced sonography of critically ill patients. J Ultrasound Med 2014;33:1685-93. [Crossref] [PubMed]
- Brass P, Hellmich M, Kolodziej L, et al. Ultrasound guidance versus anatomical landmarks for internal jugular vein catheterization. In: Brass P, editor. Cochrane Database of Systematic Reviews. Vol 1. Chichester, UK: John Wiley & Sons, Ltd; 2015:CD006962.
- Merrer J, De Jonghe B, Golliot F, et al. Complications of femoral and subclavian venous catheterization in critically ill patients: a randomized controlled trial. JAMA 2001;286:700-7. [Crossref] [PubMed]
- Sznajder JI, Zveibil FR, Bitterman H, et al. Central vein catheterization. Failure and complication rates by three percutaneous approaches. Arch Intern Med. 1986;146:259-61. [Crossref] [PubMed]
- Zhang Z, Xu X, Ye S, et al. Ultrasonographic Measurement of the Respiratory Variation in the Inferior Vena Cava Diameter Is Predictive of Fluid Responsiveness in Critically Ill Patients: Systematic Review and Meta-analysis. Ultrasound Med Biol 2014;40:845-53. [Crossref] [PubMed]
- Ferrada P, Anand RJ, Whelan J, et al. Qualitative assessment of the inferior vena cava: useful tool for the evaluation of fluid status in critically ill patients. Am Surg 2012;78:468-70. [PubMed]
- De Backer D, Fagnoul D. Intensive care ultrasound: VI. Fluid responsiveness and shock assessment. Ann Am Thorac Soc 2014;11:129-36. [Crossref] [PubMed]
- Wallace DJ, Allison M, Stone MB. Inferior vena cava percentage collapse during respiration is affected by the sampling location: an ultrasound study in healthy volunteers. Acad Emerg Med 2010;17:96-9. [Crossref] [PubMed]
- Rudski LG, Lai WW, Afilalo J, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr 2010;23:685-713. [Crossref] [PubMed]
- Kent A, Bahner DP, Boulger CT, et al. Sonographic evaluation of intravascular volume status in the surgical intensive care unit: a prospective comparison of subclavian vein and inferior vena cava collapsibility index. J Surg Res 2013;184:561-6. [Crossref] [PubMed]
- Stawicki SP, Braslow BM, Panebianco NL, et al. Intensivist use of hand-carried ultrasonography to measure IVC collapsibility in estimating intravascular volume status: correlations with CVP. J Am Coll Surg 2009;209:55-61. [Crossref] [PubMed]
- Ando Y, Yanagiba S, Asano Y. The inferior vena cava diameter as a marker of dry weight in chronic hemodialyzed patients. Artif Organs 1995;19:1237-42. [Crossref] [PubMed]
- Stawicki SPA, Bahner DP. Evidence tables: Inferior vena cava collapsibility index (IVC-CI). OPUS 12 Scientist 2012;6:3-5.
- Gargani L, Volpicelli G. How I do it: lung ultrasound. Cardiovasc Ultrasound 2014;12:25. [Crossref] [PubMed]
- Doerschug KC, Schmidt GA. Intensive care ultrasound: III. Lung and pleural ultrasound for the intensivist. Ann Am Thorac Soc 2013;10:708-12. [Crossref] [PubMed]
- Lichtenstein DA. Lung ultrasound in the critically ill. Ann Intensive Care 2014;4:1. [Crossref] [PubMed]
- Lichtenstein DA, Menu Y. A bedside ultrasound sign ruling out pneumothorax in the critically ill. Lung sliding. Chest 1995;108:1345-8. [Crossref] [PubMed]
- Chen L, Zhang Z. Bedside ultrasonography for diagnosis of pneumothorax. Quant Imaging Med Surg 2015;5:618-23. [PubMed]
- Lichtenstein D, Mezière G, Biderman P, et al. The lung point: an ultrasound sign specific to pneumothorax. Intensive Care Med 2000;26:1434-40. [Crossref] [PubMed]
- Zhang Z. Double lung point in an 18-month-old child: a case report and literature review. J Thorac Dis 2015;7:E50-3. [PubMed]
- Zhang Z, Chen L. A Physiological Sign That Mimics Lung Point in Critical Care Ultrasonography. Crit Care 2015;19:155. [Crossref] [PubMed]
- Volpicelli G, Audino B. The double lung point: an unusual sonographic sign of juvenile spontaneous pneumothorax. Am J Emerg Med 2011;29:355.e1-355.e2. [PubMed]
- Aspler A, Pivetta E, Stone MB. Double-lung point sign in traumatic pneumothorax. Am J Emerg Med 2014;32:819.e1-2. [Crossref] [PubMed]
- Mongodi S, Via G, Girard M, et al. Lung ultrasound for early diagnosis of ventilator-associated pneumonia. Chest 2016;149:969-80. [Crossref] [PubMed]
- Matamis D, Soilemezi E, Tsagourias M, et al. Sonographic evaluation of the diaphragm in critically ill patients. Technique and clinical applications. Intensive Care Med 2013;39:801-10. [Crossref] [PubMed]
- Ferrari G, De Filippi G, Elia F, et al. Diaphragm ultrasound as a new index of discontinuation from mechanical ventilation. Crit Ultrasound J 2014;6:8. [Crossref] [PubMed]
- Zhou P, Zhang Z, Hong Y, et al. The predictive value of serial changes in diaphragm function during the spontaneous breathing trial for weaning outcome: a study protocol. BMJ Open 2017;7:e015043. [Crossref] [PubMed]
- Boniface KS, Calabrese KY. Intensive care ultrasound: IV. Abdominal ultrasound in critical care. Ann Am Thorac Soc 2013;10:713-24. [Crossref] [PubMed]
- Zhang Z, Hong Y, Liu N, et al. Diagnostic accuracy of contrast enhanced ultrasound in patients with blunt abdominal trauma presenting to the emergency department: a systematic review and meta-analysis. Sci Rep 2017;7:4446. [Crossref] [PubMed]
- Deshpande R, Akhtar S, Haddadin AS. Utility of ultrasound in the ICU. Curr Opin Anaesthesiol 2014;27:123-32. [Crossref] [PubMed]
- Raphael DT, Conard FU. Ultrasound confirmation of endotracheal tube placement. J Clin Ultrasound 1987;15:459-62. [Crossref] [PubMed]
- Teoh WH, Kristensen MS. Utility of ultrasound in airway management. Trends Anaesth Crit Care 2014;4:84-90. [Crossref]
- Sutherasan Y, Theerawit P, Hongphanut T, et al. Predicting laryngeal edema in intubated patients by portable intensive care unit ultrasound. J Crit Care 2013;28:675-80. [Crossref] [PubMed]
- Nicolaou S, Talsky A, Khashoggi K, et al. Ultrasound-guided interventional radiology in critical care. Crit Care Med 2007;35:S186-97. [Crossref] [PubMed]
- Legler D, Nugent M. Doppler localization of the internal jugular vein facilitates central venous cannulation. Anesthesiology 1984;60:481-2. [Crossref] [PubMed]
- Feller-Kopman D. Ultrasound-guided central venous catheter placement: the new standard of care? Crit Care Med 2005;33:1875-7. [Crossref] [PubMed]
- Wigmore TJ, Smythe JF, Hacking MB, et al. Effect of the implementation of NICE guidelines for ultrasound guidance on the complication rates associated with central venous catheter placement in patients presenting for routine surgery in a tertiary referral centre. Br J Anaesth 2007;99:662-5. [Crossref] [PubMed]
- Royse CF, Canty DJ, Faris J, et al. Core review: Physician-performed ultrasound: The time has come for routine use in acute care medicine. Anesth Analg. 2012;115:1007-1028. [Crossref] [PubMed]
- Troianos CA, Hartman GS, Glas KE, et al. Guidelines for performing ultrasound guided vascular cannulation: recommendations of the American Society of Echocardiography and the Society of Cardiovascular Anesthesiologists. J Am Soc Echocardiogr 2011;24:1291-318. [Crossref] [PubMed]
- Chacko J, Brar G. Bedside ultrasonography-Applications in critical care: Part II. Indian J Crit Care Med 2014;18:376-81. [Crossref] [PubMed]
- Blaivas M, Brannam L, Fernandez E. Short-axis versus long-axis approaches for teaching ultrasound-guided vascular access on a new inanimate model. Acad Emerg Med 2003;10:1307-11. [Crossref] [PubMed]
- Karakitsos D, Labropoulos N, De Groot E, et al. Real-time ultrasound-guided catheterisation of the internal jugular vein: a prospective comparison with the landmark technique in critical care patients. Crit Care 2006;10:R162. [Crossref] [PubMed]
- Leung J, Duffy M, Finckh A. Real-time ultrasonographically-guided internal jugular vein catheterization in the emergency department increases success rates and reduces complications: a randomized, prospective study. Ann Emerg Med 2006;48:540-7. [Crossref] [PubMed]
- Sigaut S, Skhiri A, Stany I, et al. Ultrasound guided internal jugular vein access in children and infant: A meta-analysis of published studies. Paediatr Anaesth 2009;19:1199-206. [Crossref] [PubMed]
- Hind D, Calvert N, McWilliams R, et al. Ultrasonic locating devices for central venous cannulation: meta-analysis. BMJ 2003;327:361. [Crossref] [PubMed]
- Frankel HL, Kirkpatrick AW, Elbarbary M, et al. Guidelines for the appropriate use of bedside general and cardiac ultrasonography in the evaluation of critically Ill Patients-Part I: General Ultrasonography. Crit Care Med 2015;43:2479-502. [Crossref] [PubMed]
- Kwon TH, Kim YL, Cho DK. Ultrasound-guided cannulation of the femoral vein for acute haemodialysis access. Nephrol Dial Transplant 1997;12:1009-12. [Crossref] [PubMed]
- Prabhu MV, Juneja D, Gopal PB, et al. Ultrasound-guided femoral dialysis access placement: a single-center randomized trial. Clin J Am Soc Nephrol 2010;5:235-9. [Crossref] [PubMed]
- Hilty WM, Hudson PA, Levitt MA, et al. Real-time ultrasound-guided femoral vein catheterization during cardiopulmonary resuscitation. Ann Emerg Med 1997;29:331-6; discussion 337. [Crossref] [PubMed]
- Aouad MT, Kanazi GE, Abdallah FW, et al. Femoral vein cannulation performed by residents: a comparison between ultrasound-guided and landmark technique in infants and children undergoing cardiac surgery. Anesth Analg 2010;111:724-8. [Crossref] [PubMed]
- Shah PR, Wagner C, Shaw A. Echocardiography and Ultrasound in the Intensive Care Unit. In: Evans AS, Kerr GE, Chung I, et al, editors. Modern Concepts and Practices in Cardiothoracic Critical Care. Hershey, PA: IGI Global; 2015. p. 890-907.
- Sekiguchi H. Tools of the trade: point-of-care ultrasonography as a stethoscope. Semin Respir Crit Care Med 2016;37:68-87. [Crossref] [PubMed]
- Levin PD, Sheinin O, Gozal Y. Use of ultrasound guidance in the insertion of radial artery catheters. Crit Care Med 2003;31:481-4. [Crossref] [PubMed]
- Shiver S, Blaivas M, Lyon M. A prospective comparison of ultrasound-guided and blindly placed radial arterial catheters. Acad Emerg Med 2006;13:1275-9. [Crossref] [PubMed]
- Shiloh AL, Savel RH, Paulin LM, et al. Ultrasound-guided catheterization of the radial artery. Chest 2011;139:524-9. [Crossref] [PubMed]
- Narasimhan M, Koenig SJ, Mayo PH. A whole-body approach to point of care ultrasound. Chest 2016;150:772-6. [Crossref] [PubMed]
- Herridge MS, Cheung AM, Tansey CM, et al. One-Year Outcomes in Survivors of the Acute Respiratory Distress Syndrome. N Engl J Med 2003;348:683-93. [Crossref] [PubMed]
- Galindo Martín CA, Monares Zepeda E, Lescas Méndez OA. Bedside ultrasound measurement of rectus femoris: a tutorial for the nutrition support clinician. J Nutr Metab 2017;2017:2767232.
- Hiesmayr M. Nutrition risk assessment in the ICU. Curr Opin Clin Nutr Metab Care 2012;15:174-80. [Crossref] [PubMed]
- Mourtzakis M, Wischmeyer P. Bedside ultrasound measurement of skeletal muscle. Curr Opin Clin Nutr Metab Care 2014;17:389-95. [Crossref] [PubMed]
- Turton P, Hay R, Taylor J, et al. Human limb skeletal muscle wasting and architectural remodeling during five to ten days intubation and ventilation in critical care - an observational study using ultrasound. BMC Anesthesiol 2016;16:119. [Crossref] [PubMed]
- Gordon D. Ultrasound as a Diagnostic & Surgical Tool: Based on the International Symposium held at the Royal College of Surgeons, London, on December 5th and 6th, 1962. [Internet]. Ann Arbor. Williams and Wilkins; 1964. [cited 2017 Aug 13]. Available online: https://books.google.com/books/about/Ultrasound_as_a_diagnostic_surgical_tool.html?id=-0FrAAAAMAAJ
- Seidel G, Kaps M, Dorndorf W. Transcranial color-coded duplex sonography of intracerebral hematomas in adults. Stroke 1993;24:1519-27. [Crossref] [PubMed]
Cite this article as: Balmert N, Espinosa J, Arafeh MO, Costello J, Markle S. Integration of bedside ultrasound into the ICU—a review of indications, techniques and interventions. J Emerg Crit Care Med 2018;2:17.