
Clinical applications of lactate testing in patients with sepsis and septic shock
Introduction
In 1940s, the entire glycolytic pathway was elucidated. Lactic acid has been known as a product of glycolysis during hypoxia (1). It has been recognized as a metabolite associated with sepsis and with tissue hypoxia for a long time (2). However, a number of studies have suggested that lactate formation during sepsis is due to not only hypoxia but also metabolic processes (3,4). Lactic acidosis results from the accumulation of lactate and protons in the body fluids and is often associated with poor clinical outcomes (5). Moreover, lactate is a parameter of global tissue hypoperfusion and is essential in identifying patients with “cryptic” shock who require focused early goal-directed therapy (EGDT) (6,7). Most of the lactate produced in shock state is due to inadequate oxygen delivery resulting in tissue hypoxia and causing anaerobic glycolysis. Moreover, a hypermetabolic state, with glycolysis enhanced by catecholamines, contributes to the accumulation of lactate (4). Thus, hyperlactatemia and lactic acidosis are common in patients with septic shock and are associated with significant morbidity and mortality (2). As a result, the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) has included hyperlactatemia over 2 mmol/L in the revised definition of septic shock (8). Since three independent international multicenter randomized controlled trials (ProCESS, ARISE, and ProMISe) confirmed that EGDT did not confer a mortality benefit compared with usual resuscitation, the Surviving Sepsis Campaign (SSC) focused on implementing the lactate-guided sepsis management bundle (9-12). The campaign guideline suggests guiding resuscitation to normalize lactate in patients with elevated lactate levels (12). A few recent studies showed that an early lactate clearance strategy and a lactate-guided resuscitation reduced mortality in patients with sepsis and septic shock (7,13-15). This study aimed to review the clinical aspect of lactic acidosis in patients with sepsis and septic shock.
Lactic acid formation and clearance
Lactic acid is normally produced in excess by about 20 mmol/kg/day, which enters the bloodstream (16,17). The tissues that normally produce excess lactic acid include the skin, red cells, brain tissue, muscles, and gastrointestinal tract. During heavy exercise, the skeletal muscles produce most of the excess lactic acid (17). Moreover, the lungs can produce lactate during acute lung injury without tissue hypoxia, and leukocytes generate lactate during phagocytosis or when activated in sepsis (18-20). In pathological conditions in which oxygen delivery is limited, lactate production occurs in other tissues (2).
Lactate is formed from pyruvate in the cytosol as part of glycolysis. Its concentration is in equilibrium with that of pyruvate and is maintained by lactate dehydrogenase (LDH), an enzyme that favors lactate production and normally maintains a constant lactate to pyruvate ratio of approximately 10:1 (21). Therefore, lactate increases when the production of pyruvate exceeds its utilization in the mitochondria. Pyruvate is essentially produced via glycolysis; hence, any increase in glycolysis, regardless of its origin, can increase lactatemia (4). Meanwhile, pyruvate is essentially metabolized to acetyl coenzyme A (acetyl-CoA) by pyruvate dehydrogenase (PDH), which enters the tricarboxylic acid (TCA) cycle under aerobic conditions (2). The TCA cycle also called Krebs cycle. Under anaerobic conditions, the Krebs cycle cannot metabolize pyruvate; thus, pyruvate is shunted toward lactate (Figure 1) (2).
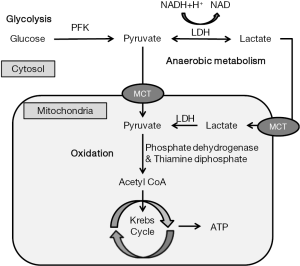
Lactate can be metabolized by the liver and kidneys either by direct oxidation or as a source of glucose (21). Generated lactate can be transformed into oxaloacetate or alanine via the pyruvate pathway or can be utilized directly by periportal hepatocytes (60%) to produce glycogen and glucose (neoglycogenesis and neoglucogenesis; Cori cycle) (4). Furthermore, the kidneys participate in 30% of lactate metabolism, with the cortex classically acting as the metabolizer by neoglucogenesis and the medulla as a producer of lactate (4).
Lactate is not only transformed into glucose via the Cori cycle, it is also removed through oxidation (22). This oxidative compartment which is likely close to the mitochondria is considered responsible for lactate uptake by mono-carboxylate transporter (MCT) into mitochondria and oxidation via pyruvate and the Krebs cycle with adenosine triphosphate (ATP) production (3). This intracellular lactate shuttle balances the lactate level between producing by glycolysis and clearance by oxidation (21) (Figure 1).
The role of lactic acid
In addition of glucose metabolism, lactate plays a crucial role in various functions of the neurologic system, cancer metabolism, in various functions of the immune system, wound healing, and ischemic injuries (1).
Aerobic glycolysis in the brain is very important process in gene expression of neonate (23) and is connected to the development of synapses, neuron projections, and learning (24). Specifically, lactic acid which comes from glycolysis in astrocytes is entered to neurons through MCTs. And it plays a signaling function and stimulation of gene expression, which can lead to a long-term memory formation (25). Moreover, chronic stress is associated with sustained elevation of cyclic adenosine monophosphate (cAMP) and cognitive impairment (26). Lactate could potentially modulate the over-activated signaling cascades by reducing cAMP, thus preventing memory loss and enhancing neuronal protection (1).
Cancer cells, especially rapid growing type, are known to use an aerobic glycolysis which called Warburg effect. Lactic acid surrounding the tumor tissues can reach up to 40 mmol/L, and lactic acidosis in cancer patients is correlated with rapid cancer growth, metastasis, and poor survival (1,27). Moreover, lactic acid contributes to the reduced immunity of the tumor-infiltration host inflammatory cells such as macrophages and lymphocytes (28-30).
Lactic acid can modulate inflammation and promote immune tolerance (1). Lactic acid increases cellular production of anti-inflammatory cytokines such as interleukin-10 (1,31). On the other hand, it reduces the activities of pro-inflammatory cytokines such as interleukin-12, macrophages, natural killer cells, and tumor necrosis factors (32,33). Aerobic glycolysis is prominently involved in wound healing. Lactic acid around the healing wounds would reach between 5 and 15 mmol/L (34-36). When acute tissue ischemia occur ischemia-induced lactic acid formation is an important cellular response and which is activated by the plasma membrane sodium proton exchanges (37,38). It increases intracellular sodium, and it leads to calcium overload via calcium-sodium exchange and inducing cell death (1,39). In the setting of sepsis related lactic acidosis, animals which pretreated with sodium-proton exchanger blockers develop less hemodynamic instability and better survival compared with non-treated control groups (40,41).
Hyperlactatemia in sepsis and septic shock
Hyperlactatemia in sepsis and septic shock occurs as a result of tissue hypoxia, in which the whole body oxygen delivery fails to meet the whole body oxygen requirements (2,42). Therefore, increased blood lactate concentration indicates anaerobic metabolism and tissue hypoxia. It follows from this reasoning that patients with an elevated blood lactate level should be treated by increasing oxygen delivery (21). Although enough oxygen was delivered to the tissues, in the setting of tissue oxygen extraction impairment, anaerobic metabolism generates lactate. Normally, most tissues can extract as much as 70% of the delivered oxygen before anaerobic metabolism. However, in sepsis and septic shock state, this critical oxygen extraction ratio is decreased to 50% or less so that lactic acid formation increases at oxygen deliveries that would normally be sufficient to meet the aerobic oxygen demand (2,43). Microcirculatory dysfunction, which impairs oxygen delivery to the tissues, and mitochondrial dysfunction, which impairs oxygen utility, occur in patients with sepsis so that, even in an adequate oxygenation, anaerobic metabolism occurs and pyruvate is shunted toward lactate production (2,21).
Endogenous and exogenous catecholamines are highly associated with lactic acid production in sepsis and septic shock (21). Because aerobic glycolysis is stimulated by high levels of circulating epinephrine. By binding to the β2-adrenergic receptor on the plasma membrane, epinephrine increases the glycolytic flux both directly and by stimulation of the ubiquitous adenosine triphosphatase sodium/potassium pump (Na+/K+-ATPase) and the resultant consumption of ATP (2,4,5,44). Thereby, ATP consumption generates adenosine diphosphate (ADP) via phosphofructokinase stimulation, thus reactivating glycolysis (4). Glycolytic flux can exceed the capacity of PDH to catalyze the conversion of pyruvate into acetyl-CoA. Therefore, pyruvate is inevitably converted to lactate by LDH (Figure 2) (2).
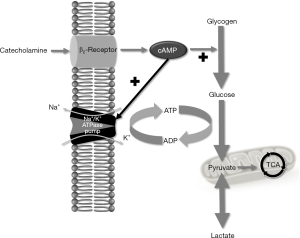
Reduced lactate clearance enhanced hyperlactatemia. In sepsis patients whose vital signs were stable, hyperlactatemia might be induced by the dysfunction of hepatic lactate clearance, which is primarily due to PDH inhibition (2,45). In patients with sepsis and low-flow state, chronic liver disease further compromises lactate clearance (5,46). PDH converts pyruvate into acetyl-CoA, allowing pyruvate to enter the mitochondria. PDH activity was decreased in patients with septic muscle and is restored by dichloroacetate, decreasing lactatemia in patients with sepsis (4). However, chronic liver disease alone causes only minimal hyperlactatemia, and kidney failure adds to the impairment in lactate clearance (5).
Role of lactic acid: prognosis marker of sepsis and septic shock
Lactic acidosis results from the accumulation of lactate and protons in the body fluid (5). However, glycolytic flux from glucose to pyruvate generates H+, but conversion of pyruvate to lactate consumes the molar equivalent H+ flux; therefore, increased generation of lactate resulting in hyperlactatemia is not, by itself, acidosis, but ATP hydrolysis is the major generator of H+ and becomes the source of acidosis (2).
Regardless of the source, increased lactate levels have been associated with worse outcomes (17). Moreover high initial lactate level as well as longer normalization time was associated with increased hazard of mortality (47). Lactic acidosis can cause a reduction of cardiac contractility and vascular hypo-responsiveness to vasopressors through various mechanisms. It is a precipitator of mortality and contributes to a worsening of underlying comorbidities (17). In normotensive patients with sepsis, a lactate concentration more than 4 mmol/L was found to be independently correlated with higher mortality and therefore needs urgent recognition and proper resuscitation (48). However, patients with septic shock with intermediate concentrations of lactate (2–4 mmol/L) have poorer prognosis than those with normal lactate concentration (49). Moreover, in the severity score, lactate weighted scoring system discriminated mortality significantly than others such as sequential organ failure assessment score (50).
In the Third International Consensus Definitions for Sepsis and Septic Shock, elevated lactate level was included as the third important variable along with hypotension and sustained need for vasopressor therapy to define septic shock (8). The risk adjusted hospital mortality was significantly higher in patients with fluid-resistant hypotension requiring vasopressors and hyperlactatemia compared with those with either hyperlactatemia alone or with fluid resistant hypotension requiring vasopressors but with a lactate level of <2 mmol/L (8).
Role of lactic acid: lactate-guided septic shock management
Since the time Rivers et al. first proposed EGDT, central venous oxygen saturation (ScvO2) has been widely used as a surrogate marker of the balance between oxygen delivery and consumption (6,51). Moreover, lactate is a useful biomarker of tissue hypoxia and anaerobic metabolism, reflecting disease severity and lactate clearance, and can be used as a therapeutic target instead of ScvO2 (51,52). However, current clinical trials have shown that EGDT targeting ScvO2 fails to improve outcomes compared with usual therapy or lactate-based protocols (9-11,51).
Recent SSC guideline recommended guiding resuscitation to normalize lactate in patients with elevated lactate levels, a marker of tissue hypoperfusion (12). Since 2013, they recommended bundle therapy for sepsis and septic shock; it consisted of four components that should be performed within 3 hours and three components that should be performed within 6 hours. The 3-hour bundle recommended the measurement of lactate levels, while the 6-hour bundle recommended the re-measurement of lactate if the initial lactate level was elevated (53).
The Sepsis-3 task force recommended that the monitoring of lactate should not be used as a guide to evaluate patient’s therapeutic response or should not be used as an indicator of illness severity. They recognized that serum lactate measurements are commonly, but not universally, available, especially in developing countries (8). However, there were five randomized controlled trials with 647 patients, which have evaluated the lactate-guided resuscitation of patients who had septic shock (7,13,14,52,54). Results showed that mortality was reduced in patients who received lactate-guided resuscitation compared with those who received resuscitation without lactate monitoring [risk ratio, 0.67; 95% confidence interval (CI), 0.53–0.84] (12). Two other meta-analyses of 647 patients demonstrated a moderate evidence of decreasing mortality in lactate guiding resuscitation strategy, compared with either usual critical care or with ScvO2 guiding strategy (15,55).
Lactate versus lactate clearance in patients with sepsis and septic shock
Repeated measurements of blood lactate levels after quantitative resuscitation can serve as a surrogate marker of patient’s response to therapy and may be more predictive of mortality than the initial lactate value. While the current surviving sepsis guidelines recommended the re-measurement of lactate levels within 6 hours if the initial lactate levels were elevated, no study has yet examined which time point is the most significant prognostic value of lactate from the recognition of shock at the emergency department in patients with septic shock. Nguyen et al. re-measured the lactate levels of patients 6 hours after the initial lactate level check and found that an optimal cutoff lactate clearance <10% had a sensitivity of 44.7% and specificity of 84.4% for predicting in-hospital mortality (56). Several studies also reported about lactate kinetics and clearance. These studies showed that lactate clearance greater than 10%, based on the initial measurement obtained during the first 2 to 6 hours of resuscitation, predicted survival in patients with septic shock (56-58). Moreover, it was demonstrated that for every 10% increase in lactate clearance, there was a corresponding 11% decrease in in-hospital mortality (56). In general, <10% of lactate clearance 6 hours from initial resuscitation was an independent predictor of in-hospital mortality (57,59). There was a systemic review and meta-analysis about lactate clearance and mortality in critically ill patients. They show that lactate clearance is strongly associated with all-cause mortality and rapid clearance is a strong predictor of survivor (60). However, there is not enough evidence to suggest a specific cutoff value of lactate clearance for resuscitation target goal, because among the recent studies there was a significant heterogeneity such as different time point and severity. Thus, we recommend to the clinicians to follow the current guidelines implementing a guided resuscitation to normalize lactate levels in patients with septic shock, although it supported with weak recommendations and low-quality evidence.
Marty et al. measured the lactate levels at time 0 (T0), T6, T12, and T24 and showed that the best predictor of death was the T24 clearance (61). Similarly, Herwanto et al. investigated the role of 6-, 12-, and 24-h lactate clearance in patients with sepsis and septic shock and showed only the 24-h lactate clearance measurement to be associated with mortality (62). Chertoff et al. reported that there was a delay in lactate clearance measurement 24–48 hours after initial resuscitation and that the median clearance of 31.6% was significantly associated with mortality (63). Although some changes in lactate kinetics were clearly significant within 6 to 24 hours after resuscitation, it is currently not possible to define the best time interval between lactate measurements (64).
Furthermore, the interesting issue is whether lactate or lactate clearance is more useful in guiding septic shock management. Lokhandwala et al. presented that sensitivity and specificity were significantly different when comparing subsequent lactate levels less than the recommended level vs. <10% lactate reduction in the non-vasopressor therapy hyperlactatemia group; however, unlike the complete cohort, no statistical difference was found when comparing a <20% lactate reduction to either of the previous metrics (65). Table 1 shows a comprehensive summary of the reports regarding the roles of lactate as a prognostic indicator of sepsis and septic shock (7,56,57,59,61,63-67).
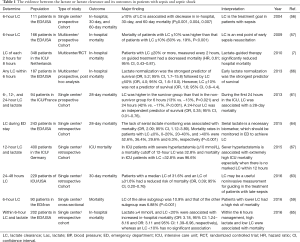
Full table
After the release of Sepsis-3, our knowledge on the prognostic value of lactate kinetics in patients with septic shock (12) remained insufficient as data on the prognostic value of lactate levels and clearance in patients with septic shock is limited. Thus, further research is needed to determine the prognostic value of lactate or lactate kinetics in patients with septic shock, as defined by Sepsis-3.
Sepsis and septic shock management enhancing lactate clearance
The important management of lactic acidosis is to treat the underlying cause. Thus, sepsis should be treated immediately by early administration of appropriate antibiotics and infection source control (2).
To reduce lactate production, the macro-circulatory oxygen delivery should improve first. The oxygen delivery depends on the patient’s cardiac output, hemoglobin, and oxygen saturation. Adequate volume resuscitation using inotropes, red blood cell transfusion, and provision of adequate oxygen supply are essential (2). The use of catecholamine should be limited as stimulation of β-adrenergic receptors increases glycolytic flux (44). In patients with septic shock, reduction of the norepinephrine dose by adding a low-dose vasopressin improved survival by 10% in patients initially receiving <15 µg/min norepinephrine in the vasopressin and septic shock trial (68). To reduce lactate production caused by overstimulation of the respiratory muscles, a mechanical ventilator support is required and sometimes neuromuscular blocker may help too (2).
To increase lactate removal, hepatic function should be preserved and monitored. Evidence of decreased hepatic function should be sought, and reversible contributors to hepatic dysfunction should be treated (2). In addition, potential hepatotoxins or renal toxins should be avoided. Continuous renal replacement therapy can be performed in critically ill patients with severe lactic acidosis and acute kidney injury (69). Sodium bicarbonate administration should be avoided, because it increases carbon dioxide production and decreases serum ionized calcium, which may decrease ventricular and vascular contractility (2).
Inducing a pyruvate metabolism in Krebs cycle decreases serum lactate levels. Thiamine administration may enhance aerobic metabolism by converting pyruvate to acetyl-CoA (70). Moreover, dichloroacetate lowers lactate concentrations and improves acidemia when oxygen is available by enhancing the activity of PDH. However, it does not improve the hemodynamic parameters or survival (71).
Conclusions
In patients with sepsis and septic shock, hyperlactatemia is promoted by glycolytic flux via anaerobic metabolism with tissue hypoxia, β-adrenergic receptor stimulation by endo/exogenous catecholamine, and decreased clearance due to hepatic and renal dysfunction. It reduces cardiac contractility and vascular hypo-responsiveness to vasopressors; however, it is closely associated with poor prognosis. Therefore, during sepsis and septic shock management, lactate levels should be re-measured and normalized. To normalize the lactate levels, we have to reduce glycolytic flux, enhance lactate removal, and induce pyruvate metabolism in the Krebs cycle. However, the most important treatment is to control the underlying infection.
Acknowledgements
We thank to Hye Won Jung for making the illustration.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Sun S, Li H, Chen J, et al. Lactic Acid: No Longer an Inert and End-Product of Glycolysis. Physiology (Bethesda) 2017;32:453-63. [Crossref] [PubMed]
- Suetrong B, Walley KR. Lactic acidosis in sepsis: it's not all anaerobic: implications for diagnosis and management. Chest 2016;149:252-61. [Crossref] [PubMed]
- Gladden LB. Lactate metabolism: a new paradigm for the third millennium. J Physiol 2004;558:5-30. [Crossref] [PubMed]
- Levy B. Lactate and shock state: the metabolic view. Curr Opin Crit Care 2006;12:315-21. [Crossref] [PubMed]
- Kraut JA, Madias NE. Lactic acidosis. N Engl J Med 2014;371:2309-19. [Crossref] [PubMed]
- Rivers E, Nguyen B, Havstad S, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001;345:1368-77. [Crossref] [PubMed]
- Jansen TC, van Bommel J, Schoonderbeek FJ, et al. Early lactate-guided therapy in intensive care unit patients: a multicenter, open-label, randomized controlled trial. Am J Respir Crit Care Med 2010;182:752-61. [Crossref] [PubMed]
- Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA 2016;315:801-10. [Crossref] [PubMed]
- Pro CI, Yealy DM, Kellum JA, et al. A randomized trial of protocol-based care for early septic shock. N Engl J Med 2014;370:1683-93. [Crossref] [PubMed]
- Investigators A, Group ACT, Peake SL, et al. Goal-directed resuscitation for patients with early septic shock. N Engl J Med 2014;371:1496-506. [Crossref] [PubMed]
- Mouncey PR, Osborn TM, Power GS, et al. Trial of early, goal-directed resuscitation for septic shock. N Engl J Med 2015;372:1301-11. [Crossref] [PubMed]
- Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Crit Care Med 2017;45:486-552. [Crossref] [PubMed]
- Lyu X, Xu Q, Cai G, et al. Efficacies of fluid resuscitation as guided by lactate clearance rate and central venous oxygen saturation in patients with septic shock. Zhonghua Yi Xue Za Zhi 2015;95:496-500. [PubMed]
- Yu B, Tian HY, Hu ZJ, et al. Comparison of the effect of fluid resuscitation as guided either by lactate clearance rate or by central venous oxygen saturation in patients with sepsis. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 2013;25:578-83. [PubMed]
- Gu WJ, Zhang Z, Bakker J. Early lactate clearance-guided therapy in patients with sepsis: a meta-analysis with trial sequential analysis of randomized controlled trials. Intensive Care Med 2015;41:1862-3. [Crossref] [PubMed]
- Connor H, Woods HF. Quantitative aspects of L(+)-lactate metabolism in human beings. Ciba Found Symp 1982;87:214-34. [PubMed]
- Foucher CD, Tubben RE. Lactic Acidosis. StatPearls. Treasure Island (FL)2017.
- Kellum JA, Kramer DJ, Lee K, et al. Release of lactate by the lung in acute lung injury. Chest 1997;111:1301-5. [Crossref] [PubMed]
- Iscra F, Gullo A, Biolo G. Bench-to-bedside review: lactate and the lung. Crit Care 2002;6:327-9. [Crossref] [PubMed]
- Borregaard N, Herlin T. Energy metabolism of human neutrophils during phagocytosis. J Clin Invest 1982;70:550-7. [Crossref] [PubMed]
- Garcia-Alvarez M, Marik P, Bellomo R. Sepsis-associated hyperlactatemia. Crit Care 2014;18:503. [Crossref] [PubMed]
- Miller BF, Fattor JA, Jacobs KA, et al. Lactate and glucose interactions during rest and exercise in men: effect of exogenous lactate infusion. J Physiol 2002;544:963-75. [Crossref] [PubMed]
- Petanjek Z, Judas M, Simic G, et al. Extraordinary neoteny of synaptic spines in the human prefrontal cortex. Proc Natl Acad Sci U S A 2011;108:13281-6. [Crossref] [PubMed]
- Goyal MS, Hawrylycz M, Miller JA, et al. Aerobic glycolysis in the human brain is associated with development and neotenous gene expression. Cell Metab 2014;19:49-57. [Crossref] [PubMed]
- Barros LF. Metabolic signaling by lactate in the brain. Trends Neurosci 2013;36:396-404. [Crossref] [PubMed]
- Liu X, Betzenhauser MJ, Reiken S, et al. Role of leaky neuronal ryanodine receptors in stress-induced cognitive dysfunction. Cell 2012;150:1055-67. [Crossref] [PubMed]
- Walenta S, Mueller-Klieser WF. Lactate: mirror and motor of tumor malignancy. Semin Radiat Oncol 2004;14:267-74. [Crossref] [PubMed]
- Fischer K, Hoffmann P, Voelkl S, et al. Inhibitory effect of tumor cell-derived lactic acid on human T cells. Blood 2007;109:3812-9. [Crossref] [PubMed]
- Mendler AN, Hu B, Prinz PU, et al. Tumor lactic acidosis suppresses CTL function by inhibition of p38 and JNK/c-Jun activation. Int J Cancer 2012;131:633-40. [Crossref] [PubMed]
- Sica A, Larghi P, Mancino A, et al. Macrophage polarization in tumour progression. Semin Cancer Biol 2008;18:349-55. [Crossref] [PubMed]
- Nasi A, Fekete T, Krishnamurthy A, et al. Dendritic cell reprogramming by endogenously produced lactic acid. J Immunol 2013;191:3090-9. [Crossref] [PubMed]
- Errea A, Cayet D, Marchetti P, et al. Lactate Inhibits the Pro-Inflammatory Response and Metabolic Reprogramming in Murine Macrophages in a GPR81-Independent Manner. PLoS One 2016;11:e0163694. [Crossref] [PubMed]
- Husain Z, Huang Y, Seth P, et al. Tumor-derived lactate modifies antitumor immune response: effect on myeloid-derived suppressor cells and NK cells. J Immunol 2013;191:1486-95. [Crossref] [PubMed]
- Ghani QP, Wagner S, Hussain MZ. Role of ADP-ribosylation in wound repair. The contributions of Thomas K. Hunt, MD. Wound Repair Regen 2003;11:439-44. [Crossref] [PubMed]
- Trabold O, Wagner S, Wicke C, et al. Lactate and oxygen constitute a fundamental regulatory mechanism in wound healing. Wound Repair Regen 2003;11:504-9. [Crossref] [PubMed]
- Hunt TK, Conolly WB, Aronson SB, et al. Anaerobic metabolism and wound healing: an hypothesis for the initiation and cessation of collagen synthesis in wounds. Am J Surg 1978;135:328-32. [Crossref] [PubMed]
- Mahnensmith RL, Aronson PS. The plasma membrane sodium-hydrogen exchanger and its role in physiological and pathophysiological processes. Circ Res 1985;56:773-88. [Crossref] [PubMed]
- Malo ME, Fliegel L. Physiological role and regulation of the Na+/H+ exchanger. Can J Physiol Pharmacol 2006;84:1081-95. [Crossref] [PubMed]
- Regli L, Anderson RE, Meyer FB. Effects of intermittent reperfusion on brain pHi, rCBF, and NADH during rabbit focal cerebral ischemia. Stroke 1995;26:1444-51; discussion 51-2. [Crossref] [PubMed]
- Sikes PJ, Zhao P, Maass DL, et al. Sodium/hydrogen exchange activity in sepsis and in sepsis complicated by previous injury: 31P and 23Na NMR study. Crit Care Med 2005;33:605-15. [Crossref] [PubMed]
- Wu D, Kraut JA. Potential role of NHE1 (sodium-hydrogen exchanger 1) in the cellular dysfunction of lactic acidosis: implications for treatment. Am J Kidney Dis 2011;57:781-7. [Crossref] [PubMed]
- Huckabee WE. Lactic Acidosis. Am J Cardiol 1963;12:663-6. [Crossref] [PubMed]
- Herbertson MJ, Werner HA, Russell JA, et al. Myocardial oxygen extraction ratio is decreased during endotoxemia in pigs. J Appl Physiol 1985;1995:479-86. [PubMed]
- Levy B, Desebbe O, Montemont C, et al. Increased aerobic glycolysis through beta2 stimulation is a common mechanism involved in lactate formation during shock states. Shock 2008;30:417-21. [Crossref] [PubMed]
- Levraut J, Ciebiera JP, Chave S, et al. Mild hyperlactatemia in stable septic patients is due to impaired lactate clearance rather than overproduction. Am J Respir Crit Care Med 1998;157:1021-6. [Crossref] [PubMed]
- Sterling SA, Puskarich MA, Jones AE. The effect of liver disease on lactate normalization in severe sepsis and septic shock: a cohort study. Clin Exp Emerg Med 2015;2:197-202. [Crossref] [PubMed]
- Zhang Z, Chen K, Ni H, et al. Predictive value of lactate in unselected critically ill patients: an analysis using fractional polynomials. J Thorac Dis 2014;6:995-1003. [PubMed]
- Casserly B, Phillips GS, Schorr C, et al. Lactate measurements in sepsis-induced tissue hypoperfusion: results from the Surviving Sepsis Campaign database. Crit Care Med 2015;43:567-73. [Crossref] [PubMed]
- Tang Y, Choi J, Kim D, et al. Clinical predictors of adverse outcome in severe sepsis patients with lactate 2-4 mM admitted to the hospital. QJM 2015;108:279-87. [Crossref] [PubMed]
- Zhang Z, Hong Y. Development of a novel score for the prediction of hospital mortality in patients with severe sepsis: the use of electronic healthcare records with LASSO regression. Oncotarget 2017;8:49637-45. [PubMed]
- Shin TG, Jo IJ, Hwang SY, et al. Comprehensive Interpretation of Central Venous Oxygen Saturation and Blood Lactate Levels During Resuscitation of Patients With Severe Sepsis and Septic Shock in the Emergency Department. Shock 2016;45:4-9. [Crossref] [PubMed]
- Jones AE, Shapiro NI, Trzeciak S, et al. Lactate clearance vs central venous oxygen saturation as goals of early sepsis therapy: a randomized clinical trial. JAMA 2010;303:739-46. [Crossref] [PubMed]
- Dellinger RP, Levy MM, Rhodes A, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med 2013;41:580-637. [Crossref] [PubMed]
- Tian HH, Han SS, Lv CJ, et al. The effect of early goal lactate clearance rate on the outcome of septic shock patients with severe pneumonia. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue 2012;24:42-5. [PubMed]
- Simpson SQ, Gaines M, Hussein Y, et al. Early goal-directed therapy for severe sepsis and septic shock: A living systematic review. J Crit Care 2016;36:43-8. [Crossref] [PubMed]
- Nguyen HB, Rivers EP, Knoblich BP, et al. Early lactate clearance is associated with improved outcome in severe sepsis and septic shock. Crit Care Med 2004;32:1637-42. [Crossref] [PubMed]
- Arnold RC, Shapiro NI, Jones AE, et al. Multicenter study of early lactate clearance as a determinant of survival in patients with presumed sepsis. Shock 2009;32:35-9. [Crossref] [PubMed]
- Chertoff J, Chisum M, Garcia B, et al. Lactate kinetics in sepsis and septic shock: a review of the literature and rationale for further research. J Intensive Care 2015;3:39. [Crossref] [PubMed]
- Bolvardi E, Malmir J, Reihani H, et al. The role of lactate clearance as a predictor of organ dysfunction and mortality in patients with severe sepsis. Mater Sociomed 2016;28:57-60. [Crossref] [PubMed]
- Zhang Z, Xu X. Lactate clearance is a useful biomarker for the prediction of all-cause mortality in critically ill patients: a systematic review and meta-analysis*. Crit Care Med 2014;42:2118-25. [Crossref] [PubMed]
- Marty P, Roquilly A, Vallee F, et al. Lactate clearance for death prediction in severe sepsis or septic shock patients during the first 24 hours in Intensive Care Unit: an observational study. Ann Intensive Care 2013;3:3. [Crossref] [PubMed]
- Herwanto V, Lie KC, Suwarto S, et al. Role of 6-hour, 12-hour, and 24-hour lactate clearance in mortality of severe sepsis and septic shock patients. Critical Care 2014;18:2. [Crossref]
- Chertoff J, Chisum M, Simmons L, et al. Prognostic utility of plasma lactate measured between 24 and 48 h after initiation of early goal-directed therapy in the management of sepsis, severe sepsis, and septic shock. J Intensive Care 2016;4:13. [Crossref] [PubMed]
- Dettmer M, Holthaus CV, Fuller BM. The impact of serial lactate monitoring on emergency department resuscitation interventions and clinical outcomes in severe sepsis and septic shock: an observational cohort study. Shock 2015;43:55-61. [Crossref] [PubMed]
- Lokhandwala S, Andersen LW, Nair S, et al. Absolute lactate value vs relative reduction as a predictor of mortality in severe sepsis and septic shock. J Crit Care 2017;37:179-84. [Crossref] [PubMed]
- Puskarich MA, Trzeciak S, Shapiro NI, et al. Whole blood lactate kinetics in patients undergoing quantitative resuscitation for severe sepsis and septic shock. Chest 2013;143:1548-53. [Crossref] [PubMed]
- Haas SA, Lange T, Saugel B, et al. Severe hyperlactatemia, lactate clearance and mortality in unselected critically ill patients. Intensive Care Med 2016;42:202-10. [Crossref] [PubMed]
- Russell JA, Walley KR, Singer J, et al. Vasopressin versus norepinephrine infusion in patients with septic shock. N Engl J Med 2008;358:877-87. [Crossref] [PubMed]
- De Corte W, Vuylsteke S, De Waele JJ, et al. Severe lactic acidosis in critically ill patients with acute kidney injury treated with renal replacement therapy. J Crit Care 2014;29:650-5. [Crossref] [PubMed]
- Berg KM, Gautam S, Salciccioli JD, et al. Intravenous thiamine is associated with increased oxygen consumption in critically ill patients with preserved cardiac index. Ann Am Thorac Soc 2014;11:1597-601. [Crossref] [PubMed]
- Stacpoole PW, Wright EC, Baumgartner TG, et al. A controlled clinical trial of dichloroacetate for treatment of lactic acidosis in adults. The Dichloroacetate-Lactic Acidosis Study Group. N Engl J Med 1992;327:1564-9. [Crossref] [PubMed]
Cite this article as: Ryoo SM, Kim WY. Clinical applications of lactate testing in patients with sepsis and septic shock. J Emerg Crit Care Med 2018;2:14.


