
Anticoagulation in extracorporeal membrane oxygenation
Introduction
Extracorporeal membrane oxygenation (ECMO) supports patients in refractory cardiac and/or respiratory failure (1). ECMO has evolved over the years with advancements in hardware and expert skills, such that increasingly complex patients are being supported with more complex circuits (e.g., incorporating plasmapheresis, dialysis, molecular adsorbent recirculating system) (2-4). Concurrent supportive care such as nutrition, physiotherapy, mobilization regimes and transportation during ECMO support continue to be developed (5). Experience is increasing with the Extracorporeal Life Support Organization Registry (ELSO) reporting more than 85,000 ECMO runs as of January 2017 (6). Nevertheless, anticoagulation, an essential component of ECMO support, remains an area of ongoing challenge with patients suffering haemostatic imbalance and physicians still struggling to establish the optimal management strategy for anticoagulation on ECMO (7).
Bleeding and thrombotic complications are the most frequent causes of death and morbidity affecting patients on ECMO (8,9). Bleeding (e.g., surgical site bleeding, cannula site bleeding and intracranial haemorrhage) and thrombosis (e.g., circuit clots, oxygenator failure and hemolysis) occurred in 38–70% and 31–44% of patients, respectively (8,9). Both bleeding and thrombosis were associated with decreased survival {relative risk (RR) 0.59 [95% confidence interval (CI): 0.53–0.66] and 0.67 (95% CI: 0.60–0.74), respectively}. Anticoagulation practices vary among centres and may impact bleeding/thrombotic outcomes as well as overall cost of ECMO support (10,11). In addition to describing the fundamental effect of ECMO on coagulation pathways, we will in this review, also present a summary of the medical literature on the newer alternative anticoagulants used in ECMO and new monitoring strategies.
Methods
We reviewed studies on all age groups including neonates, pediatric and adult subjects, and all types of ECMO including cardiac, respiratory and extracorporeal cardiopulmonary resuscitation. Anticoagulation use in ventricular assist devices, Berlin heart devices, and extracorporeal carbon dioxide (CO2) removal are beyond the scope of this review. In view of the limited medical literature, we included all published studies, including retrospective studies, to comprehensively assess this topic. We summarized the medical literature into the following subheadings: (I) the pathophysiology of impaired haemostasis on ECMO; (II) the use of anticoagulants; (III) the role of haemostatic adjuncts; (IV) tests of anticoagulation; and (V) future directions.
The pathophysiology of impaired haemostasis on ECMO
During ECMO support, impairments in haemostasis are in part due to exposure of blood to non-biological surfaces of the extracorporeal circuit that activates both the coagulation and inflammatory pathways, and in part due to ongoing disease processes that cause varying degrees of immune dysregulation, endothelial dysfunction and consumption of coagulation factors (12-15). Underlying disease processes (e.g., sepsis, systemic autoimmune syndromes and severe burns) result in a systemic inflammatory response. The widespread cytokine storm [such as interleukin (IL)-6, IL-1, IL-8 and tumour necrosis factor alpha], complement activation and interactions between inflammatory cells and endothelium further impair endothelial integrity (16). Changes in the haemostatic system can be broadly divided into effects on platelets and on the coagulation system.
Platelets are activated upon contact with ECMO circuit surfaces and with high shear stress and turbulence within the circuit (Figure 1) (17). Activated platelets amplify coagulation pathways, aggregate and adhere onto the circuit surfaces and to damaged endothelium, resulting in both thrombosis and consumptive coagulopathy (18). Fibrinogen adsorption onto artificial surfaces also contributes to platelet activation via glycoprotein IIb/IIIa binding (18). Platelet counts are expected to drop within the first few hours of ECMO and patients who remain on ECMO longer often have declining platelet counts over time (19). Platelet function has also been shown to be affected, with impairment of platelet aggregation noted in a time-dependent manner (17,20). Acquired von Willebrand syndrome (AVWS) is a phenomenon that has been recognized to occur during ECMO runs (21-23). AVWS is characterized by loss of high molecular weight multimers of von Willebrand factor (VWF) as a result of high shear stress, and leads to impaired binding of VWF to platelets and to the subendothelial matrix, which is not reversed by repeated platelet transfusions (17,20). A small adult study reported a high prevalence of AVWS in 31/32 (97%) patients on ECMO and 22/32 (69%) had bleeding complications (22). Severe qualitative platelet dysfunction for adenosine diphosphate (ADP)- and arachidonic acid (AA)-mediated aggregation were found on thromboelastography-platelet mapping studies which were associated with severe bleeding and mortality on ECMO (24).
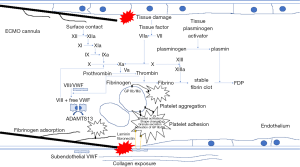
Effects on the coagulation system include contact activation of the intrinsic pathway via exposure of circulating factor XII and prekallikrein to non-biological surfaces, which triggers fibrinolysis and bradykinin release downstream (initiating inflammatory and the renin-angiotensin pathways) (16). Exposure of negatively charged collagen in damaged endothelium also triggers activation of high molecular weight kininogen, prekallikrein and factor XII which form a complex on collagen and further amplifies the intrinsic pathway (16). The extrinsic coagulation pathway is activated via the exposure of subendothelial tissue factor on damaged endothelia to circulating factor VII (16). This is the major pathway responsible for thrombosis and disseminated intravascular coagulation (18).
Acquired antithrombin (AT) deficiency is commonly seen upon initiation of ECMO. Produced in the liver, AT is an endogenous inhibitor of activated clotting factors, including factor Xa and thrombin. AT deficiency that develops during ECMO runs can be attributed to a combination of accelerated consumption and reduced synthesis (25). Heparin acts by potentiating the anticoagulant effects of AT. AT deficiency leads to heparin resistance and potentially thrombosis (26).
Anticoagulant agents
An international cross-sectional survey (n=121 respondents) demonstrated that unfractionated heparin (UFH) was universally used for anticoagulation in ECMO (10). UFH is inexpensive, familiar to most physicians and has an age-related, dose-dependent short half-life (range 1–2 hours) (27). The effect of UFH is mediated via potentiation of the anticoagulant action of AT and induction of tissue factor pathway inhibitor, and results in inhibition of free thrombin, factors XIa, Xa, IXa and the tissue factor/factor VIIa complex (28). However, limitations of UFH include ineffective inhibition of platelet-bound factor Xa, phospholipid-bound factor Va–Xa complex and fibrin-bound thrombin. It also has the potential to cause heparin induced thrombocytopenia (HIT) (29). UFH also has undesirable pharmacokinetic properties; it binds to plasma proteins including acute‐phase reactants leading to heparin resistance especially in acutely ill patients (30). Moreover, heparin therapy itself produces a decrease in circulating AT which also leads to heparin resistance (30).
Heparin resistance is a phenomenon whereby high doses of UFH are required to achieve therapeutic activated clotting time (ACT) levels. AT concentrate or fresh frozen plasma is given to replenish AT levels. The clinical benefit of replacing AT routinely is however, controversial. A retrospective study in paediatric ECMO (n=64) aiming to raise the AT levels to 120% (normal 80–120% in children and adults) showed a reduction in the UFH infusion rate of 10.1 unit/kg/hour (95% CI: 7.6–36.6) but no change in thrombosis or bleeding rates (31). Another retrospective paediatric ECMO study (n=40) aiming for 100% AT levels showed that AT replacement was independently associated with increased risk of circuit change compared to the control group [hazard ratio 3.15 (95%CI: 1.21–8.16)] (32). A retrospective neonatal ECMO study (n=162) targeting 100% AT levels in turn, showed that the AT group had less thrombotic/haemorrhagic complications (40.3% vs. 66.7%; P<0.001), less transfusions per day (54.7±20.1 vs. 67.4±34.9 mL/kg per day, P=0.001) and higher anti-Xa assay levels (0.48±0.16 vs. 0.3±0.14; P<0.001) compared to controls (33).
HIT, an immune-mediated side effect of heparin therapy characterized by thrombocytopenia and by a paradoxical prothrombotic state following heparin exposure, occurs in around 0.8–7% of adult patients on ECMO (29,34). Its occurrence is rare in children, being only reported in scattered case reports (35,36). These reports do not evaluate outcomes related to HIT, however, extrapolated from other groups of patients who develop HIT, the management involves immediate withdrawal of heparin and switching to alternative anticoagulants to maintain circuit patency and to prevent thrombotic complications (37).
Alternative anticoagulants (e.g., argatroban, bivalirudin and lepirudin [direct thrombin inhibitors (DTI)] are used only by a minority of providers (Table 1) (10). Of these, bivalirudin is supported by evidence derived from retrospective adult and pediatric studies (38-40). Unlike UFH, the DTI are not dependent on AT for their anticoagulant effect but directly inhibit both free circulating and fibrin bound thrombin. The short half-life (25 minutes) of bivalirudin in particular makes it suitable for rapid titration (41). Of note, the doses of bivalirudin would need to be adjusted in patients with renal impairment (42). Bivalirudin is administered as a continuous infusion with the dose ranging between 0.025–0.48 mg/kg/hour. It is usually titrated to target a therapeutic APTT range. A positive correlation was demonstrated between bivalirudin dose and APTT (r2=0.267; P=0.044) (40). Two retrospective studies compared bivalirudin to UFH and found that bivalirudin resulted in more stable APTT measurements (38,39). The largest study (n=21) which included both adults and children, showed better preservation of platelet count and AT levels, less bleeding and counterintuitively, lower overall cost despite the higher cost of bivalirudin (38).

Full table
Another DTI, argatroban, has a slightly longer half-life (39–51minutes) (43). However, it is metabolized in the liver and hence contraindicated in severe hepatic dysfunction. Case reports and case series in adult, paediatric and neonatal ECMO have reported the use of argatroban in place of UFH in suspected/proven HIT (35,44-48). Some utilized an initial bolus dose of 100–200 µg/kg, while the maintenance dose reported ranged from 0.1–0.65 µg/kg/min. Dose titration was guided by APTT and ACT levels. Lepirudin as an anticoagulant in ECMO has been reported in only a small number of case reports (49,50). Infrequent usage is likely due to the less favourable, longer half-life (1.3 hours) and renal excretion, thus requiring caution and dose adjustment in renal impairment (51).
Regional anticoagulation in ECMO is currently in its developmental infancy. The benefit of regional anticoagulation as opposed to systemic anticoagulation is reduced bleeding since only the extracorporeal circuit is anticoagulated (52). A regional anticoagulant, Nafamostat mesylate, which is a protease inhibitor of the coagulation (thrombin, Xa, and XIIa), fibrinolytic and complement systems has been used as a regional anticoagulant for continuous renal replacement therapy in Japan (53). Its use in ECMO has been reported in a group of 13 patients with bleeding complications from ECMO (54). It was infused into the venous/drainage limb of the ECMO circuit and anticoagulation measured by ACT and APTT were lower in the samples taken from the patient compared to samples taken from the circuit. Citrate, long used for regional anticoagulation in continuous renal replacement therapy is also being evaluated for use in ECMO (clinicaltrials.gov identifier: NCT00968565) (55).
In summary, UFH remains the default anticoagulant used in ECMO. However, UFH is associated with several serious limitations. With increasing experience with the use of newer anticoagulants, these limitations may be overcome. With the current state of evidence, it is uncertain if the use of these new anticoagulants may one day surpass UFH.
The role of haemostatic adjuncts
Bleeding is a common and serious complication of ECMO occurring in up to 60–70% of adults or children put on ECMO (9,56). It includes cannula site bleeding, surgical site bleeding, gastro-intestinal bleeding, hemothorax, hemopericardium and intracranial hemorrhage. The most devastating, are intracranial hemorrhages which are reported to occur in up to 16% pediatric/neonatal ECMO runs and was associated with higher rates of brain death (9). Whereas in adults, the rate of intracranial hemorrhage is around 2% (56). Bleeding is independently associated with higher daily risk of mortality (56). As such, hemostatic adjuncts are employed to minimize this risk of bleeding and reduce transfusion requirements (10). These drugs provide weak haemostatic effects and do not replace transfusion of blood products and replacement of coagulation factors. Haemostatic adjuncts include antifibrinolytics (e.g., tranexamic acid and aminocaproic acid, aprotinin), desmopressin and recombinant factor VIIa (rFVIIa). However, evidence for their use specifically in ECMO is scarce.
Aminocaproic acid and tranexamic acid, derivatives of the amino acid lysine, are competitive inhibitors of plasminogen preventing its conversion to plasmin (57). They also competitively inhibit tissue plasminogen activator and directly inhibit plasmin. In the adult population, both have been used successfully in other types of traumatic, surgical (including cardiac surgery) and post-partum haemorrhage to reduce bleeding and reduce transfusion requirements (56,58,59). In neonatal ECMO, retrospective studies have reported the use of aminocaproic acid to be safe, and effective in reducing the incidence of cannulation site and surgical site bleeding (27,41,42). The efficacy of tranexamic acid in ECMO was reported in a small retrospective cohort (n=10) of neonates with congenital diaphragmatic hernia where the use of tranexamic acid was associated with lower incidence of surgical site bleeding and lower transfusion requirements (43). However, thrombotic complications were reported in the intervention arm. Aprotinin is a non-specific protease inhibitor with a complex mechanism of action (57,60). It interferes with contact factor activation (factor XII), fibrinolysis, renin-angiotensin system, and neutrophil activation—hence having anti-inflammatory, anticoagulation and procoagulation effects. Aprotinin use in the adult cardiac surgery population was associated with increased mortality, renal, cardiovascular and cerebrovascular events, hence, its use is limited (59,61,62). A large, single center retrospective study (n=564) in neonatal ECMO found no increase in mortality in patients on tranexamic acid and aprotinin. However, this study did not report the efficacy on haemostasis (63). Desmopressin, a synthetic vasopressin analogue, induces release of factor VIII and VWF. It produces a small reduction in postoperative blood loss in adult cardiac surgery especially in patients on aspirin (64). Its use in ECMO has been reported but there are no dedicated studies evaluating its efficacy in this group of patients (51).
rFVIIa was originally developed for the treatment of haemophilia with inhibitors (58). However, its use has expanded to include treatment of any haemorrhage that does not respond to plasma transfusion or other conventional therapy (65). Coagulation is triggered locally at the site of vascular injury as rFVIIa binds to tissue factor, and activates factors IX and X, ultimately leading to thrombin generation and clot formation. Given its local effects at the site of vascular injury, rFVIIa may have a role in achieving haemostasis in patients experiencing refractory postoperative bleeding complications. An international survey reported that more than half of ECMO providers use rFVIIa (10).
Paediatric and adult retrospective ECMO studies demonstrated a reduction in bleeding and transfusion requirements with the use of rFVIIa (66-68). Patients were deemed to be adequately transfused and surgical bleeds were excluded prior to rFVIIa administration. These studies utilised doses ranging from 45–90 µg/kg/dose. An adult ECMO study (n=66) reported that the rate of effective haemostasis was 93.3% (66). The rate of thromboembolic events was 3.3% (1 case) which was not significantly different from the control group. There was also no difference in the need for circuit change, ventilation time, infectious complications or survival between patients who received rFVIIa or not. However, the use of rFVIIa is limited due to the fear that it may cause overt thrombosis in the patient or the circuit as has been reported in small case series (68,69).
Some centres have reported success in the use of bleeding bundles encompassing standard initial steps in the control of bleeding: (I) ensuring correction of acidosis, hypothermia, hypocalcemia; (II) ensuring platelets, coagulation factors and fibrinogen are adequately replaced; (III) use of adjuncts like tranexamic acid, aprotinin, aminocaproic acid and desmopressin; and (IV) excluding surgical bleeding prior to administration of rFVIIa. First dose of rFVIIa was given in the presence of perfusion staff and a fully primed backup circuit ready for the possibility of circuit thrombosis (70).
Specific ECMO data on antifibrinolytic agents are lacking and its use is extrapolated from cardiac surgical data. Data on the use of rFVIIa reveals it to be a double-edged sword as there is a definite thrombotic risk. Moderate sized retrospective studies report its efficacy in reducing bleeding and transfusion. However, the increased thrombotic rate though not statistically significant is worrisome. Importantly, a bleeding bundle to ensure all contributory factors are addressed in a multipronged manner is rational.
Monitoring of anticoagulation in ECMO
Most ECMO programs routinely monitor ACT, full blood counts (FBC), prothrombin and activated partial thromboplastin time (PT and APTT) and fibrinogen (10). Additionally, AT levels 60/119 (50%), anti-factor Xa assay 46/115 (40%) and thromboelastography (TEG) 21/116 (18%) have been used at varying time points or on a “as needed” basis (10). There are limitations with each test and hence, a combination of tests are required for optimal anticoagulation management (71).
ACT remains the preferred choice for monitoring anticoagulation in ECMO (72). Minimum ranges of 140–220 and maximum ranges of 170–240 have been used (10,11). Its main advantages are that it is an inexpensive, point-of-care test, thus allowing for immediate heparin titration. There is also extrapolated experience from its use in cardiopulmonary bypass. There are, however, mechanistic differences between cardiac bypass and ECMO that affects the performance of ACT (72). These factors include higher doses of heparin used in bypass, the relatively prolonged nature of ECMO and its common association with systemic inflammation and multiorgan dysfunction (72). ACT measures the time to fibrin clot formation after adding a contact activator (e.g., glass, celite or kaolin) to whole blood. In the presence of haemodilution, hypothermia, abnormalities in coagulation factors, fibrinogen and platelets, and the use of adjuncts like aprotinin, ACT would not be able to measure the effect of heparin accurately. These co-existing conditions are common in ECMO, and renders ACT inaccurate as a measure of the anticoagulant effect of heparin (73).
APTT, an assessment of the intrinsic pathway, is measured in the laboratory (74). This test involves the addition of platelet poor plasma to the partial thromboplastin reagent. The result reflects the time taken for clot formation as detected by optical or electromechanical methods. Although this method can be affected by a variety of factors (e.g., drugs, hematocrit, acute phase reactants and abnormalities in coagulation factors) it has better correlation with heparin dose than ACT in the neonatal, paediatric and adult population (73-76).
In the anti-Xa assay, known amounts of factor Xa and AT are added to the sample (77). Heparin forms an inhibitory complex with AT and inactivates factor Xa. Therefore, excess amount of factor Xa remaining in the sample is inversely proportional to the original amount of heparin. This is detected using chromogenic methods. The results are then compared to a standard curve and are provided as a concentration of anti-Xa (units/mL). Hence, anti- Xa assay directly measures UFH activity and is considered the gold standard for monitoring UFH in ECMO. Compared with APTT, anti-Xa has a higher degree of correlation with heparin dose and less variation (73). The accuracy of the anti-Xa assay can however, be affected by hyperbilirubinemia, haemolysis and lipaemia which are not uncommon occurrences in patients on ECMO (78). Moreover, some centers may have limited availability and or turn-around time of anti-Xa assays.
A prospective adult ECMO study (n=22) correlating the clinical anti-thrombotic effect of ACT, APTT and anti-Xa levels, showed that every unit decrease in anti-Xa level was associated with increased odds of developing deep vein thrombosis [OR 7.28 (95% CI: 1.61–32.94)], whereas there was no relationship with ACT and APTT (79). A retrospective study in paediatric ECMO (n=62) showed that patients who did not require a circuit change had higher heparin doses and anti-Xa levels compared to patients who did require it. Each decrease of 0.01 IU/mL anti-Xa level increased the odds of requiring a circuit change [OR 1.105 (95% CI: 1.00–1.10)].
Lastly, other point-of-care tests like TEG, also called thromboelastometry (TEM), provides global information on the dynamics of clot development, stabilization and dissolution (80). Its use is extrapolated from experience in complex major surgery including cardiopulmonary bypass, having shown to be associated with decreased blood product administration and mortality (81,82). A prospective, observational study assessing the use of TEM in adult ECMO showed that reliable and timely information on haemostatic parameters could be obtained during bleeding episodes and guide transfusion decisions (83). However, there is lack of evidence supporting its routine use in ECMO with no published threshold parameters or therapeutic goals.
The currently available anticoagulation assays measure either specific pathways or global haemostasis, and each test may be influenced by a variety of factors. As such, the bedside physician must use this knowledge to make an overall judgement on how to manage a bleeding patient and minimise the thrombotic risk at the same time. Further advancement is required in the development of anticoagulation management protocols and algorithms. One such protocol which includes monitoring of ACT, FBC, APTT, anti-Xa, AT and TEG, was shown to reduce the occurrence of cannula site bleeding (22% to 12%; P=0.04), surgical site bleeding (38% to 25%; P=0.02) and increase the median ECMO circuit life (3.6 to 4.3 days; P=0.02). (11).
Future directions
UFH anticoagulation is associated with several limitations including heparin resistance, need for AT replacement and HIT. As such, there is growing experience with alternative anticoagulants. These drugs especially bivalirudin which has favourable pharmacokinetic properties are currently being evaluated in randomised trial. A study comparing bivalirudin and UFH in neonatal and pediatric ECMO is underway (clinicaltrials.gov identifier: NCT03318393) (55). Increasing knowledge about the safety and efficacy of these alternative anticoagulants may eventually challenge UFH as the anticoagulant of choice. We also anticipate the emergence of feasibility data on the use of regional anticoagulation in ECMO.
The cause of bleeding in a patient on ECMO is multifactorial, as demonstrated in the various pathways that can be affected. As such, the efficacy of an individual haemostatic agent may be difficult to evaluate in isolation in a research or clinical setting. Over the next few years, we expect more development of evidenced based bleeding bundles/protocols tailored to the capabilities of each ECMO centre.
Knowledge of various anticoagulant tests to map the different haemostatic pathways may soon be combined and incorporated into these bundles/protocols as these seem to translate into improved clinical outcomes. ACT and APTT, being crude measures of anticoagulation and influenced by many concomitant factors in ECMO, may be superseded by a combination of tests like anti-Xa assay, TEG/TEM and platelet mapping. We await trials evaluating anticoagulation monitoring algorithms based on TEG versus conventional algorithms based on ACT or APTT (clinicaltrials.gov identifier: NCT02271126) (55).
Conclusions
Alterations in almost every haemostatic pathway exist in the patient on ECMO producing both bleeding and thrombotic risks. As such, anticoagulation remains one of the most challenging aspects of ECMO management. Newer anticoagulation agents are being evaluated for use in ECMO and we can expect more data on regional anticoagulation methods in the next few years. Advancement in the form of bleeding bundles and anticoagulation management protocols are also anticipated to emerge.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Paden ML, Conrad SA, Rycus PT, et al. Extracorporeal Life Support Organization Registry Report 2012. ASAIO J 2013;59:202-10. [Crossref] [PubMed]
- Jhang J, Middlesworth W, Shaw R, et al. Therapeutic plasma exchange performed in parallel with extra corporeal membrane oxygenation for antibody mediated rejection after heart transplantation. J Clin Apher 2007;22:333-8. [Crossref] [PubMed]
- Santiago MJ, Sanchez A, Lopez-Herce J, et al. The use of continuous renal replacement therapy in series with extracorporeal membrane oxygenation. Kidney Int 2009;76:1289-92. [Crossref] [PubMed]
- Sparks BE, Cavarocchi NC, Hirose H. Extracorporeal membrane oxygenation with multiple-organ failure: Can molecular adsorbent recirculating system therapy improve survival? J Heart Lung Transplant 2017;36:71-6. [Crossref] [PubMed]
- Wong JJ, Cheifetz IM, Lee JH. Extracorporeal membrane oxygenation for severe pediatric respiratory failure. J Emerg Crit Care Med 2017;1:11. [Crossref]
- Extracorporeal Life Support Organization. International Summary; January, 2017. ECLS Registry Report.
- Ranucci M. The conundrum of anticoagulation and hemostatic management in ECMO patients. Minerva Anestesiol 2016;82:147-8. [PubMed]
- Dalton HJ, Garcia-Filion P, Holubkov R, et al. Association of bleeding and thrombosis with outcome in extracorporeal life support. Pediatr Crit Care Med 2015;16:167-74. [Crossref] [PubMed]
- Dalton HJ, Reeder R, Garcia-Filion P, et al. Factors Associated with Bleeding and Thrombosis in Children Receiving Extracorporeal Membrane Oxygenation. Am J Respir Crit Care Med 2017;196:762-71. [Crossref] [PubMed]
- Bembea MM, Annich G, Rycus P, et al. Variability in anticoagulation management of patients on extracorporeal membrane oxygenation: an international survey. Pediatr Crit Care Med 2013;14:e77-84. [Crossref] [PubMed]
- Northrop MS, Sidonio RF, Phillips SE, et al. The use of an extracorporeal membrane oxygenation anticoagulation laboratory protocol is associated with decreased blood product use, decreased hemorrhagic complications, and increased circuit life. Pediatr Crit Care Med 2015;16:66-74. [Crossref] [PubMed]
- Urlesberger B, Zobel G, Zenz W, et al. Activation of the clotting system during extracorporeal membrane oxygenation in term newborn infants. J Pediatr 1996;129:264-8. [Crossref] [PubMed]
- Grossmann R, Babin-Ebell J, Misoph M, et al. Changes in coagulation and fibrinolytic parameters caused by extracorporeal circulation. Heart Vessels 1996;11:310-7. [Crossref] [PubMed]
- Saini A, Spinella PC. Management of anticoagulation and hemostasis for pediatric extracorporeal membrane oxygenation. Clin Lab Med 2014;34:655-73. [Crossref] [PubMed]
- McManus ML, Kevy SV, Bower LK, et al. Coagulation factor deficiencies during initiation of extracorporeal membrane oxygenation. J Pediatr 1995;126:900-4. [Crossref] [PubMed]
- Millar JE, Fanning JP, McDonald CI, et al. The inflammatory response to extracorporeal membrane oxygenation (ECMO): a review of the pathophysiology. Crit Care 2016;20:387. [Crossref] [PubMed]
- Robinson TM, Kickler TS, Walker LK, et al. Effect of extracorporeal membrane oxygenation on platelets in newborns. Crit Care Med 1993;21:1029-34. [Crossref] [PubMed]
- Hoffman M, Monroe DM 3rd. A cell-based model of hemostasis. Thromb Haemost 2001;85:958-65. [PubMed]
- Abrams D, Baldwin MR, Champion M, et al. Thrombocytopenia and Extracorporeal Membrane Oxygenation in Adults with Acute Respiratory Failure: A Cohort Study. Intensive Care Med 2016;42:844-52. [Crossref] [PubMed]
- Cheung PY, Sawicki G, Salas E, et al. The mechanisms of platelet dysfunction during extracorporeal membrane oxygenation in critically ill neonates. Crit Care Med 2000;28:2584-90. [Crossref] [PubMed]
- Pasala S, Fiser RT, Stine KC, et al. von Willebrand factor multimers in pediatric extracorporeal membrane oxygenation support. ASAIO J 2014;60:419-23. [Crossref] [PubMed]
- Heilmann C, Geisen U, Beyersdorf F, et al. Acquired von Willebrand syndrome in patients with extracorporeal life support (ECLS). Intensive Care Med 2012;38:62-8. [Crossref] [PubMed]
- Shim K, Anderson PJ, Tuley EA, et al. Platelet-VWF complexes are preferred substrates of ADAMTS13 under fluid shear stress. Blood 2008;111:651-7. [Crossref] [PubMed]
- Saini A, Hartman ME, Gage BF, et al. Incidence of Platelet Dysfunction by Thromboelastography-Platelet Mapping in Children Supported with ECMO: A Pilot Retrospective Study. Front Pediatr 2016;3:116. [PubMed]
- Hirose H, Sarik J, Pitcher H, et al. Antithrombin III deficiency in patients requiring mechanical circulatory support. J Cardiol Clin Res 2014;2:1017.
- Sarik J, Unai S, Pitcher H, et al. Antithrombin III Deficiency in Patients Requiring Extracorporeal Membrane Oxygenation (ECMO) Support. Chest 2013;144:319A. [Crossref]
- Downard CD, Betit P, Chang RW, et al. Impact of AMICAR on hemorrhagic complications of ECMO: a ten-year review. J Pediatr Surg 2003;38:1212-6. [Crossref] [PubMed]
- Buckley LF, Reardon DP, Camp PC, et al. Aminocaproic acid for the management of bleeding in patients on extracorporeal membrane oxygenation: Four adult case reports and a review of the literature. Heart Lung 2016;45:232-6. [Crossref] [PubMed]
- Glick D, Dzierba AL, Abrams D, et al. Clinically suspected heparin-induced thrombocytopenia during extracorporeal membrane oxygenation. J Crit Care 2015;30:1190-4. [Crossref] [PubMed]
- Anderson JA, Saenko EL. Heparin resistance. Br J Anaesth 2002;88:467-9. [Crossref] [PubMed]
- Wong TE, Delaney M, Gernsheimer T, et al. Antithrombin concentrates use in children on extracorporeal membrane oxygenation: a retrospective cohort study. Pediatr Crit Care Med 2015;16:264-9. [Crossref] [PubMed]
- Byrnes JW, Swearingen CJ, Prodhan P, et al. Antithrombin III supplementation on extracorporeal membrane oxygenation: impact on heparin dose and circuit life. ASAIO J 2014;60:57-62. [Crossref] [PubMed]
- Stansfield BK, Wise L, Ham PB 3rd, et al. Outcomes following routine antithrombin III replacement during neonatal extracorporeal membrane oxygenation. J Pediatr Surg 2017;52:609-13. [Crossref] [PubMed]
- Sokolovic M, Pratt AK, Vukicevic V, et al. Platelet Count Trends and Prevalence of Heparin-Induced Thrombocytopenia in a Cohort of Extracorporeal Membrane Oxygenator Patients. Crit Care Med 2016;44:e1031-7. [Crossref] [PubMed]
- Scott LK, Grier LR, Conrad SA. Heparin-induced thrombocytopenia in a pediatric patient receiving extracorporeal support and treated with argatroban. Pediatr Crit Care Med 2006;7:255-7. [Crossref] [PubMed]
- Pollak U, Yacobobich J, Tamary H, et al. Heparin-induced thrombocytopenia and extracorporeal membrane oxygenation: a case report and review of the literature. J Extra Corpor Technol 2011;43:5-12. [PubMed]
- Alving BM. How I treat heparin-induced thrombocytopenia and thrombosis. Blood 2003;101:31-7. [Crossref] [PubMed]
- Ranucci M, Ballotta A, Kandil H, et al. Bivalirudin-based versus conventional heparin anticoagulation for postcardiotomy extracorporeal membrane oxygenation. Crit Care 2011;15:R275. [Crossref] [PubMed]
- Pieri M, Agracheva N, Bonaveglio E, et al. Bivalirudin versus heparin as an anticoagulant during extracorporeal membrane oxygenation: a case-control study. J Cardiothorac Vasc Anesth 2013;27:30-4. [Crossref] [PubMed]
- Nagle EL, Dager WE, Duby JJ, et al. Bivalirudin in pediatric patients maintained on extracorporeal life support. Pediatr Crit Care Med 2013;14:e182-8. [Crossref] [PubMed]
- Wilson JM, Bower LK, Fackler JC, et al. Aminocaproic acid decreases the incidence of intracranial hemorrhage and other hemorrhagic complications of ECMO. J Pediatr Surg 1993;28:536-40; discussion 540-1. [Crossref] [PubMed]
- Horwitz JR, Cofer BR, Warner BW, et al. A multicenter trial of 6-aminocaproic acid (Amicar) in the prevention of bleeding in infants on ECMO. J Pediatr Surg 1998;33:1610-3. [Crossref] [PubMed]
- van der Staak FH, de Haan AF, Geven WB, et al. Surgical repair of congenital diaphragmatic hernia during extracorporeal membrane oxygenation: hemorrhagic complications and the effect of tranexamic acid. J Pediatr Surg 1997;32:594-9. [Crossref] [PubMed]
- Dolch ME, Frey L, Hatz R, et al. Extracorporeal membrane oxygenation bridging to lung transplant complicated by heparin-induced thrombocytopenia. Exp Clin Transplant 2010;8:329-32. [PubMed]
- Mejak B, Giacomuzzi C, Heller E, et al. Argatroban usage for anticoagulation for ECMO on a post-cardiac patient with heparin-induced thrombocytopenia. J Extra Corpor Technol 2004;36:178-81. [PubMed]
- Mejak B, Giacomuzzi C, Shen I, et al. Cardiopulmonary bypass using argatroban as an anticoagulant for a 6.0-kg pediatric patient. J Extra Corpor Technol 2005;37:303-5. [PubMed]
- Phillips MR, Khoury AI, Ashton RF, et al. The dosing and monitoring of argatroban for heparin-induced thrombocytopenia during extracorporeal membrane oxygenation: a word of caution. Anaesth Intensive Care 2014;42:97-8. [PubMed]
- Beiderlinden M, Treschan T, Gorlinger K, et al. Argatroban in extracorporeal membrane oxygenation. Artif Organs 2007;31:461-5. [Crossref] [PubMed]
- Dager WE, Gosselin RC, Yoshikawa R, et al. Lepirudin in heparin-induced thrombocytopenia and extracorporeal membranous oxygenation. Ann Pharmacother 2004;38:598-601. [Crossref] [PubMed]
- Balasubramanian SK, Tiruvoipati R, Chatterjee S, et al. Extracorporeal membrane oxygenation with lepirudin anticoagulation for Wegener's granulomatosis with heparin-induced thrombocytopenia. ASAIO J 2005;51:477-9. [Crossref] [PubMed]
- Lotz C, Streiber N, Roewer N, et al. Therapeutic Interventions and Risk Factors of Bleeding During Extracorporeal Membrane Oxygenation. ASAIO J 2017;63:624-30. [Crossref] [PubMed]
- Zhang Z, Hongying N. Efficacy and safety of regional citrate anticoagulation in critically ill patients undergoing continuous renal replacement therapy. Intensive Care Med 2012;38:20-8. [Crossref] [PubMed]
- Choi JY, Kang YJ, Jang HM, et al. Nafamostat Mesilate as an Anticoagulant During Continuous Renal Replacement Therapy in Patients With High Bleeding Risk: A Randomized Clinical Trial. Medicine (Baltimore) 2015;94:e2392. [Crossref] [PubMed]
- Park JH, Her C, Min HK, et al. Nafamostat mesilate as a regional anticoagulant in patients with bleeding complications during extracorporeal membrane oxygenation. Int J Artif Organs 2015;38:595-9. [Crossref] [PubMed]
- Medicine USNLo. ClinicalTrials.gov. Available online: https://www.clinicaltrials.gov/ct2/home
- Hunt BJ. The current place of tranexamic acid in the management of bleeding. Anaesthesia 2015;70 Suppl 1:50-3, e18.
- Acid Epsilon Aminocaproic. N Engl J Med 1965;273:336-7. [Crossref] [PubMed]
- Ker K, Edwards P, Perel P, et al. Effect of tranexamic acid on surgical bleeding: systematic review and cumulative meta-analysis. BMJ 2012;344:e3054. [Crossref] [PubMed]
- Henry DA, Carless PA, Moxey AJ, et al. Anti-fibrinolytic use for minimising perioperative allogeneic blood transfusion. Cochrane Database Syst Rev 2011.CD001886. [PubMed]
- Ortmann E, Besser MW, Klein AA. Antifibrinolytic agents in current anaesthetic practice. BJA: British Journal of Anaesthesia 2013;111:549-63. [Crossref] [PubMed]
- Mangano DT, Tudor IC, Dietzel C. The risk associated with aprotinin in cardiac surgery. N Engl J Med 2006;354:353-65. [Crossref] [PubMed]
- Fergusson DA, Hebert PC, Mazer CD, et al. A comparison of aprotinin and lysine analogues in high-risk cardiac surgery. N Engl J Med 2008;358:2319-31. [Crossref] [PubMed]
- Lin CY, Shuhaiber JH, Loyola H, et al. The Safety and Efficacy of Antifibrinolytic Therapy in Neonatal Cardiac Surgery. PLoS One 2015;10:e0126514. [Crossref] [PubMed]
- Wademan BH, Galvin SD. Desmopressin for reducing postoperative blood loss and transfusion requirements following cardiac surgery in adults. Interact Cardiovasc Thorac Surg 2014;18:360-70. [Crossref] [PubMed]
- Dutta TK, Verma SP. Rational Use of Recombinant Factor VIIa in Clinical Practice. Indian J Hematol Blood Transfus 2014;30:85-90. [Crossref] [PubMed]
- Anselmi A, Guinet P, Ruggieri VG, et al. Safety of recombinant factor VIIa in patients under extracorporeal membrane oxygenation. Eur J Cardiothorac Surg 2016;49:78-84. [Crossref] [PubMed]
- Niebler RA, Punzalan RC, Marchan M, et al. Activated recombinant factor VII for refractory bleeding during extracorporeal membrane oxygenation. Pediatr Crit Care Med 2010;11:98-102. [Crossref] [PubMed]
- Repesse X, Au SM, Brechot N, et al. Recombinant factor VIIa for uncontrollable bleeding in patients with extracorporeal membrane oxygenation: report on 15 cases and literature review. Crit Care 2013;17:R55. [Crossref] [PubMed]
- Long MT, Wagner D, Maslach-Hubbard A, et al. Safety and efficacy of recombinant activated factor VII for refractory hemorrhage in pediatric patients on extracorporeal membrane oxygenation: a single center review. Perfusion 2014;29:163-70. [Crossref] [PubMed]
- Walker A, Davidson M, Chalmers E. Use of activated recombinant factor VII in pediatric extracorporeal membrane oxygenation. Pediatr Crit Care Med 2010;11:537-8; author reply 8-9. [Crossref] [PubMed]
- Nguyen T, Musick M, Teruya J. Anticoagulation monitoring during extracorporeal membrane oxygenation: is anti-factor Xa assay (heparin level) a better test?. Pediatr Crit Care Med 2014;15:178-9. [Crossref] [PubMed]
- Hattersley PG. Activated coagulation time of whole blood. JAMA 1966;196:436-40. [Crossref] [PubMed]
- Liveris A, Bello RA, Friedmann P, et al. Anti-factor Xa assay is a superior correlate of heparin dose than activated partial thromboplastin time or activated clotting time in pediatric extracorporeal membrane oxygenation. Pediatr Crit Care Med 2014;15:e72-9. [Crossref] [PubMed]
- Maul TM, Wolff EL, Kuch BA, et al. Activated partial thromboplastin time is a better trending tool in pediatric extracorporeal membrane oxygenation. Pediatr Crit Care Med 2012;13:e363-71. [Crossref] [PubMed]
- Atallah S, Liebl M, Fitousis K, et al. Evaluation of the activated clotting time and activated partial thromboplastin time for the monitoring of heparin in adult extracorporeal membrane oxygenation patients. Perfusion 2014;29:456-61. [Crossref] [PubMed]
- Khaja WA, Bilen O, Lukner RB, et al. Evaluation of heparin assay for coagulation management in newborns undergoing ECMO. Am J Clin Pathol 2010;134:950-4. [Crossref] [PubMed]
- Kitchen S. Problems In Laboratory Monitoring Of Heparin Dosage. Br J Haematol 2000;111:397-406. [Crossref] [PubMed]
- Vera-Aguilera J, Yousef H, Beltran-Melgarejo D, et al. Clinical Scenarios for Discordant Anti-Xa. Adv Hematol 2016;2016:6.
- Chu DC, Abu-Samra AG, Baird GL, et al. Quantitative measurement of heparin in comparison with conventional anticoagulation monitoring and the risk of thrombotic events in adults on extracorporeal membrane oxygenation. Intensive Care Med 2015;41:369-70. [Crossref] [PubMed]
- Whiting D, DiNardo JA. TEG and ROTEM: technology and clinical applications. Am J Hematol 2014;89:228-32. [Crossref] [PubMed]
- Ak K, Isbir CS, Tetik S, et al. Thromboelastography-based transfusion algorithm reduces blood product use after elective CABG: a prospective randomized study. J Card Surg 2009;24:404-10. [Crossref] [PubMed]
- Shore-Lesserson L, Manspeizer HE, DePerio M, et al. Thromboelastography-guided transfusion algorithm reduces transfusions in complex cardiac surgery. Anesth Analg 1999;88:312-9. [PubMed]
- Nair P, Hoechter DJ, Buscher H, et al. Prospective observational study of hemostatic alterations during adult extracorporeal membrane oxygenation (ECMO) using point-of-care thromboelastometry and platelet aggregometry. J Cardiothorac Vasc Anesth 2015;29:288-96. [Crossref] [PubMed]
Cite this article as: Wong JJ, Lam JC, Mok YH, Lee JH. Anticoagulation in extracorporeal membrane oxygenation. J Emerg Crit Care Med 2018;2:12.


