
Cons: should universal decolonization be performed routinely for critically ill patients?
“It turns out that things are not so close as we imagined.”
—Lord Kelvin
Introduction
The decolonization strategy is gradually applied in intensive care unit (ICU) through the years. Theoretical basis for supporting the strategy is based upon the notion that decolonization reduces the chance of healthcare-associated infections (HAIs) (1). Therefore, recommendation of taking universal decolonization program was proposed in some countries (2). Nevertheless, the evidence to support universal decolonization strategy in ICU is still lacking and adverse effects related to universal decolonization need serious concern.
Increasing bacterial resistance
Mupirocin
Mupirocin ointment is commonly used in universal decolonization programme either alone or with chlorhexidine and other antiseptics. However, mupirocin has gained more and more attentions duo to the data of increasing drug resistance which was reported from 1% to 81% these years (3). It has been proved that previous mupirocin exposure is a great risk for both high-level [minimal inhibitory concentration (MIC) is above 512 mg/L] or low-level (MIC is in the range from 8 to 256 mg/L) resistance (4). Mupirocin resistance was first reported in staphylococci in 1986, just 1 year after it was introduced into clinical use. Beginning from the next year (1986 to 1987), resistant strains had been slowly spreading in England from 3% to 6% (5). The date of mupirocin resistance was also revealed in the following years in the other countries of Europe and in the North America (6).
Point mutations in ileS gene encoded by chromosomally is the main source of low-level mupirocin resistance. Meanwhile, high-level mupirocin resistance (HLMR) is thought to arise from the plasmid acquisition, which contains both the mupA resistance element and the modified ileS-2 gene. The emergence of HLMR is also linked to multi-drug resistant (MDR) which leads to clinical treatment failure easily (7). Plasmid-mediated resistance genes can spread between different staphylococcal species clonally and horizontally. Although ileS-2 does not encode resistance to other antibiotics, it is reported that the emergence of ileS-2 carrying plasmids has been associated with resistance to other categories of antibiotics such as erythromycin levofloxacin, clindamycin, tetracycline and so on (4,8).
Some types of local skin and soft tissue infections were treated by mupirocin due to the pathogen isolated were S. aureus and streptococcal species. It also contributes to mupirocin resistance (9). An observational study about bloodstream infection found that the percentage of coagulase negative staphylococci isolated which carried ileS-2 gene was elevated from 8% in 2006 to 22% in 2011, P<0.01. This drug resistance crisis went with the sustained growth of mupirocin consumption in each of the corresponding years (10). Therefore, the more mupirocin use, the higher rate of resistance it may occur.
Chlorhexidine
Chlorhexidine is a major component in most decolonization programs. It is often used in various forms, such as hand rubs, mouth wash and skin antisepsis prior to surgery, although the real benefit and evidence are not clear (11). However, the inappropriate and off-label use of chlorhexidine led to more and more drug resistant organisms isolated which had attracted high attention from WHO.
Low concentrations of chlorhexidine could disrupt the cell wall together with the membranes which leads to the leakage of intracellular contents. High concentrations of chlorhexidine induce coagulation of intracellular contents (12). The first case of chlorhexidine resistance was reported in 1995 (13). QacA/B and smr are the two gene families recognized which are conferred to chlorhexidine resistance (14). QacA/B genes are mediated by plasmid which encodes proton-dependent multidrug efflux pumps. The expression of qacA/B genes leads to high-level chlorhexidine resistance, whereas the smr gene results in low-level resistance (15).
The increasing rate of methicillin-resistant Staphylococcus aureus (MRSA) with qacA and/or qacB genes isolation was reported in Taiwan, where chlorhexidine has been used in hand hygiene programme for 20 years. Only 1.7% among the 240 isolates was verified with a chlorhexidine MIC of ≥4 mg/L in 1990 while it increased to 40% in 2000 and almost 50% in 2005 (16).
Wand and his colleagues identified that Klebsiella pneumoniae with chlorhexidine-resistant also has the feature of cross-resistant to colistin which is the last defense of multidrug-resistant (MDR) bacteria (17). They found that this sort of Klebsiella pneumoniae firstly adapted to chlorhexidine which could survive under antiseptic exposure and then developed cross-resistance to colistin. When the investigators explored the mechanism of the cross-resistance, they found the critical role of the progress was the efflux pump (smvR). They also took notice of the fact that the same efflux pump could be found in other different species (Figure 1).
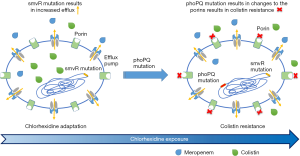
The finding can be taken as a warning because chlorhexidine is usually applied in decolonization strategy. This surprising new finding implied that exposure to chlorhexidine was associated with acquired resistance to colistin which stands as the last resort of antibiotics for treating MDR infections. So this discovery has significant clinical implications not only for infection prevention procedures but the therapeutic schedule of MDR infections as well. The new discovery revealed that inappropriate use of disinfectant might bring about a new path for pathogens to gain the ability of resisting to other sorts of antibiotics. Whether the application of decolonization strategy contained chlorhexidine in ICU patients with Klebsiella pneumoniae infection and/or colonization will affect the prognosis is still unknown. More researches about the important outcomes [e.g., mortality, length of ICU/hospital stay or ventilator-associated pneumonia (VAP) mortality] are needed to resolve the controversies.
We have to acknowledge that the appearance of resistance to mupirocin and chlorhexidine is so soon after their introduction to clinical application which is disappointing. Any decolonization strategy using mupirocin and/or chlorhexidine should take account of the possibility of resistance developing.
Decolonization failure and colonization re-occurrence
The rate of qacA or qacB gene detected among MRSA elevated from 65% to 91% in the last decade. The raise of resistance to chlorhexidine together with mupirocin, is associated with decolonization failure (18,19).
In Lona Mody’s randomized study, intranasal mupirocin ointment (given twice-daily treatment for 2 weeks) was compared with a placebo in patients with persistent colonization of S. aureus or MRSA (n=127). During the study period (6 months) they found that mupirocin did have the significant effect of eradicating S. aureus, including MRSA primely [60/64 (94%)] while the clearance rate was only 86% (54/63) in the placebo group. However, it is incredible that the recolonization rate was up to 39% after 90 days in the mupirocin group while the rate of recolonization in placebo group was only 17%, P<0.05 (20). Although recolonization was both occurred in the two groups, the development of resistance was uncommon which led to further controversies.
Another double-blind trial applied the decolonization programme with mupirocin ointment and chlorhexidine soap to evaluate the rates of bacterial eradication. At the 4 weeks study period, almost 44% patients in decolonization group (19/43) were free of MRSA in nasal while the control group was only 25% (21). This trial revealed that even though mupirocin was used with chlorhexidine as a decolonization strategy, the decolonization failure rate was as high as 56%. Although the failure rate was lower than the control group (56% vs. 75%, P<0.05), it indicated that the decolonization strategy applied at present might not the best choice.
One meta-analysis published in 2016 has concerned the trouble of carbapenem-resistant Enterobacteriaceae (CRE) and extended spectrum beta lactamase (ESBL) remained colonization or recolonization after decolonization in ICU. The systematic review included 37 studies, showing that a significant proportion of ESBL and CRE carriers remain colonized up to 1 year in the healthcare setting. It was similar to the control group (35.2% vs. 37.6%, P>0.05). Meanwhile, the development of resistance to decolonization antibiotics was reported in four studies including colistin and gentamicin (22).
It is well known that nose cavity is the main site of S. aureus colonization in human beings. However, the determinants of the host are not completely understood. Recent randomised controlled trials (RCTs) in including non-surgical patients could not verify the efficacy of S. aureus eliminating from nose cavity to prevent subsequent infection. Therefore, it is important for us to explore the mechanisms of S. aureus carriage in the host. Patients could be classified as an intermittent carrier or as a persistent carrier. The mechanisms of S. aureus nasal cavity carriage are multifactorial. Some scholars deduced that host characteristics co-determine the S. aureus colonization state in general and that an optimal fit between bacteria and host seems to be essential (23).
There are four essential conditions lead to S. aureus colonization in nasal. First, the contact of the two main components (nose and S. aureus). Second, S aureus has the chance or ability to adhere to specific receptors in nasal cavity. Third, immune defences of the host cannot eliminate S aureus initially. Last, S. aureus has the ability of propagate in the nose. It is imaginable that the innate immune response tries to prevent S. aureus from invading the mucosa heavily and causing a great deal of colonization or even severe infection. Thus, it is of great necessary for us to resolve the immunological mechanisms of colonization. Therefore, the best way to solve the problem of colonization is to adjust immunity of the host. Decolonization strategy cannot solve the root of the problem nor even the universal decolonization strategy.
The real effect of universal decolonization is uncertain
Whether the universal decolonization strategy could reduce the incidence of HAIs is still unknown. A meta-analysis showed the high heterogeneity of the five RCTs included (24). Different implementation such as the concentration and treatment duration of chlorhexidine might lead to different results, even in reducing the incidence of healthcare associated bloodstream infections (25,26).
In a prospective crossover study including 9,340 patients from five ICUs, the intervention group applied daily bathing programme with 2% chlorhexidine cloths while the control group use non-antibacterial cloths. The first stage lasted for 10 weeks followed by a 2 weeks washout period. Then all the units switched to the alternate group for another 10 weeks. During the whole study period, every unit included crossed over between bathing strategy 3 times. The results revealed that daily bathing strategy with chlorhexidine did not decrease the incidence of HAIs including CLABSI, CRUTI, VAP or CDI. The findings of this study could not provide evidence that critically ill patients will benefit from the universal decolonization strategy (27).
In conclusion, with the lack of evidence to support universal decolonization in ICU and the possibility of inducing bacterial resistance, universal decolonization cannot be routinely recommended. In order to decrease the additional antibiotics selection pressure in nosocomial pathogens and to protect the last defender of antibiotics, it is wise to restrict the application of universal decolonization strategy to those patients who will not get benefit from it. Adjusting immunity of the high risk host who was colonized might be a better way than universal decolonization to reduce the incidence of HAIs.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Solomkin JS, Mazuski J, Blanchard JC, et al. Introduction to the Centers for Disease Control and Prevention and the Healthcare Infection Control Practices Advisory Committee Guideline for the Prevention of Surgical Site Infections. Surg Infect (Larchmt) 2017;18:385-93. [Crossref] [PubMed]
- Allen-Bridson K, Gross C, Anttila A, et al. Health care-associated infections studies project: An American Journal of Infection Control and National Healthcare Safety Network data quality collaboration. Am J Infect Control 2017;45:1394-5. [Crossref] [PubMed]
- Liu X, Deng S, Huang J, et al. Dissemination of macrolides, fusidic acid and mupirocin resistance among Staphylococcus aureus clinical isolates. Oncotarget 2017;8:58086-97. [PubMed]
- Mahmoodzadeh Hosseini H, Kiyani N, Amin M, et al. Distribution of high-level mupirocin resistance among clinical MRSA. J Chemother 2017;29:215-9. [Crossref] [PubMed]
- Kavi J, Anderews JM, Wise R. Mupirocin-resistant Staphylococcus aureus. Lancet 1987;2:1472-3. [Crossref] [PubMed]
- Semret M, Miller MA. Topical mupirocin for eradication of MRSA colonization with mupirocin-resistant strains. Infect Control Hosp Epidemiol 2001;22:578-80. [Crossref] [PubMed]
- González-Domínguez M, Seral C, Potel C, et al. Genotypic and phenotypic characterization of methicillin-resistant Staphylococcus aureus (MRSA) clones with high-level mupirocin resistance. Diagn Microbiol Infect Dis 2016;85:213-7. [Crossref] [PubMed]
- Han LL, McDougal LK, Gorwitz RJ, et al. High frequencies of clindamycin and tetracycline resistance in methicillin-resistant Staphylococcus aureus pulsed-field type USA300 isolates collected at a Boston ambulatory health center. J Clin Microbiol 2007;45:1350-2. [Crossref] [PubMed]
- Hetem DJ, Bonten MJ. Clinical relevance of mupirocin resistance in Staphylococcus aureus. J Hosp Infect 2013;85:249-56. [Crossref] [PubMed]
- Bathoorn E, Hetem DJ, Alphenaar J, et al. Emergence of high-level mupirocin resistance in coagulase-negative staphylococci associated with increased short-term mupirocin use. J Clin Microbiol 2012;50:2947-50. [Crossref] [PubMed]
- Swanson JM. The Building Case for Chlorhexidine Decolonization in the Prevention of Healthcare-Associated Infections. Crit Care Med 2016;44:1938-9. [Crossref] [PubMed]
- Noto MJ, Wheeler AP. Understanding chlorhexidine decolonization strategies. Intensive Care Med 2015;41:1351-4. [Crossref] [PubMed]
- McDonnell G, Russell AD. Antiseptics and disinfectants: activity, action, and resistance. Clin Microbiol Rev 1999;12:147-79. [PubMed]
- George S, Leasure AR, Horstmanshof D. Effectiveness of Decolonization With Chlorhexidine and Mupirocin in Reducing Surgical Site Infections: A Systematic Review. Dimens Crit Care Nurs 2016;35:204-22. [Crossref] [PubMed]
- Roghmann MC, Lydecker AD, Langenberg P, et al. Microbiological effect of mupirocin and chlorhexidine for Staphylococcus aureus decolonization in community and nursing home based adults. Diagn Microbiol Infect Dis 2017;88:53-7. [Crossref] [PubMed]
- Wang JT, Sheng WH, Wang JL, et al. Longitudinal analysis of chlorhexidine susceptibilities of nosocomial methicillin-resistant Staphylococcus aureus isolates at a teaching hospital in Taiwan. J Antimicrob Chemother 2008;62:514-7. [Crossref] [PubMed]
- Wand ME, Bock LJ, Bonney LC, et al. Mechanisms of Increased Resistance to Chlorhexidine and Cross-Resistance to Colistin following Exposure of Klebsiella pneumoniae Clinical Isolates to Chlorhexidine. Antimicrob Agents Chemother 2016;61:e01162-16. [PubMed]
- Nakaminami H, Noguchi N, Nishijima S, et al. Characterization of the pTZ2162 encoding multidrug efflux gene qacB from Staphylococcus aureus. Plasmid 2008;60:108-17. [Crossref] [PubMed]
- Nakaminami H, Noguchi N, Nishijima S, et al. Transduction of the plasmid encoding antiseptic resistance gene qacB in Staphylococcus aureus. Biol Pharm Bull 2007;30:1412-5. [Crossref] [PubMed]
- Mody L, Kauffman CA, McNeil SA, et al. Mupirocin-based decolonization of Staphylococcus aureus carriers in residents of 2 long-term care facilities: a randomized, double-blind, placebo-controlled trial. Clin Infect Dis 2003;37:1467-74. [Crossref] [PubMed]
- Harbarth S, Dharan S, Liassine N, et al. Randomized, placebo-controlled, double-blind trial to evaluate the efficacy of mupirocin for eradicating carriage of methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother 1999;43:1412-6. [PubMed]
- Bar-Yoseph H, Hussein K, Braun E, et al. Natural history and decolonization strategies for ESBL/carbapenem-resistant Enterobacteriaceae carriage: systematic review and meta-analysis. J Antimicrob Chemother 2016;71:2729-39. [Crossref] [PubMed]
- Nouwen J, Boelens H, van Belkum A, et al. Human factor in Staphylococcus aureus nasal carriage. Infect Immun 2004;72:6685-8. [Crossref] [PubMed]
- Choi EY, Park DA, Kim HJ, et al. Efficacy of chlorhexidine bathing for reducing healthcare associated bloodstream infections: a meta-analysis. Ann Intensive Care 2015;5:31. [Crossref] [PubMed]
- Huang HP, Chen B, Wang HY, et al. The efficacy of daily chlorhexidine bathing for preventing healthcare-associated infections in adult intensive care units. Korean J Intern Med 2016;31:1159-70. [Crossref] [PubMed]
- Rupp ME, Cavalieri RJ, Lyden E, et al. Effect of hospital-wide chlorhexidine patient bathing on healthcare-associated infections. Infect Control Hosp Epidemiol 2012;33:1094-100. [Crossref] [PubMed]
- Noto MJ, Domenico HJ, Byrne DW, et al. Chlorhexidine bathing and health care-associated infections: a randomized clinical trial. JAMA 2015;313:369-78. [Crossref] [PubMed]
Cite this article as: Yu Y, Zhu C, Xu C, Chen E. Cons: should universal decolonization be performed routinely for critically ill patients? J Emerg Crit Care Med 2018;2:7.


