
The role of bedside point-of-care ultrasound evaluation of the lung in the critically ill patient
Our ability to obtain real-time clinical information and to apply it to crucial decision-making processes is a crucial factor that often will determine success in the medical intensive care units. The use of bedside ultrasound (US) technology in the assessment of critically unstable patient allows us to understand hemodynamics in a better way. Thus, the use of the bedside US has moved from the domain of radiologists to critical care practitioners with widespread acceptance of the need for structured training and protocols (1,2). Bedside US has always been widely used for procedural guidance, evaluation of shock states (3), assessment of preload responsiveness (4), and in examination of the heart as a part of heart-lung interaction as in acute respiratory distress syndrome (ARDS) (5,6). The lungs being usually full of air does not reflect sound waves as other organs do. Hence, the application of lung US was limited in the healthy patient. The injured lung on the other hand, has an abnormal echotexture because of the presence of fluid, secretions or air in a space that should not have either of these. This produces US artifacts resulting from the abnormal gas/tissue interface. It is crucial for the trainee to identify normal patterns of such air-lung tissue interaction before moving on to abnormal patterns produced by the interaction of air-filled lungs and the pleural tissue in diseased states.
The reasons for using lung US in the critical care environment are many. Thoracic CT scans may be the gold standard for diagnostic imaging for most critical care conditions, but it is expensive and often the patient will not be able to be moved out of the intensive care unit. The process of obtaining CT scans is time consuming and occasionally the presence of a radiologist reading the scan can be a limiting factor as well. Finally, the images are an accurate representation of the state of the lungs at the time the image was obtained and repeated scanning to assess for changes with treatment are often not possible due to costs as well as risks associated with exposure to radiation. Lung US can reliably identify pneumothorax, consolidation/atelectasis, interstitial syndromes, pleural effusions with a high degree of sensitivity (7-9) Experts consider it to be a reliable tool on the basis of which clinical decision can be taken (10). Standard radiological imaging such as X-rays do not have the ability to identify short interval differences in aeration among patients with ARDS. This is possible with using lung US and can aid in making crucial decisions such as for extubating patients (11-13). Diaphragmatic thickening fraction determined by US can help to determine readiness for extubating patients undergoing mechanical ventilation (14,15). Lung US has also proven to be an efficient diagnostic tool in the detection of pleural septations (16), necrotic consolidations (17), dynamic air bronchograms (18), and diaphragmatic function in respiratory failure (19). In view of these compelling evidences, several experts feel that the use of lung US in the assessment of critically ill patients is justified and should be regularly practiced (20).
Normal lungs US (Figure 1)
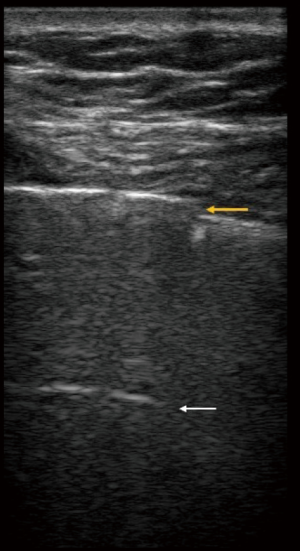
The US study of a normal lung and a clear idea of the nature of “healthy” artifacts is crucial before evaluation of diseased lungs are attempted. The US probe is to be help vertically over the chest wall in the intercostal space. View should be obtained in between the ribs so that the pleural line is visible. The “pleural line” slides craniocaudally with each breath and is usually located 0.5 cm below the rib line. This sliding pattern is referred to as “lung sliding” (21). On the M-mode, this line and the aerated lung below create the “seashore sign” (22). An isolated vertical B line may be visible in some normal lungs (23). These are described to be:
- They arise from the pleural line;
- They move with the pleural line (if the pleural line is mobile);
- They are hyperechoic;
- They reach the bottom of the screen;
- They flare out (if using a lower frequency phased array probe);
- They efface A-lines at their point of intersection. They also traverse them.
US probes of different frequencies can be used for examination of the lungs. High frequency probe such as the vascular probe are more useful to look for ‘lung sliding’ and other signs of pneumothorax, as well as studying B-lines. Lower frequency cardiac probe help with the imaging of deep lung tissues as in looking at consolidation and pleural effusion.
Lung US patterns in disease states
We will discuss the common signs on lung US specific to different diseases.
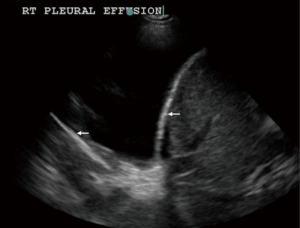
- Pleural effusion, empyema and complicated parapneumonic effusion (Figure 2): pleural effusion is detected best with the transducer in the craniocaudal orientation placed in a dependent position with the diaphragm, liver or spleen below and lungs margins visible in the same view. It appears as a hypoechoic and homogeneous structure with no gas inside and is present during expiration and inspiration. M-mode display demonstrates the sinusoidal sign due to movement of the visceral pleura from depth to periphery (24,25). Presence of septations on US predicts the need for pigtail drainage and intra-pleural fibrinolytic therapy in cases of pleural space infection (26). Lung US can also help in estimation of pleural fluid volume in a loculated effusion (27). Remérand et al. described a technique of multiplying the length of the pleural effusion measured in paravertebral regions by the cross-sectional area measured at the mid length to estimate the volume of the effusion which was found to be an accurate representation of the volume of effusion drained by thoracentesis (28).
- Pneumothorax: the presence of air in the pleural space leads to separation of the visceral and parietal pleural interposition. This leads to a series of findings on lung US. There is absence of lung sliding and B-lines. Absence of lung-sliding has a sensitivity of 95% and 100% negative predictive value in diagnosing pneumothorax (21). It is to be remembered that this is not a specific sign by itself and may be seen in pleural fibrosis, atelectasis, cardiac arrest, ARDS and in those undergoing jet ventilation (29). The lung US, in this case, reveals a space occupied by series of A lines which is caused by reverberation of the parietal pleural surface on M-mode (22). This appearance is referred to as the stratosphere sign. The partially inflated lungs can sometimes be seen to slide into the view with inspiration and a zone of transition between normal lung findings and zone with A-lines only. This is referred to as the “lung-point” (30). Location of lung point anteriorly indicates the presence of a moderate sized pneumothorax whereas a posteriorly located lung-point indicates a large pneumothorax. The lung-point mentioned above should be distinguished from a “physiologic lung-point” which is an artifact mimicking the lung point but in the absence of a pneumothorax. The distinction can be made by the presence of soft tissue with cardiac pulse can be visualized at the no-lung region instead of the A-line pattern seen at areas without lung sliding and pneumothorax (31).
- Pneumonia/consolidation/atelectasis: loss of a substantial area of lung aeration due to pus (pneumonia), blood (alveolar hemorrhage or contusion) allows the sound waves to transmit to greater depth from the pleural line than usual. This will appear hypoechoic overall but with areas that appear punctiform and are hyperechoic, corresponding to air bronchograms (air-filled bronchi) (32). A translobar consolidation gives the lung a liver-like appearance in echotexture. This is referred to as “tissue-sign”. The “shred-sign” (Figure 3) and “tissue-sign” have 90% sensitivity and 98% specificity in diagnosing consolidation (34). Lung US can reliably diagnose such areas and even aid with transthoracic needle aspiration if an abscess if suspected or cryptogenic pneumonia is strongly considered (35-38). Small areas of lung consolidations suggest pneumonia as a cause of hypoxic respiratory failure eighteen times more likely than a diagnosis of pulmonary embolism (PE) (39). The overall sensitivity of lung US in detection of community acquired pneumonia varies between 93.4% and 98% with specificity between 97.7% and 95% (40,41).
- Acute exacerbation of interstitial lung disease (AEILD): in the correct clinical setting, the presence of an alveolar-interstitial syndrome points towards exacerbation of interstitial lung diseases. The presence of three or more B-lines in between two ribs indicates the presence of interstitial syndrome with 93% accuracy (43). Interstitial syndrome describes a pattern of diffuse presence of three or more B-lines in the anterolateral lung fields (29). Such a pattern in the posterior dependent portions if the lung may be a result of gravitational accumulation of fluid in the interstitial. Lichtenstein et al. described the presence of 3–4 B-lines as septal rockets (29). The presence of twice as many was named ground-glass rockets which was suggested to correlate with ground-glass on chest X-rays. The presence of interstitial syndrome include left heart failure and pulmonary edema, ARDS, acute exacerbations of interstitial lung disease. The distance between each two adjacent B-lines has been reported to correlate with the degree of interstitial involvement on chest HRCT (44). When the B-lines appear widely separated from each other (7 mm). They suggest the presence of well-established fibrosis and honeycombing. On the other hand, a shorter distance between such B-lines (3 mm), produces a white lung appearance of chest X-rays as seen in early stage of alveolar-interstitial disease such as AEILD (44).
- Distinguishing pulmonary edema and multifocal pneumonia: it is often difficult to distinguish between multifocal pneumonia and pulmonary edema on chest X-rays. Both these conditions have excess lung water and can have predominant B line pattern on lug US examination. The presence of a “shred sign” can reliably help to distinguish between the two and direct management towards the right diagnosis (45). The “shred sign” was described by Lichtenstein et al. (34) as a sub-pleural hypoechoic area with an irregular deep border (fractal or shred sign) that is representative of areas of consolidated lung. This is representative of a non-translobar consolidation. Patients with pulmonary edema should not have the “shred sign” on their examination.
- PE: Reissig et al. has evaluated the utility of point of care lung US in the evaluation of suspected PE. Multiple, triangular, hypoechoic, and pleural-based parenchymal lesions with a localized and/or basal effusion were noted to be typical of pulmonary infarctions associated with PE. The sensitivity of US for detecting PEs was 80% and the specificity was 92%. Those with more central severe PE did not have peripheral parenchymal lesions but were more likely to have pleural effusions (46).
Areas of atelectasis can be recognized by a combination of absent lung sliding and visible heart activity at the pleural line. This is referred to as “lung-pulse” (42).

Lung US protocols in critical care medicine
There are three specific situations where a protocolized application of lung US can help us in diagnosis in a time-efficient and inexpensive manner.
- Acute hypoxic respiratory failure: the BLUE-protocol (Bedside Lung Ultrasound in Emergency) is a rapidly performed sequence of steps designed to sequentially rule out different etiologies of respiratory failure until the diagnosis reached. It takes about three minutes to perform in the hands of experts. The protocol requires that the examiner be trained in efficient application of lung US principles and can identify the presence of specific US features and hence recognize different profiles. The profiles are named based on the most important distinguishing feature as shown in Table 1.
- Shock: a systematic examination of the heart and lungs is designed to rule out obstructive shock followed by cardiogenic shock and finally hypovolemic hypovolemic shock. If these are excluded then a diagnosis of septic/distributive shock will likely be the underlying process. At first, a pericardial effusion with tamponade physiology is excluded followed by systematic evaluation of the right ventricle. A dilated right ventricle raises the possibility of a massive PE. If either is negative, then focused lung US is performed to search for an A′ profile looking for pneumothorax. If negative, lung US is performed to look for B-profile. Presence of B-profile would suggest a left sided cardiac failure and cardiogenic shock. In the absence of B-profile and A′ profile, intravenous fluid challenge is performed. If this results in resolution of the shock, a diagnosis of hypovolemic shock is made. Finally, shock refractory to fluid challenge itself and change on lung US exam findings from A-profile to B-profile without any improvement of shock points towards septic shock as being the cause (23,47). The BLUE-protocol has the highest sensitivity for detecting acute pulmonary edema (97%) and least for detecting PE (81%) with COPD exacerbation (89%), pneumonia (89%) and pneumothorax (88%) in between (39).
- Cardiac arrest: the SESAME (“Sequential Emergency Scanning Assessing Mechanism)-protocol is a fast examination of the lungs and heart in patients with cardiac arrest of an unknown cause. The first step is to exclude a pneumothorax by searching for lung sliding or the A′ sign. One must be cautious of detecting a pneumothorax just by the absence of lung sliding on the left if right mainstem endotracheal intubation has not yet been excluded. The protocol suggests scanning the veins of the calf looking for deep vein thrombosis (PE) in case of a non-traumatic arrest and the abdomen (fluid in the abdomen) for hemorrhagic shock if there is a history of trauma. If these are negative, an attempt should be made to obtain subcostal windows to look at the heart and diagnose pericardial fluid and tamponade physiology. The protocol requires practice and coordination with the staff so as to avoid stoppage of compressions during the examination (48).

Full table
The impact of lung US on the decision making process
Other than the obvious diagnostic utility of lung US in the intensive care unit, it also provides crucial information that helps to define the course of action when faced with a specific clinical question. Xirouchaki et al. published data on this particular aspect of the utility of lung US and reported that lung US directly changed the management of the patients in half of the included cases (10). Similar results have also been reported by other authors strengthening the justification for using lung US when dealing with a clinical question in the intensive care environment (36,39).
Training programs in US in ICU
A structured curriculum is necessary for the scientific training required to achieve mastery over a skill that is potentially life-saving in the ICU. Learning time for diagnosing certain conditions such as pneumothorax may be longer than other conditions due to its low incidence in the ICU (49). A 4-day long training course has been reported to be able produce technical skills among trainees that were described to range from “moderate” for the detection of pneumothorax and consolidation to “excellent” for detection of pleural effusion and interstitial syndrome (50). In view of the utility and excellent value for this skill, there should be a more concerted effort to teach this technique universally.
Conclusions
Despite the consolidated evidence supporting the use of lung US in the critical care setting, response among intensivist has been varied. Approximately one-third of intensivists in Australasian ICUs are experts with using lung US (51). A validated curriculum for universal application of lung US in the ICU as well as in every teaching program should be proposed. Lung US has already been recognized as an integral component to fulfilling overall competency requirements in general critical care US training (1,20). It is time to initiate a dedicated movement towards teaching lung US to every physician involved in the care of critically ill patients in academic centers around the world.
Acknowledgements
None.
Footnote
Conflicts of Interest: The author has no conflicts of interest to declare.
References
- Expert Round Table on Ultrasound in ICU. International expert statement on training standards for critical care ultrasonography. Intensive Care Med 2011;37:1077-83. [Crossref] [PubMed]
- Cholley BP, Vieillard-Baron A, Mebazaa A. Echocardiography in the ICU: time for widespread use! Intensive Care Med 2006;32:9-10. [Crossref] [PubMed]
- Kaplan A, Mayo PH. Echocardiography performed by the pulmonary/critical care medicine physician. Chest 2009;135:529-35. [Crossref] [PubMed]
- Charron C, Caille V, Jardin F, et al. Echocardiographic measurement of fluid responsiveness. Curr Opin Crit Care 2006;12:249-54. [Crossref] [PubMed]
- Repessé X, Charron C, Vieillard-Baron A. Acute respiratory distress syndrome: the heart side of the moon. Curr Opin Crit Care 2016;22:38-44. [Crossref] [PubMed]
- Biswas A. Right heart failure in acute respiratory distress syndrome: An unappreciated albeit a potential target for intervention in the management of the disease. Indian J Crit Care Med 2015;19:606. [Crossref] [PubMed]
- Lichtenstein D, Goldstein I, Mourgeon E, et al. Comparative diagnostic performances of auscultation, chest radiography, and lung ultrasonography in acute respiratory distress syndrome. Anesthesiology 2004;100:9-15. [Crossref] [PubMed]
- Volpicelli G, Mussa A, Garofalo G, et al. Bedside lung ultrasound in the assessment of alveolar-interstitial syndrome. Am J Emerg Med 2006;24:689-96. [Crossref] [PubMed]
- Bouhemad B, Liu ZH, Arbelot C, et al. Ultrasound assessment of antibiotic-induced pulmonary reaeration in ventilator-associated pneumonia. Crit Care Med 2010;38:84-92. [Crossref] [PubMed]
- Xirouchaki N, Kondili E, Prinianakis G, et al. Impact of lung ultrasound on clinical decision making in critically ill patients. Intensive Care Med 2014;40:57-65. [Crossref] [PubMed]
- Soummer A, Perbet S, Brisson H, et al. Ultrasound assessment of lung aeration loss during a successful weaning trial predicts postextubation distress. Crit Care Med 2012;40:2064-72. [Crossref] [PubMed]
- Bouhemad B, Brisson H, Le-Guen M, et al. Bedside ultrasound assessment of positive end-expiratory pressure–induced lung recruitment. Am J Respir Crit Care Med 2011;183:341-7. [Crossref] [PubMed]
- Caltabeloti F, Monsel A, Arbelot C, et al. Early fluid loading in acute respiratory distress syndrome with septic shock deteriorates lung aeration without impairing arterial oxygenation: a lung ultrasound observational study. Crit Care 2014;18:R91. [Crossref] [PubMed]
- DiNino E, Gartman EJ, Sethi JM, et al. Diaphragm ultrasound as a predictor of successful extubation from mechanical ventilation. Thorax 2014;69:423-7. [Crossref] [PubMed]
- Ferrari G, De Filippi G, Elia F, et al. Diaphragm ultrasound as a new index of discontinuation from mechanical ventilation. Crit Ultrasound J 2014;6:8. [Crossref] [PubMed]
- Lichtenstein DA. The LUCI-FLR Project: Lung Ultrasound in the Critically Ill – A Bedside Alternative to Irradiating Techniques, Radiographs and CT. In: Lung Ultrasound in the Critically Ill. Springer, Cham, 2016:217-25.
- Lichtenstein D, Peyrouset O. Is lung ultrasound superior to CT? The example of a CT occult necrotizing pneumonia. Intensive Care Med 2006;32:334-5. [Crossref] [PubMed]
- Lichtenstein D, Mezière G, Seitz J. The dynamic air bronchogram: a lung ultrasound sign of alveolar consolidation ruling out atelectasis. Chest 2009;135:1421-5. [Crossref] [PubMed]
- Lerolle N, Guérot E, Dimassi S, et al. Ultrasonographic diagnostic criterion for severe diaphragmatic dysfunction after cardiac surgery. Chest 2009;135:401-7. [Crossref] [PubMed]
- Georgopoulos D, Xirouchaki N, Volpicelli G. Lung ultrasound in the intensive care unit: let’s move forward. Springer, 2014.
- Lichtenstein DA, Menu Y. A bedside ultrasound sign ruling out pneumothorax in the critically III: lung sliding. Chest 1995;108:1345-8. [Crossref] [PubMed]
- Lichtenstein DA, Mezière G, Lascols N, et al. Ultrasound diagnosis of occult pneumothorax. Crit Care Med 2005;33:1231-8. [Crossref] [PubMed]
- Lichtenstein DA. Lung ultrasound in the critically ill. Ann Intensive Care 2014;4:1. [Crossref] [PubMed]
- Lichtenstein D, Hulot JS, Rabiller A, et al. Feasibility and safety of ultrasound-aided thoracentesis in mechanically ventilated patients. Intensive Care Med 1999;25:955-8. [Crossref] [PubMed]
- Doust BD, Baum JK, Maklad NF, et al. Ultrasonic evaluation of pleural opacities. Radiology. 1975;114:135-40. [Crossref] [PubMed]
- Chen KY, Liaw YS, Wang HC, et al. Sonographic septation: a useful prognostic indicator of acute thoracic empyema. J Ultrasound Med 2000;19:837-43. [Crossref] [PubMed]
- Vignon P, Chastagner C, Berkane V, et al. Quantitative assessment of pleural effusion in critically ill patients by means of ultrasonography. Crit Care Med 2005;33:1757-63. [Crossref] [PubMed]
- Remérand F, Dellamonica J, Mao Z, et al. Multiplane ultrasound approach to quantify pleural effusion at the bedside. Intensive Care Med 2010;36:656-64. [Crossref] [PubMed]
- Lichtenstein D. Lung ultrasound in the critically ill. Clin Intensive Care 2005;16:79-87. [Crossref]
- Lichtenstein D, Mezière G, Biderman P, et al. The" lung point": an ultrasound sign specific to pneumothorax. Intensive Care Med 2000;26:1434-40. [Crossref] [PubMed]
- Zhang Z, Chen L. A physiological sign that mimics lung point in critical care ultrasonography. Crit Care 2015;19:155. [Crossref] [PubMed]
- Weinberg B, Diakoumakis E, Kass E, et al. The air bronchogram: sonographic demonstration. AJR Am J Roentgenol 1986;147:593-5. [Crossref] [PubMed]
- Biswas A. Ultrasound study of the right lung in the anterior chest with a straight linear array probe (5–13 MHz) demonstrates sliding pleura with each breath. Asvide 2017;4:506. Available online: http://www.asvide.com/articles/1825
- Lichtenstein DA, Lascols N, Mezière G, et al. Ultrasound diagnosis of alveolar consolidation in the critically ill. Intensive Care Med 2004;30:276-81. [Crossref] [PubMed]
- Yang PC, Luh KT, Chang DB, et al. Ultrasonographic Evaluation of Pulmonary Consolidation1, 2. Am Rev Respir Dis 1992;146:757-62. [Crossref] [PubMed]
- Yu C-J, Yang P-C, Chang D, et al. Diagnostic and therapeutic use of chest sonography: value in critically ill patients. AJR Am J Roentgenol 1992;159:695-701. [Crossref] [PubMed]
- Yang PC, Chang DB, Yu CJ, et al. Ultrasound guided percutaneous cutting biopsy for the diagnosis of pulmonary consolidations of unknown aetiology. Thorax 1992;47:457-60. [Crossref] [PubMed]
- Yang PC, Luh KT, Lee YC, et al. Lung abscesses: US examination and US-guided transthoracic aspiration. Radiology 1991;180:171-5. [Crossref] [PubMed]
- Lichtenstein DA, Meziere GA. Relevance of lung ultrasound in the diagnosis of acute respiratory failure. Chest 2008;134:117-25. [Crossref] [PubMed]
- Cortellaro F, Colombo S, Coen D, et al. Lung ultrasound is an accurate diagnostic tool for the diagnosis of pneumonia in the emergency department. Emerg Med J 2012;29:19-23. [Crossref] [PubMed]
- Reissig A, Copetti R, Mathis G, et al. Lung ultrasound in the diagnosis and follow-up of community-acquired pneumonia: a prospective, multicenter, diagnostic accuracy study. Chest 2012;142:965-72. [Crossref] [PubMed]
- Lichtenstein DA, Lascols N, Prin S, et al. The “lung pulse”: an early ultrasound sign of complete atelectasis. Intensive Care Med 2003;29:2187-92. [Crossref] [PubMed]
- Lichtenstein D, Meziere G, Biderman P, et al. The comet-tail artifact: an ultrasound sign of alveolar-interstitial syndrome. Am J Respir Crit Care Med 1997;156:1640-6. [Crossref] [PubMed]
- Hasan AA, Makhlouf HA. B-lines: Transthoracic chest ultrasound signs useful in assessment of interstitial lung diseases. Ann Thorac Med 2014;9:99-103. [Crossref] [PubMed]
- Biswas A, Lascano JE, Mehta HJ, et al. The Utility of the “Shred Sign” in the Diagnosis of Acute Respiratory Distress Syndrome Resulting from Multifocal Pneumonia. Am J Respir Crit Care Med 2017;195:e20-e22. [Crossref] [PubMed]
- Reissig A, Heyne JP, Kroegel C. Sonography of lung and pleura in pulmonary embolism: sonomorphologic characterization and comparison with spiral CT scanning. Chest 2001;120:1977-83. [Crossref] [PubMed]
- Lichtenstein DA. BLUE-protocol and FALLS-protocol: two applications of lung ultrasound in the critically ill. Chest 2015;147:1659-70. [Crossref] [PubMed]
- Lichtenstein DA. How can the use of lung ultrasound in cardiac arrest make ultrasound a holistic discipline. The example of the SESAME-protocol. Med Ultrason 2014;16:252. [PubMed]
- Bouhemad B, Zhang M, Lu Q, et al. Clinical review: bedside lung ultrasound in critical care practice. Crit Care 2007;11:205. [Crossref] [PubMed]
- Mozzini C, Pasini AMF, Garbin U, et al. Lung ultrasound in internal medicine: training and clinical practice. Crit Ultrasound J 2016;8:10. [Crossref] [PubMed]
- Yang Y, Royse C, Royse A, et al. Survey of the training and use of echocardiography and lung ultrasound in Australasian intensive care units. Crit Care 2016;20:339. [Crossref] [PubMed]
Cite this article as: Biswas A. The role of bedside point-of-care ultrasound evaluation of the lung in the critically ill patient. J Emerg Crit Care Med 2017;1:34.


