Surgical interventions for severe traumatic brain injury
Introduction
Traumatic brain injury (TBI) is a worldwide threat, affecting specifically a younger population demographic. Results, including those from the Centers for Disease Control and Prevention (CDC), estimated that 2.8 million patients visited the emergency department, were hospitalized, or died due to TBI related in events in 2013 (1). The World Health Organization estimates that globally, TBI affects 10 million patients every year (2).
With a wide array of heterogeneity in etiology, presentation, pathophysiology, complexity, and outcome, caring for patients with TBI proves challenging. Programs in the “pre-injury” and pre-hospital setting have played an important role in primary injury prevention (3). Patient triage for initial resuscitative efforts and for definitive care after injury has been a topic of much debate (4-6). In the US the American College of Surgeons provides criteria that deem patients necessary for transfer between trauma facilities (7). With rapidly advancing technologies, namely with telemedicine, instantaneous inter-institutional communications and patient evaluations may be the key to pre-hospital management and patient triage within trauma systems of the future (8,9). Secondary injury prevention and management has been the main focus on patient care after TBI. Live with debate and controversy, medical management strategies, surgical indications and ethical implications continue to be studied with great interest (10).
We aim to highlight the current practice guidelines, review recent literature, and discuss surgical indications, outcomes, and short- and long-term socio-economic and ethical considerations following severe TBI.
Evaluation of the TBI patient
Evidence guided recommendations to avoid hypotension [systolic blood pressure (SBP) <90], to avoid hypoxemia (arterial O2 saturation <90%), and to establish the Glasgow Coma Scale (GCS) post-resuscitation to define neurological status and guide clinical progression have been outlined (3). Endotracheal intubation is indicated for patients with a GCS <9, or to gain adequate airway access. Only the patients showing signs of cerebral herniation should be hyperventilated to an ETCO2 of 30–35 mmHg (3) as a temporary measure of last resort.
Key in the initial steps of evaluating a TBI patient is to assess the severity of the injury itself. Following necessary cardiopulmonary resuscitation, the evaluation of TBI severity is essential for not only determining appropriate clinical decisions, but also for communication between hospital systems and multi-disciplinary teams. Numerous scales have been developed in an effort to rapidly communicate severity and ultimately predict outcomes (11-13). An advantage to standardized scoring systems is that it creates a common language across care systems and allows for data collection and multi-center investigations to provide valued care and improve outcomes based on studying these scores. A well-understood disadvantage is that these scoring systems can be confounded by variables such as multi-system trauma, sedative mediations, paralytics, illicit drug use, and seizures, which are all not uncommon in trauma settings. Therefore, the skill level of the examiner and timing of the assessment is critical, creating the subsequent issues of intra- and inter-examiner variability (14).
The GCS is one of the most widely accepted scales in neuro-trauma and was first described in 1974 by Teasdale and Jennett (11). It assigns points to eye [1–4], verbal [1–5] and [1–6] motor examinations to produce a score from 3–15. Severe TBI, as defined by the Brain Trauma Foundation (BTF), is a GCS of 3–8. The Injury Severity Score (ISS) and Abbreviated Injury Severity Score (AIS) are anatomic based grading scales. It has been suggested that an AIS ≥5 defines severe TBI (15). GCS combined with AIS have been found to predict 2-week mortality for TBI patients (16). These can be helpful in giving further prognosis in TBI, however carry the same advantages and disadvantages as other scoring systems.
Medical management for TBI
The BTF published guidelines for the management of severe TBI based on current evidence (10). In this latest 4th edition 2017 update, the following recommendations (and level of evidence) were re-visited: steroid use is not recommended for improving outcomes as it is associated with increased mortality and contraindicated (I); prolonged prophylactic hyperventilation for PaCO2 ≤25 mmHg is not recommended, but is recommended as a temporizing measure (IIB); prophylactic hypothermia is not recommended to improve outcome (IIB); continuous drainage from an external ventricular drain (EVD) may be more effective than intermittent use; basal caloric replacement is recommended before 7 days to reduce mortality (IIA); early tracheostomy may reduce ventilator days (IIA); early tracheostomy has not been shown to reduce nosocomial pneumonia or mortality in patients (IIA); anti-microbial catheters may prevent EVD-related infections (III); chemoprophylaxis for deep venous thrombosis (DVT) may be use at increased hemorrhage risk and there is no evidence for agent, dose, or timing (III); early post traumatic seizures (PTS) have not been shown to worsen outcome, but seizure prophylaxis with phenytoin may be used to help prevent PTS (IIA); no evidence to recommend levetiracetam vs. phenytoin (IIA). New Level IIB and III recommendations for monitoring and thresholds were also were made: severe TBI requires some form of intracranial pressure (ICP) monitoring, SBP thresholds based on age, treat ICP values >22 mmHg, ICP and radiographic studies should be used together for decision making, maintain CPP 60–70 mmHg but avoid aggressive CPP >70 mmHg.
Simple measures can also be taken, such as keeping the head in a neutral position and elevating the head of the bed 15 to 30 degrees to promote cerebral venous outflow and decrease intracranial venous congestion (17). Cervical collars may be loosened if possible. Further investigations and interventions should be made thereafter with the goal of decreasing the cerebral metabolic rate of oxygen consumption (CMRO2). Treating fevers and preventing seizures are two examples that apply these principles. This is also where the BTG guidelines and recommendations may be employed and executed effectively.
Role of initial imaging
The non-contrast computed tomography (CT) scan is indispensable when assessing the trauma patient. A CT scan of the head (CTH) provides the ability to quickly identify various traumatic pathologies including, epidural hematomas (EDH), subdural hematomas (SDH), subarachnoid hemorrhage, intraventricular hemorrhage, contusions, and severe deep white matter shear injuries. Clinical decisions can be made rapidly and neurosurgical intervention can be swiftly considered. Not every patient in the setting of reported trauma however should undergo a CTH and certain clinical correlations must be applied. Recommendations for acquiring scans can be based on the presenting risk factors. Low risk patients are asymptomatic, or have headaches, dizziness or present with superficial soft tissue scalp injuries. These low risk patients may not require a CTH (18-20). Criteria listed for patients who should have a CTH have been published (18,20). Vomiting, headaches, altered mental status, loss of consciousness, intoxication, PTSs, post traumatic amnesia, signs of a basal skull fracture, facial injury, and multiple traumatic injuries are amongst the factors considered. These types of criteria have the ability to improved valued care and reduce unnecessary radiation to the patients (21).
The timing of the CTH is also important. When assessing the trauma patient, the CTH provides only a snapshot in time. Depending upon the injury type, there may be a concern for progression of the insult. A study from UCLA demonstrated that progressive hemorrhagic injury (PHI) occurs in 50% of patients who undergo a CTH within 2 hours of their injury (22) suggesting that TBI patients that present with imaging obtained within the first 2 hours of presentation may not have completed their injury. A CTH taken during this timeframe may worsen significantly to warrant a change in management. The study found that intraparenchymal contusions were most likely to display PHI supporting the practice of a routine repeat scan to characterize progression. The recommendation from this study was to perform a repeat CT within 4 hours of the first CT, however other studies have reported different timing intervals (23). Some studies recommended no repeat CTH is indicated for patients with mild TBI and/or patients on anticoagulation without neurological changes (24,25). A systematic review revealed that up to 67% of TBI patients have progression on repeated CT scans (26). The utility of repeat imaging is evident in severe TBI, however specific timing and the number of CTH to be completed is still unclear. Repeat CTH completed due to inter-hospital transfers within trauma systems has also been evaluated (27). Even with hemorrhage progression with worsening radiographic findings, specific recommendations for surgical intervention are difficult to establish, and should be individualized, due to heterogeneous populations studied and results reported (26,28-30).
Surgery for severe TBI
The decision for surgical intervention for craniotomy is dependent on the injury type and the patient’s neurologic exam. Skull fractures, EDHs, SDHs, large vessel injuries, and intraparenchymal contusions trigger treatment specific algorithms, each supported by their respective data.
ICP monitoring
ICP monitoring is helpful to guide both medical and surgical decision-making. It is a marker to guide understanding of cerebral perfusion, but more specifically it is a function of intracranial compliance. The dynamics of perfusion and ICP are delineated in the Monroe-Kellie Hypothesis which posits that the skull is a rigid container of non-compressible contents: cerebrospinal fluid (CSF), blood, and brain tissue (31). When a new space-occupying lesion is introduced, the system requires displacement of another element to maintain a safe ICP. The BTF updates chose to renew their previous recommendations for ICP monitoring though there are no available studies to support any formal recommendations. Therefore, ICP monitoring should be performed in all salvageable patients, after cardiopulmonary resuscitation, with a GCS ≤8 with an abnormal CTH. It is also indicated for severe TBI with a normal CTH scan with two or more of the following criteria: age >40 years, motor posturing, or SBP <90 mmHg. For severe TBI, the TBF also report Level III recommendations for EVD, used as a CSF draining device, to help manage ICPs in patient who have a GCS ≤6 during the initial 12 hours after injury (as EVD placement in patients with a GCS >6 may increase mortality rates). Level IIB evidence indicates that ICPs above 22 mmHg were associated with increased mortality (10).
There are multiple tools to evaluate ICP in the TBI patient. Camino OLM ICP monitor (Integra LifeScience, Plainsboro, New Jersey, USA) is a fiberoptic cable attached to a microtransducer that is inserted into brain parenchyma through a bolt that is drilled and secured to the skull. This can be done at the bedside quickly in the trauma setting. Generally, this device is placed in the right frontal lobe to avoid areas of eloquence but it can be placed in any location to achieve the desired goal. Different than an intraparenchymal ICP monitor, a ventriculostomy (EVD) has both a diagnostic and therapeutic value. It can also be utilized to monitor pressure but it also can drain CSF to aid in ICP reduction. Kocher’s point (2–3 cm to the midline and 11 cm posterior to the nasion) on the right is often utilized to avoid both the sagittal sinus and the motor cortex. Subdural ICP monitors have also been shown to be effective and safe (32). Lastly, Licox (Integra LifeScience, Plainsboro, New Jersey, USA) monitor is also an option that employs the use of fiberoptics, microdialysis and sensors to measure multiple modalities with the goal of fine tuning management. In addition to measuring ICP, it allows monitoring of local brain tissue oxygen (PbO2). While studies have demonstrated that the use of ICP monitoring in conjunction with PbO2 monitoring and therapy directed at brain oxygenation reduces mortality following severe TBI. Others did not show any difference in outcome when PbO2 monitors were compared to EVDs (33,34).
Surgical decompression using craniotomies
Craniotomies may predate much of historical documentation. Archeologists have found evidence of trephined skulls that date as far back as 8000 BC (35). These trephinations were hypothesized to be a part of rituals or religious ceremonies. Over time, it became a proposed method of treating neurologic ailments such as headaches and dizziness. Hippocrates also suggested urgent craniotomies for TBI (35). During the Renaissance, as the use of firearms in battle grew and head trauma was surgically treated, the introduction of antibiotics, anesthetics and advances in hemostasis improved survival of those undergoing surgery and the practice of craniotomies was expanded. In 1914, the frontotemporal craniotomy was developed by George Heuer (36). This served as the launching pad for wider craniotomies to be performed such as bifrontal and biparietal craniotomies and revolutionized neurosurgery. The following guidelines are recommendations from the BTF and supporting literature (37,38).
Acute EDHs
The classic presentation of an acute EDH is a loss of consciousness followed by a lucid interval and obtundation in sequence, and a lens shaped hemorrhage on CT (Figure 1). The source of this hemorrhage is typically the middle meningeal artery, however, bleeding may occur from a sinus or the bone itself in the setting of a skull fracture. Indications for operative management are based on imaging and patient status. A volume greater than 30 cc should be evacuated regardless of GCS. If the volume is less than 30 cc or less than 15 mm in thickness on the CT scan or with less than 5 mm of midline shift and GCS greater than 8, the patient may be managed non-operatively. If the decision to proceed to the OR is made, it should be done as soon as possible especially in the setting of GCS <9 or anisocoria. There are no studies that support a single treatment method. A craniotomy, however, does provide more complete hematoma evacuation (39).
Acute SDHs
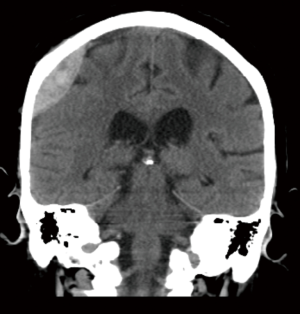
For patients presenting with an acute SDH the generally accepted indications for surgery is a thickness greater than 10 mm and midline shift greater than 5 mm on the CT scan (Figure 2). It is recommended that patients presenting with these findings be operated on regardless of their GCS. Furthermore, patients with a GCS less than 9 an acute SDH can warrant some form of ICP monitoring. This is due to the higher likelihood of underlying cerebral injury, parenchymal contusions, deep shear injury, or cortical injury, which relate to the mechanism of injury. Recommendations do not distinguish between intraparenchymal monitors, EVDs or Licox. Observation, medical management and repeat CTs are acceptable with an acute SDH less than 10 mm thickness or less than 5 mm of midline shift. However, should the GCS fall by 2 or more points from injury to presentation, the ICP rise to greater than 20 mmHg or if abnormal pupillary responses are observed, surgery is recommended. If the decision to proceed to surgery is made, the timing should be emergent. The preferred method is a craniotomy with or without bone flap removal or duraplasty depending on intraoperative findings (39). There is controversy with regards to performing a craniotomy or a craniectomy in the setting of acute SDH (40).
Traumatic parenchymal lesions
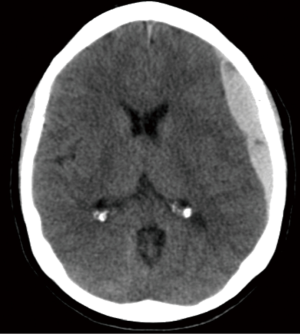
The parenchyma of the brain can be subject to injury and warrant surgical intervention as well. Indication for surgery in this type of injury is an intracerebral lesion with neurologic deterioration, and intracranial hypertension that is not responsive to medical management or signs of mass effect on CT (Figure 3). Furthermore, patients with a GCS less than 9 and a frontal or temporal lesion, with volume greater than 20 cc with midline shift greater than 5 mm, or cisternal compression or any lesion volume greater than 50 cc, may be considered for evacuation. Non-operative management can be considered when the parenchymal lesion causes no neurologic deficit, has no signs of mass effect and ICPs are controlled. The timing of proceeding to the OR should be emergent. Methods for surgical intervention depend on the location and nature of the injury. A craniotomy is generally recommended for any of these identified indications. If the patient is suffering from medically refractory intracranial hypertension, a bifrontal decompressive craniectomy (DC) may be considered. This will be reviewed in detail below. Other options for decompression include a temporal lobectomy, hemispheric DC and subtemporal decompression (41).
Depressed cranial fractures
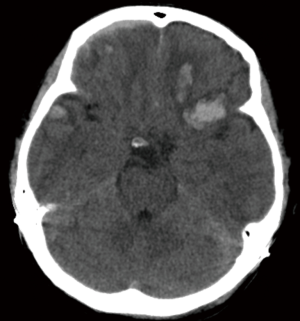
Certain types of skull fractures warrant surgical intervention. Indications for surgery include open skull fractures depressed to a degree greater than the thickness of the skull. These should be operated on to prevent infection and further damage to underlying brain parenchyma (Figure 4). At times, an open depressed skull fracture can be treated non-operatively if there is no evidence of dural penetration, no pneumocephalus, no significant intracranial hematoma, the depressed section is less than 1 cm, no involvement of the frontal sinus, no gross deformity and no wound infection or gross contamination are observed. Whether open depressed skull fractures are treated surgically, they should be treated with anti-epileptic drugs and antibiotics may be considered. Closed depressed skull fractures may be treated non-operatively. If the decision to proceed to the operating room is made, it should be done in a timely fashion, but is not necessarily emergent depending on the presentation and neurologic status. Methods for repair include elevation of the bone flap and debridement of the overlying tissue. Foreign objects if present should be removed and copious irrigation performed. If the fracture is open, complex and comminuted, the fractured cranium may be removed and the defect may be repaired later to help preventing subsequent infection. If the wound has no signs of gross contamination or infection, or if it was a closed fracture, using the original bone flap is an option (42).
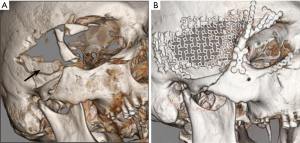
Penetrating trauma
This is a unique area of TBI in that penetrating trauma has no predilection for any particular age or group. In the civilian population, however, penetrating trauma to the head can take the form of a variety of objects, not limited to weapons such as firearms. Antibiotics and antiepileptic drugs should be started for these patients upon presentation. The primary indication for surgery is control of ICPs and secondary injury, but the removal of the penetrating foreign object may be considered. The injury itself most often includes the overlying skin, skull, dura, parenchyma, ventricles, cisterns and vascular structures. Great care must be made to determine the involvement of each of these structures in order to surgically manage these patients. Surgical intervention is rarely straightforward and case studies have outlined several considerations (43).
Gunshot wounds (GSW) to the head are a unique category of penetrating trauma (Figure 5). According to the CDC, they account for 35% of all deaths attributed to TBI. It is associated with a very high mortality with about 90% of patients dying before reaching the hospital. After presenting to the emergency room with aggressive resuscitation, about 50% die in the emergency room. Of those who survive, only about 5% go on to have any meaningful quality of life. Positive predictors of outcome for GSW to the head are presenting GCS >8, absence of an abnormal pupillary response, patency of basal cisterns and the trajectory of the bullet traversing fewer deep midline structures or brainstem (44,45). Surgery is not recommended for patients presenting with minimal neurologic function when not in shock or hypoxic (46). If the patient can be resuscitated and their blood pressure and oxygenation maintained, surgery can be considered with the primary goal of debridement, evacuation of hematoma and infection control. Retrieving the projectile is never the goal of surgery and is accomplished only as part of the superficial debridement. The timing of surgical intervention should be emergent. Options for surgical intervention include craniectomy to excise the damaged bone, evacuation of hematoma, dura repair and ICP monitoring. Patients suffering GSW to the head often suffer from secondary injury and an ICP monitor can be inserted to optimize management. Suspicion of vascular injury would necessitate vessel studies to identify and possibly repair traumatic aneurysms (47).
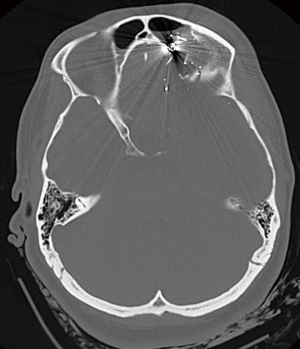
Decompressive craniectomies
Indications for DC)for TBI can plainly be described as sustained elevated ICPs not responsive to medical management (38). The patient’s injury type, clinical status, and intraoperative observations are the main determinants of surgical decision-making. By convention, decompressive craniectomies are noted as life-saving procedures to prevent further compounding secondary injures and central herniation (48). DCs are typically considered for diffuse brain injury vs. focal extra-axial or parenchymal injury. Open skull fractures, considered “dirty” may also be removed and left off for reasons other than for decompression (38). Severe TBI, by definition, with low GCS, brainstem involvement, and intraventricular involvement are associated with poor outcome with or without a DC (49-53). With such a wide array of both clinical and radiographic presentations with a high degree of patient-specific individuality, the role for DC in severe TBI is yet to be defined (Figure 6).
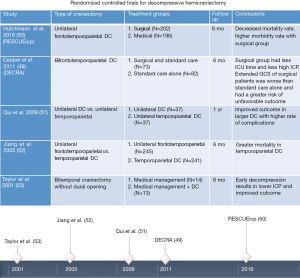
Within the last decade, two large multicenter randomized controlled trials evaluating the utility of DC in TBI patients are noteworthy (49,50). Military studies have also evaluated the role of DC and surgical evaluation for penetrating injuries, but are beyond the scope of this review. The DC in diffuse TBI (DECRA) trial evaluated outcomes after a bifrontotemporoparietal DC in 155 TBI patients (49). The study revealed a significant decrease in ICP, shorter ventilator needs, and a shorter ICU stay in the DC group. A few considerations must be noted. Only bifrontotemporoparietal DCs were evaluated. A significant number of patients with severe TBI were excluded from the statistical analysis. The patients in the DC group had worse pre-surgical clinical status and were the patients who had worse outcomes. Despite the noted patient crossover from patients initially intended for medical management to the DC group, intention-to-treat analysis was used for data reporting. This consideration may explain a more favorable outcome in the medically managed group as some did end up with a DC.
The trial of DC for traumatic intracranial hypertension (RESCUEicp) is another large multicenter randomized controlled trial, which evaluated the 6-month outcomes data of 398 severe TBI patients after medical management vs. DC (a unilateral frontotemporoparietal craniectomy) (50). A “favorable outcome,” was defined as an upper severe disability or better in the GOS-E scale. The authors acknowledged these concerns and furthermore explained similar definitions of outcome in stroke literature (54-56). These stroke articles also have similar criticisms (57). Contrary to DECRA, patients with mass lesions were not excluded from this study. The results demonstrated, a decreased mortality rate (26.9% vs. 48.9% deaths), but high morbidity rate (8.5%, 21.9%, 15.4%, 23.4%, vs. 2.1%, 14.4%, 8.0%, 19.7% vegetative state, lower severe, upper severe, and moderate disability) in the DC group vs. the intended medical arm.
From the current studies, it is reasonable to conclude there may be a role for DC in younger patients, but current conclusions leave much to be desired. It remains unclear which subset of patients benefit from a DC after severe TBI or if this subset of patients even exists. Not only the type of craniectomy, the laterality of the decompression, specific indications, pre-operative measures, and mass lesions, but the ethical and socio-economic factors must also be deliberated.
Ethics
Indications for craniotomies for various contributors of severe TBI have been supported by their respective literature. This information is what care providers wield when facing challenging family meetings to explain severity of injury and expected outcomes. And though the studies noted for DC do not supply hard-and-fast guidelines and protocols that aptly illustrate the role of DC, the data gathered has profound implications on family discussions and ethical and socio-economic considerations (50,57-59).
Primarily intended for further discussion on stroke outcomes, the Opinion Regarding Acceptable Outcome Following Decompressive Hemicraniectomy for Ischemic Stroke (ORACLE) study tackled many issues that are paralleled in the DC world for severe TBI (57). In the context of malignant cerebral edema, DC reduces mortality, but increases survivors who remain dependent with profound neurological disability. The ORACLE study aimed to determine opinions of healthcare providers in regards to acceptable opinions through a series of educational seminars and surveys. The article revealed that opinions of DC and subsequent outcomes change after more information can be provided and is considered. There are patients and families willing to accept a certain degree of disability over the entire mRS spectrum and this should be understood and respected. Most study participants did not find an mRS of ≥4 acceptable. The importance of religious and cultural beliefs, though variable, must also be strongly considered.
The financial burden on patient families and health care continue long after surgical interventions as increased hospital stays, rehabilitation and skilled nursing facility needs and partial or complete dependency increase expenses (60). Hospital costs were found to increase with TBI severity and older age (60,61). In an article published in 2011, Honeybul et al. discusses the “rule of rescue,” in neurotrauma, which tackles the challenges of financial risk against the innate human inclination of saving the life of another (62). An elegantly illustrated real-life conflict that stresses the appreciation of a wide spectrum of outcomes in between just life and death, which raises issues of social utility and equitable resource apportionment.
These individualized considerations must be continually analyzed and evidence-based literature should only serve as a basis to guide surgical indications and to facilitate discussions both between provider teams and patient families.
Conclusions
Despite the appreciated clinical relevance of surgical intervention for severe TBI to prevent secondary injury, sound, supporting evidence is lacking. In an injury type as devastating as severe TBI, large prospective randomized trials with standardization of interventions across multiple institutions, while considering the heterogeneity of various trauma systems, and establishing clinical equipoise is proving to be difficult. Even as life-saving surgical measures are taken, the ethical and socio-economic issues still remain as valued primary endpoints to consider.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Taylor CA, Bell JM, Breiding MJ, et al. Traumatic Brain Injury-Related Emergency Department Visits, Hospitalizations, and Deaths - United States, 2007 and 2013. MMWR Surveill Summ 2017;66:1-16. [Crossref] [PubMed]
- Hyder AA, Wunderlich CA, Puvanachandra P, et al. The impact of traumatic brain injuries: a global perspective. NeuroRehabilitation 2007;22:341-53. [PubMed]
- Badjatia N, Carney N, Crocco TJ, et al. Guidelines for prehospital management of traumatic brain injury 2nd edition. Prehosp Emerg Care 2008;12 Suppl 1:S1-52.
- Sasser SM, Hunt RC, Faul M, et al. Guidelines for field triage of injured patients: recommendations of the National Expert Panel on Field Triage, 2011. MMWR Recomm Rep 2012;61:1-20. [PubMed]
- Zhou Q, Rosengart MR, Billiar TR, et al. Factors Associated With Nontransfer in Trauma Patients Meeting American College of Surgeons' Criteria for Transfer at Nontertiary Centers. JAMA Surg 2017;152:369-76. [Crossref] [PubMed]
- Farach SM, Walford NE, Bendure L, et al. Helicopter Transport From the Scene of Injury: Are There Improved Outcomes for Pediatric Trauma Patients? Pediatr Emerg Care 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Rotondo MF, Cribari C, Smith RS. Interhospital transfer. Resour Optim Care Inj Patient Am Coll Surg 2014;30-4.
- Breen GM, Matusitz J. An evolutionary examination of telemedicine: a health and computer-mediated communication perspective. Soc Work Public Health 2010;25:59-71. [Crossref] [PubMed]
- Moya M, Valdez J, Yonas H, et al. The impact of a telehealth web-based solution on neurosurgery triage and consultation. Telemed J E Health 2010;16:945-9. [Crossref] [PubMed]
- Carney N, Totten AM, O’Reilly C, et al. Guidelines for the Management of Severe Traumatic Brain Injury, Fourth Edition. Neurosurgery 2017;80:6-15.
- Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet 1974;2:81-4. [Crossref] [PubMed]
- Zasler ND, Katz DI, Zafonte RD, et al. Brain Injury Medicine: Principles and Practice. 2nd Edition. New York: Demos Meical Publishing, 2012:529-44.
- Rating the severity of tissue damage. I. The abbreviated scale. JAMA 1971;215:277-80. [Crossref] [PubMed]
- Reith FC, Van den Brande R, Synnot A, et al. The reliability of the Glasgow Coma Scale: a systematic review. Intensive Care Med 2016;42:3-15. [Crossref] [PubMed]
- Savitsky B, Givon A, Rozenfeld M, et al. Traumatic brain injury: It is all about definition. Brain Inj 2016;30:1194-200. [Crossref] [PubMed]
- Timmons SD, Bee T, Webb S, et al. Using the abbreviated injury severity and Glasgow Coma Scale scores to predict 2-week mortality after traumatic brain injury. J Trauma 2011;71:1172-8. [Crossref] [PubMed]
- Rangel-Castilla L, Gopinath S, Robertson CS. Management of intracranial hypertension. Neurol Clin 2008;26:521-41. x. [Crossref] [PubMed]
- Stiell IG, Wells GA, Vandemheen K, et al. The Canadian CT Head Rule for patients with minor head injury. Lancet 2001;357:1391-6. [Crossref] [PubMed]
- Fong C, Chong W, Villaneuva E, et al. Implementation of a guideline for computed tomography head imaging in head injury: a prospective study. Emerg Med Australas 2008;20:410-9. [Crossref] [PubMed]
- Haydel MJ, Preston CA, Mills TJ, et al. Indications for computed tomography in patients with minor head injury. N Engl J Med 2000;343:100-5. [Crossref] [PubMed]
- Schmidt CW. CT scans: balancing health risks and medical benefits. Environ Health Perspect 2012;120:A118-21. [Crossref] [PubMed]
- Oertel M, Kelly DF, McArthur D, et al. Progressive hemorrhage after head trauma: predictors and consequences of the evolving injury. J Neurosurg 2002;96:109-16. [Crossref] [PubMed]
- Sullivan TP, Jarvik JG, Cohen WA. Follow-up of conservatively managed epidural hematomas: implications for timing of repeat CT. AJNR Am J Neuroradiol 1999;20:107-13. [PubMed]
- Sifri ZC, Homnick AT, Vaynman A, et al. A prospective evaluation of the value of repeat cranial computed tomography in patients with minimal head injury and an intracranial bleed. J Trauma 2006;61:862-7. [Crossref] [PubMed]
- Bauman ZM, Ruggero JM, Squindo S, et al. Repeat Head CT? Not Necessary for Patients with a Negative Initial Head CT on Anticoagulation or Antiplatelet Therapy Suffering Low-Altitude Falls. Am Surg 2017;83:429-35. [PubMed]
- Wang MC, Linnau KF, Tirschwell DL, et al. Utility of repeat head computed tomography after blunt head trauma: a systematic review. J Trauma 2006;61:226-33. [Crossref] [PubMed]
- Holmes JF, Siglock BG, Corwin MT, et al. Rate and Reasons for Repeat CT Scanning in Transferred Trauma Patients. Am Surg 2017;83:465-9. [PubMed]
- Park HK, Joo WI, Chough CK, et al. The clinical efficacy of repeat brain computed tomography in patients with traumatic intracranial haemorrhage within 24 hours after blunt head injury. Br J Neurosurg 2009;23:617-21. [Crossref] [PubMed]
- Kaups KL, Davis JW, Parks SN. Routinely repeated computed tomography after blunt head trauma: does it benefit patients? J Trauma 2004;56:475-80; discussion 480-1. [Crossref] [PubMed]
- Hill EP, Stiles PJ, Reyes J, et al. Repeat head imaging in blunt pediatric trauma patients: Is it necessary? J Trauma Acute Care Surg 2017;82:896-900. [Crossref] [PubMed]
- Mokri B. The Monro-Kellie hypothesis: applications in CSF volume depletion. Neurology 2001;56:1746-8. [Crossref] [PubMed]
- Hong WC, Tu YK, Chen YS, et al. Subdural intracranial pressure monitoring in severe head injury: clinical experience with the Codman MicroSensor. Surg Neurol 2006;66 Suppl 2:S8-S13. [Crossref] [PubMed]
- Martini RP, Deem S, Yanez ND, et al. Management guided by brain tissue oxygen monitoring and outcome following severe traumatic brain injury. J Neurosurg 2009;111:644-9. [Crossref] [PubMed]
- Green JA, Pellegrini DC, Vanderkolk WE, et al. Goal directed brain tissue oxygen monitoring versus conventional management in traumatic brain injury: an analysis of in hospital recovery. Neurocrit Care 2013;18:20-5. [Crossref] [PubMed]
- Sperati G. Craniotomy through the ages. Acta Otorhinolaryngol Ital 2007;27:151-6. [PubMed]
- Ormond DR, Hadjipanayis CG. The history of neurosurgery and its relation to the development and refinement of the frontotemporal craniotomy. Neurosurg Focus 2014;36:E12. [Crossref] [PubMed]
- Bullock MR, Chesnut R, Ghajar J, et al. Guidelines for the Surgical Management of Traumatic Brain Injury Author Group. Neurosurgery 2006;58:S2-vi. [Crossref]
- Grandhi R, Bonfield CM, Newman WC, et al. Surgical management of traumatic brain injury: a review of guidelines, pathophysiology, neurophysiology, outcomes, and controversies. J Neurosurg Sci 2014;58:249-59. [PubMed]
- Bullock MR, Chesnut R, Ghajar J, et al. Surgical management of acute epidural hematomas. Neurosurgery 2006;58:S7-15. discussion Si-iv. [PubMed]
- Phan K, Moore JM, Griessenauer C, et al. Craniotomy Versus Decompressive Craniectomy for Acute Subdural Hematoma: Systematic Review and Meta-Analysis. World Neurosurg 2017;101:677-85.e2. [Crossref] [PubMed]
- Bullock MR, Chesnut R, Ghajar J, et al. Surgical management of traumatic parenchymal lesions. Neurosurgery 2006;58:S25-46. discussion Si-iv. [Crossref] [PubMed]
- Bullock MR, Chesnut R, Ghajar J, et al. Surgical management of depressed cranial fractures. Neurosurgery 2006;58:S56-60. discussion Si-iv. [Crossref] [PubMed]
- Zyck S, Toshkezi G, Krishnamurthy S, et al. Treatment of Penetrating Nonmissile Traumatic Brain Injury. Case Series and Review of the Literature. World Neurosurg 2016;91:297-307. [Crossref] [PubMed]
- Aarabi B, Tofighi B, Kufera JA, et al. Predictors of outcome in civilian gunshot wounds to the head. J Neurosurg 2014;120:1138-46. [Crossref] [PubMed]
- Murano T, Mohr AM, Lavery RF, et al. Civilian craniocerebral gunshot wounds: an update in predicting outcomes. Am Surg 2005;71:1009-14. [PubMed]
- Kaufman HH. Civilian gunshot wounds to the head. Neurosurgery 1993;32:962-4; discussion 964. [Crossref] [PubMed]
- Bizhan A, Mossop C, Aarabi JA. Surgical management of civilian gunshot wounds to the head. Handb Clin Neurol 2015;127:181-93. [Crossref] [PubMed]
- Hofmeijer J, Kappelle LJ, Algra A, et al. Surgical decompression for space-occupying cerebral infarction (the Hemicraniectomy After Middle Cerebral Artery infarction with Life-threatening Edema Trial [HAMLET]): a multicentre, open, randomised trial. Lancet Neurol 2009;8:326-33. [Crossref] [PubMed]
- Cooper DJ, Rosenfeld JV, Murray L, et al. Decompressive craniectomy in diffuse traumatic brain injury. N Engl J Med 2011;364:1493-502. [Crossref] [PubMed]
- Hutchinson PJ, Kolias AG, Timofeev IS, et al. Trial of Decompressive Craniectomy for Traumatic Intracranial Hypertension. N Engl J Med 2016;375:1119-30. [Crossref] [PubMed]
- Qiu W, Guo C, Shen H, et al. Effects of unilateral decompressive craniectomy on patients with unilateral acute post-traumatic brain swelling after severe traumatic brain injury. Crit Care 2009;13:R185. [Crossref] [PubMed]
- Jiang JY, Xu W, Li WP, et al. Efficacy of standard trauma craniectomy for refractory intracranial hypertension with severe traumatic brain injury: a multicenter, prospective, randomized controlled study. J Neurotrauma 2005;22:623-8. [Crossref] [PubMed]
- Taylor A, Butt W, Rosenfeld J, et al. A randomized trial of very early decompressive craniectomy in children with traumatic brain injury and sustained intracranial hypertension. Childs Nerv Syst 2001;17:154-62. [Crossref] [PubMed]
- Vahedi K, Hofmeijer J, Juettler E, et al. Early decompressive surgery in malignant infarction of the middle cerebral artery: a pooled analysis of three randomised controlled trials. Lancet Neurol 2007;6:215-22. [Crossref] [PubMed]
- Puetz V, Campos CR, Eliasziw M, et al. Assessing the benefits of hemicraniectomy: what is a favourable outcome? Lancet Neurol 2007;6:580-author reply 580-1. [Crossref] [PubMed]
- Jüttler E, Unterberg A, Woitzik J, et al. Hemicraniectomy in older patients with extensive middle-cerebral-artery stroke. N Engl J Med 2014;370:1091-100. [Crossref] [PubMed]
- Honeybul S, Ho KM, Blacker DW. ORACLE Stroke Study: Opinion Regarding Acceptable Outcome Following Decompressive Hemicraniectomy for Ischemic Stroke. Neurosurgery 2016;79:231-6. [Crossref] [PubMed]
- Zhao HX, Liao Y, Xu D, et al. Prospective randomized evaluation of therapeutic decompressive craniectomy in severe traumatic brain injury with mass lesions (PRECIS): study protocol for a controlled trial. BMC Neurol 2016;16:1. [Crossref] [PubMed]
- Honeybul S, Ho KM, Lind CRP, et al. The current role of decompressive craniectomy for severe traumatic brain injury. J Clin Neurosci 2017;43:11-15. [Crossref] [PubMed]
- Ho KM, Honeybul S, Lind CR, et al. Cost-effectiveness of decompressive craniectomy as a lifesaving rescue procedure for patients with severe traumatic brain injury. J Trauma 2011;71:1637-44; discussion 1644. [Crossref] [PubMed]
- Alali AS, Naimark DM, Wilson JR, et al. Economic evaluation of decompressive craniectomy versus barbiturate coma for refractory intracranial hypertension following traumatic brain injury. Crit Care Med 2014;42:2235-43. [Crossref] [PubMed]
- Honeybul S, Gillett GR, Ho KM, et al. Neurotrauma and the rule of rescue. J Med Ethics 2011;37:707-10. [Crossref] [PubMed]
Cite this article as: Fong R, Konakondla S, Schirmer CM, Lacroix M. Surgical interventions for severe traumatic brain injury. J Emerg Crit Care Med 2017;1:28.